Advanced Oxidant-Assisted Nitration For Commercial Scale-Up Of Complex Nitroalkenes
Advanced Oxidant-Assisted Nitration For Commercial Scale-Up Of Complex Nitroalkenes
The chemical landscape of pharmaceutical and agrochemical synthesis is constantly evolving, driven by the need for more efficient, cost-effective, and environmentally sustainable manufacturing processes. A pivotal advancement in this domain is documented in patent CN1232480C, which details a novel process for the preparation of nitroalkenes, specifically conjugated beta-nitroalkenes. These compounds serve as versatile synthetic intermediates, crucial for the production of phenethylamines, fungicides, and complex central nervous system (CNS) therapeutics. The traditional methods for nitrating conjugated alkenes have long been plagued by inefficiencies, particularly regarding the stoichiometric usage of expensive reagents and the generation of difficult-to-remove byproducts. This patent introduces a transformative approach by incorporating an oxidizing agent into the reaction mixture of inorganic nitrite and iodine. This strategic modification allows for a dramatic reduction in the amount of iodine required, shifting the process from a wasteful stoichiometric consumption to a more catalytic-like cycle. For industry leaders, this represents not just a chemical improvement, but a significant opportunity for supply chain optimization and cost reduction in pharmaceutical intermediate manufacturing.
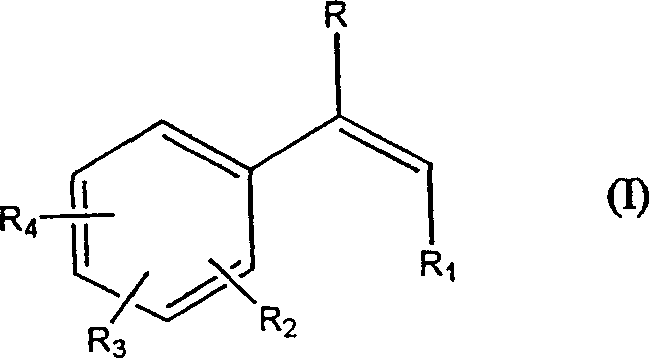
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the nitration of alkenes using nitrite and iodine has been a standard procedure in organic synthesis, yet it suffers from inherent economic and operational drawbacks. Literature precedents, such as those by Hassner and later modifications by Sy and Jew, typically rely on the use of silver nitrite or large excesses of sodium nitrite paired with substantial amounts of iodine. In many reported protocols, the molar ratio of iodine to alkene can exceed 2.75:1. This high loading of iodine creates a significant bottleneck in the downstream processing phase. At the conclusion of the reaction, the reaction mixture contains a large quantity of unreacted iodine and iodide salts. Removing this excess iodine necessitates a dedicated quenching step, usually involving the addition of bisulfite salts to reduce iodine to water-soluble iodide. This additional step increases the volume of wastewater, complicates the isolation of the organic product, and introduces potential impurities that can affect the purity profile of the final active pharmaceutical ingredient (API) intermediate. Furthermore, the reliance on silver nitrite in earlier methods imposes a heavy financial burden due to the high cost of silver salts and the regulatory complexities associated with heavy metal recovery and disposal.
The Novel Approach
The innovation presented in patent CN1232480C fundamentally alters the reaction dynamics by introducing an oxidizing agent into the standard nitrite-iodine system. This addition enables the in situ regeneration of molecular iodine from the iodide byproducts formed during the nitration event. Consequently, the initial charge of iodine can be significantly reduced to equal to or lower than 1 mole per mole of substrate, with a preferred range of 0.1 to 0.8 moles. This reduction transforms the role of iodine from a stoichiometric reagent to a quasi-catalytic mediator. The practical implication is a streamlined workup procedure where the need for extensive bisulfite treatment is either minimized or entirely avoided, depending on the residual color requirements. The process utilizes readily available inorganic nitrites, such as sodium nitrite, which are far more economical than their silver counterparts. By operating under mild acidic conditions with oxidants like peracetic acid or commercial peroxide mixtures, the method achieves high yields while maintaining a cleaner reaction profile. This approach directly addresses the productivity losses associated with high molecular weight iodine usage, offering a robust pathway for the synthesis of high-purity OLED material and pharmaceutical precursors.
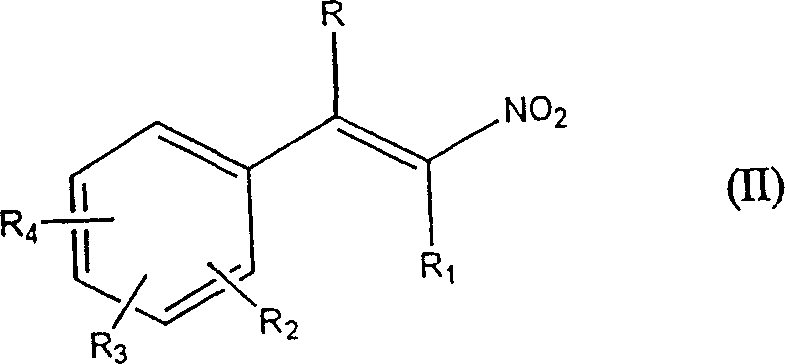
Mechanistic Insights into Oxidant-Assisted Nitration
The core mechanistic advantage of this process lies in the redox cycle facilitated by the oxidizing agent. In the conventional pathway, iodine reacts with the alkene and nitrite to form the nitroalkene and iodide ions. Without an oxidant, these iodide ions accumulate as waste. In the patented method, the added oxidizing agent, such as peracetic acid or hydrogen peroxide in an acidic medium, actively oxidizes the generated iodide ions back into molecular iodine. This regenerated iodine is then available to participate in further nitration cycles. This continuous regeneration loop means that a small initial amount of iodine can facilitate the conversion of a large amount of substrate. The reaction is typically conducted in organic solvents like esters, aromatic hydrocarbons, or chlorinated hydrocarbons, which ensure good solubility of the conjugated alkene substrates. The temperature is maintained between 20°C and 70°C, with a preferred range of 40°C to 50°C, ensuring that the reaction kinetics are favorable without promoting thermal decomposition or side reactions. The acidic environment, maintained at a pH below 5, is critical for the activation of the oxidizing agent and the stabilization of the nitrosating species. This precise control over reaction parameters ensures that the impurity profile remains tight, which is a critical consideration for R&D directors focusing on the purity and杂质谱 of complex intermediates.
Furthermore, the versatility of this mechanism allows for the nitration of a wide range of conjugated alkenes, including styrenes, dihydronaphthalenes, and benzopyrans. The substituent tolerance is high, accommodating various functional groups such as halogens, alkoxy groups, and carbonyls on the aromatic ring. For instance, the synthesis of 8-fluoro-3-nitro-2H-chromene-5-carboxamide, a key intermediate for CNS drugs, benefits significantly from this method. The mild conditions prevent the hydrolysis of sensitive amide or ester groups that might occur under harsher nitrating conditions. The ability to use sodium nitrite instead of silver nitrite also eliminates the risk of silver contamination, which is a stringent requirement for many pharmaceutical applications. The impurity control mechanism is further enhanced by the reduced formation of iodinated byproducts, which are common side reactions when excess iodine is present. By keeping the iodine concentration low through oxidative regeneration, the selectivity for the desired beta-nitroalkene is maximized. This level of control is essential for the commercial scale-up of complex polymer additives and fine chemicals where batch-to-b一致性 is paramount.
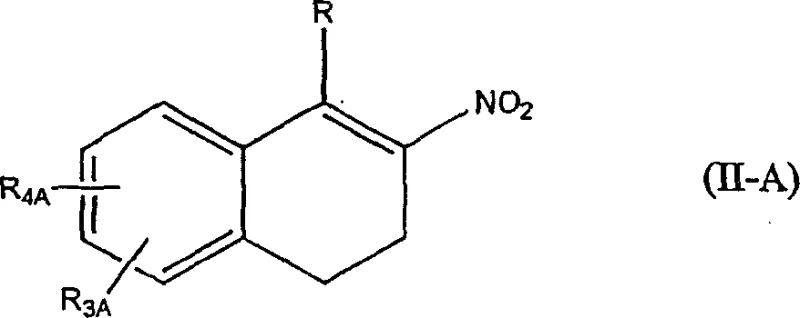
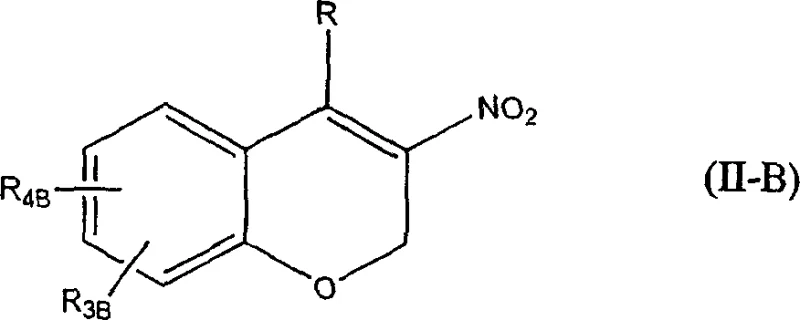
How to Synthesize Beta-Nitroalkenes Efficiently
The implementation of this synthesis route requires careful attention to the addition rates and stoichiometry to maximize the benefits of the oxidant cycle. The process begins with the suspension or solution of the conjugated alkene and sodium nitrite in a suitable solvent, followed by the addition of the reduced iodine charge. The oxidizing agent is then added slowly, typically over a period of 3 to 4 hours, to maintain a steady concentration of active iodine without causing a runaway exotherm. This controlled addition is key to managing the reaction safety and ensuring complete conversion. Detailed standardized synthesis steps see the guide below.
- Prepare a reaction mixture containing the conjugated alkene substrate, excess inorganic nitrite (preferably sodium nitrite), and a reduced amount of iodine in a suitable organic solvent.
- Slowly add an oxidizing agent solution, such as peracetic acid or a commercial peroxide mixture, over a period of 3 to 4 hours while maintaining the temperature between 20°C and 70°C.
- Upon completion, optionally treat with bisulfite to remove residual iodine color, separate phases, and isolate the beta-nitroalkene product through standard crystallization or extraction techniques.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the technical improvements in this patent translate directly into tangible business value. The primary advantage is the substantial cost savings achieved through raw material optimization. By reducing the iodine requirement from stoichiometric excess to sub-stoichiometric levels, the direct material cost per kilogram of product is significantly lowered. Iodine is a relatively expensive halogen, and minimizing its usage has a compounding effect on the overall cost of goods sold (COGS). Additionally, the switch from silver nitrite to sodium nitrite removes a high-cost precious metal from the bill of materials, further enhancing the economic viability of the process. These savings are not merely theoretical; they are rooted in the fundamental stoichiometry of the reaction. The simplified workup procedure also reduces the consumption of quenching agents like bisulfite and decreases the volume of wastewater that requires treatment. This leads to lower operational expenditures related to waste disposal and environmental compliance, which are increasingly critical cost drivers in the chemical industry.
- Cost Reduction in Manufacturing: The elimination of excess iodine and expensive silver salts directly lowers the raw material expenditure. The process avoids the need for complex heavy metal removal steps, which often require specialized resins or extraction protocols that add time and cost to the manufacturing cycle. By streamlining the reaction to use common, bulk chemicals like sodium nitrite and peracetic acid, the process becomes more resilient to price fluctuations in specialty reagents. This stability allows for more accurate long-term budgeting and pricing strategies for downstream customers. The reduction in waste treatment costs further contributes to the overall economic efficiency, making the production of high-purity pharmaceutical intermediates more competitive in the global market.
- Enhanced Supply Chain Reliability: The reagents required for this process, such as sodium nitrite, iodine, and peracetic acid, are commodity chemicals with robust global supply chains. Unlike specialized catalysts or precious metal salts that may face supply constraints or long lead times, these materials are readily available from multiple suppliers. This availability reduces the risk of production delays due to raw material shortages. Furthermore, the simplified process flow reduces the dependency on complex unit operations, making the manufacturing line less prone to bottlenecks. For supply chain heads, this means a more predictable production schedule and the ability to respond quickly to changes in market demand. The reliability of the supply is further bolstered by the process's tolerance to variations in raw material quality, ensuring consistent output even when sourcing from different vendors.
- Scalability and Environmental Compliance: The mild reaction conditions and reduced hazard profile make this process highly scalable from pilot plant to commercial production. The lower exothermic potential associated with reduced iodine loads enhances process safety, a critical factor for large-scale operations. From an environmental perspective, the reduction in iodine waste and the elimination of silver discharge align with green chemistry principles and stringent environmental regulations. This compliance reduces the regulatory burden and the risk of fines or shutdowns due to environmental violations. The ability to produce complex nitroalkenes with a smaller environmental footprint is increasingly becoming a key differentiator for suppliers seeking to partner with multinational corporations that have aggressive sustainability goals. This process facilitates the commercial scale-up of complex intermediates while maintaining a responsible environmental stance.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this oxidant-assisted nitration technology. These answers are derived directly from the patent specifications and are intended to clarify the operational benefits and feasibility for potential partners. Understanding these details is crucial for making informed decisions about adopting this technology for your specific production needs.
Q: How does the oxidant-assisted method reduce iodine consumption compared to conventional nitration?
A: Conventional methods often require stoichiometric or excess amounts of iodine (up to 2.75 moles per mole of alkene), necessitating complex workups to remove residual iodine. The patented oxidant-assisted process regenerates iodine in situ from iodide byproducts, allowing the initial iodine charge to be reduced to catalytic or sub-stoichiometric levels (0.1 to 0.8 moles), significantly simplifying purification.
Q: What are the primary commercial advantages of using sodium nitrite over silver nitrite in this process?
A: Historically, silver nitrite was preferred for solubility and reactivity but incurs high raw material costs and generates heavy metal waste. This process validates the use of inexpensive sodium nitrite in conjunction with an oxidizing agent, drastically lowering raw material costs and eliminating the environmental burden associated with silver waste disposal, thereby enhancing overall process sustainability.
Q: Is this nitration method scalable for industrial production of CNS drug intermediates?
A: Yes, the process operates under mild thermal conditions (40°C to 50°C) and uses commercially available oxidants like peracetic acid. The reduction in exothermic risk associated with lower iodine loads and the simplified workup procedure make it highly suitable for large-scale manufacturing of critical intermediates such as 8-fluoro-3-nitro-2H-chromene-5-carboxamide.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Nitroalkenes Supplier
The technological breakthroughs detailed in patent CN1232480C underscore the potential for more efficient and sustainable chemical manufacturing. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate such innovative laboratory processes into robust commercial realities. As a leading CDMO partner, we have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped to handle the specific requirements of oxidant-assisted reactions, ensuring that the benefits of reduced iodine usage and simplified workups are fully realized at scale. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of nitroalkenes meets the exacting standards required by the pharmaceutical and agrochemical industries. Our commitment to quality and efficiency makes us an ideal partner for companies looking to optimize their supply chain for critical intermediates.
We invite you to explore how our capabilities can enhance your production efficiency and reduce your overall manufacturing costs. By leveraging our expertise in process optimization, we can help you achieve a Customized Cost-Saving Analysis tailored to your specific product portfolio. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments for your target molecules. Whether you require small quantities for R&D or large volumes for commercial launch, our team is ready to provide the support and reliability you need to succeed in a competitive market. Let us collaborate to bring your next generation of products to market faster and more efficiently.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
