Revolutionizing Glycidyl Ester Production: Enhanced Purity and Scalability for Global Coatings Markets
The chemical landscape for high-performance coatings and resin systems is undergoing a significant transformation, driven by the demand for intermediates that offer superior stability and purity without compromising yield. Patent CN1325392A introduces a pivotal advancement in the synthesis of glycidyl esters of branched monocarboxylic acids, addressing long-standing challenges associated with color instability and heavy by-product formation. This technology represents a critical leap forward for manufacturers seeking a reliable glycidyl esters supplier capable of delivering consistent quality for epoxy polyester and polyurethane applications. By optimizing the reaction environment through precise control of solvent systems and catalyst loading, the process effectively suppresses the generation of colored impurities that traditionally plague this class of compounds. The implications for the global supply chain are profound, as this method enables the production of high-purity epoxy functional monomers with significantly reduced reliance on energy-intensive purification steps that historically eroded profit margins. As we delve into the technical specifics, it becomes clear that this innovation is not merely an incremental improvement but a fundamental rethinking of how branched carboxylic acid derivatives are manufactured for the modern coatings industry.
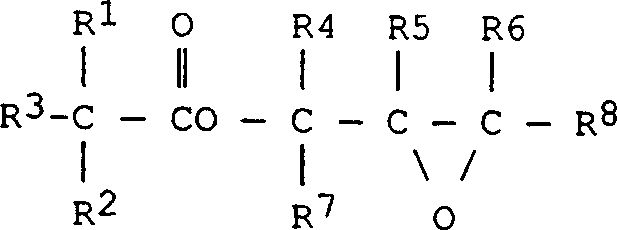
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of glycidyl esters has been hampered by the inevitable formation of heavier by-products, which typically constitute between 8% and 12% by weight of the crude reaction mixture. These impurities are chemically complex, often arising from the reaction of the desired glycidyl ester or chlorohydrin intermediates with unreacted epichlorohydrin, free acids, or water at various stages of synthesis. The presence of these species is detrimental not only to the initial color of the product but also to its thermal stability during storage and subsequent processing. Conventional attempts to rectify this issue have relied heavily on vacuum distillation techniques, which are inherently flawed for thermally sensitive molecules like glycidyl esters. The high temperatures required for distillation often trigger further reactions between the glycidyl functional groups and the existing impurities, leading to a darkening of the product that cannot be reversed. Furthermore, achieving high purity levels, such as 99%, through distillation alone has historically required sacrificing up to 30% of the theoretical yield, representing a massive inefficiency in raw material utilization and waste generation.
The Novel Approach
In stark contrast to the brute-force purification methods of the past, the process disclosed in CN1325392A adopts a preventative strategy by controlling the reaction pathway itself to minimize by-product formation at the source. This novel approach utilizes a specific combination of water and water-miscible solvents, such as aqueous alkanols, to create a reaction medium that facilitates optimal catalyst activity while suppressing side reactions. By carefully managing the molar ratios of alkali metal hydroxides and maintaining temperatures within a narrow window of 30°C to 110°C, the process ensures high conversion of the halogen-substituted epoxide without generating excessive heavy ends. The result is a crude product that already possesses a lightened initial color and a by-product content of less than 6% by weight, often dropping below 4% in preferred embodiments. This drastic reduction in impurity load means that the final purification steps can be much milder, avoiding the thermal stress that leads to discoloration and preserving the integrity of the valuable epoxy functionality required for downstream polymerization.
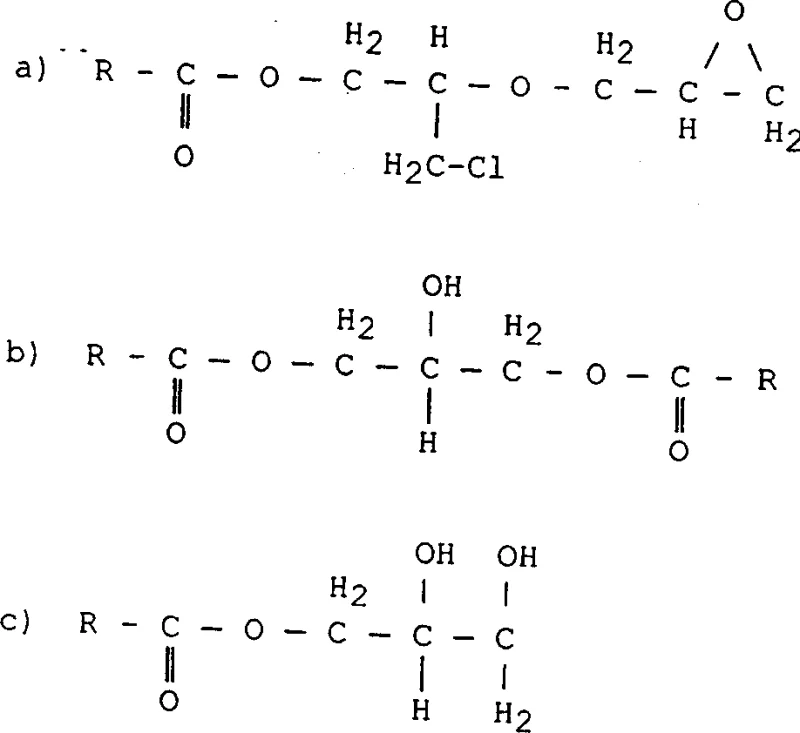
Mechanistic Insights into Catalytic Esterification and Dehydrohalogenation
The core of this technological breakthrough lies in the nuanced understanding of the catalytic cycle and the phase behavior of the reaction mixture. The process employs alkali metal hydroxides or alkoxides, preferably sodium or potassium hydroxide, in conjunction with a solvent system that balances solubility between the organic and aqueous phases. The use of isopropanol, for instance, allows for sufficient dissolution of the base into the organic phase where the reaction occurs, while the presence of water helps to manage the exotherm and solubilize the resulting salt by-products. This biphasic management is critical; it prevents the localized high concentrations of base that could otherwise lead to the hydrolysis of the newly formed glycidyl ester, a common side reaction that reduces yield and generates diols. The catalyst loading is kept relatively low, typically up to 45 mol% relative to the acid, which is sufficient to drive the esterification and subsequent ring closure without promoting excessive degradation. This precise balancing act ensures that the epichlorohydrin is selectively converted into the desired glycidyl ester structure rather than polymerizing or reacting to form the complex heavy by-products illustrated in the structural analysis.
Furthermore, the mechanism includes a strategic dehydrohalogenation step that is decoupled from the initial esterification, allowing for independent optimization of each stage. In the second stage, the addition of concentrated alkali metal hydroxide solution under anhydrous or low-water conditions completes the cyclization of the chlorohydrin intermediate to the epoxide. The patent highlights that maintaining low water content during this specific phase is crucial to prevent hydrolysis of the sensitive oxirane ring. The subsequent washing steps are designed to efficiently remove the alkali metal halide salts formed during the reaction, which is essential for achieving the low conductivity and high purity required for electronic or high-performance coating applications. By separating the salt removal from the reaction phase through phase separation and washing, the process avoids the need for filtration of solid salts, which can be a bottleneck in large-scale continuous manufacturing. This mechanistic clarity provides R&D teams with a robust framework for scaling the process while maintaining tight control over the impurity profile.
How to Synthesize Glycidyl Esters Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for replicating these high-quality results in a pilot or production setting. The procedure begins with the charging of the alpha-branched carboxylic acid and a significant excess of epichlorohydrin into a reactor equipped for temperature control and distillation. The addition of the solvent system and the initial catalyst dose initiates the esterification, which is monitored by tracking the consumption of the acid or the formation of the sodium salt. Once the initial reaction is complete, the mixture is cooled, and the aqueous phase containing the bulk of the salts is separated. The organic phase is then subjected to a second treatment with base to close the epoxide ring, followed by a rigorous washing sequence to ensure all ionic residues are removed. Detailed standardized synthesis steps see the guide below.
- React alpha-branched monocarboxylic acid with excess halogen-substituted monoepoxide in a water-miscible solvent system with controlled catalyst loading.
- Add alkali metal hydroxide or alkoxide to achieve a specific molar ratio and react at moderate temperatures to form the ester intermediate.
- Distill off excess epoxide and solvent, then treat the residue with concentrated base to complete dehydrohalogenation and wash to remove salts.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this advanced synthesis route offers tangible benefits that extend far beyond simple technical specifications. The primary value proposition lies in the substantial cost reduction in coatings intermediate manufacturing achieved through yield preservation. By eliminating the need for aggressive distillation cuts that historically discarded up to 30% of the product to achieve high purity, this process maximizes the output from every kilogram of raw material purchased. This efficiency translates directly into a more competitive cost structure, allowing buyers to secure high-purity epoxy functional monomers without the premium pricing typically associated with ultra-low impurity grades. Additionally, the simplified purification workflow reduces the consumption of utilities such as steam and cooling water, further lowering the operational expenditure associated with production. The ability to produce material with superior color stability right out of the reactor also minimizes the risk of batch rejection due to off-spec color, ensuring a more predictable and reliable supply stream for downstream formulators.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the elimination of yield-losing purification steps. Conventional methods often require sacrificing significant portions of the batch to remove heavy ends, whereas this novel approach suppresses their formation chemically. This means that the effective cost per unit of pure product is drastically lowered, as less raw material is wasted in the form of distillation bottoms. Furthermore, the reduced need for extensive thermal processing lowers energy costs, contributing to a leaner manufacturing model that is resilient against fluctuating energy prices. The qualitative improvement in process efficiency allows for a more sustainable pricing model that can be passed down the supply chain.
- Enhanced Supply Chain Reliability: From a logistics perspective, the robustness of this synthesis method enhances supply continuity. The process is less sensitive to minor variations in raw material quality due to its self-correcting washing and separation steps, reducing the frequency of production delays caused by off-spec inputs. The ability to operate in both batch and continuous modes provides flexibility to scale production up or down in response to market demand without compromising product integrity. This adaptability ensures that partners can rely on consistent delivery schedules, mitigating the risks associated with production bottlenecks that often plague complex chemical syntheses involving sensitive intermediates.
- Scalability and Environmental Compliance: The environmental footprint of the manufacturing process is significantly improved, aligning with modern sustainability goals. By reducing the volume of heavy by-products and chlorine-containing waste, the process simplifies effluent treatment and lowers the burden on waste management infrastructure. The avoidance of high-temperature distillation also reduces the potential for volatile organic compound (VOC) emissions associated with thermal degradation. This cleaner production profile facilitates easier regulatory compliance in jurisdictions with strict environmental standards, ensuring long-term operational viability and reducing the risk of shutdowns due to non-compliance issues.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this glycidyl ester production technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for decision-making. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this intermediate into their existing resin formulations or production lines. The answers reflect the specific advantages of the catalytic system and purification strategy employed in this method.
Q: How does this process improve color stability compared to conventional distillation?
A: By minimizing the formation of heavy by-products during the reaction phase through specific solvent and catalyst control, the need for aggressive distillation is reduced, preventing thermal degradation that causes discoloration.
Q: What are the key raw materials for producing these glycidyl esters?
A: The primary inputs are alpha-branched monocarboxylic acids (such as Versatic acids), epichlorohydrin, and alkali metal hydroxides, utilizing water-miscible solvents like isopropanol.
Q: Can this method be scaled for industrial production of coating resins?
A: Yes, the process is designed for both batch and continuous modes, offering high conversion rates and simplified purification steps suitable for large-scale manufacturing of epoxy-functional monomers.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Glycidyl Esters Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced intermediates requires a partner with deep technical expertise and proven manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising results seen in patent literature can be reliably translated into industrial reality. We maintain stringent purity specifications and operate rigorous QC labs to verify that every batch of glycidyl esters meets the exacting standards required for high-performance coatings and adhesives. Our commitment to quality assurance means that clients receive material with consistent epoxy content and minimal color variation, enabling them to formulate products with predictable performance characteristics.
We invite you to engage with our technical procurement team to discuss how this innovative process can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits of switching to our high-efficiency grade. We encourage you to contact us to obtain specific COA data and route feasibility assessments tailored to your specific application needs. Whether you are developing next-generation automotive clear coats or durable industrial finishes, our expertise in complex organic synthesis positions us as the ideal partner to support your growth and innovation objectives in the global market.
