Optimizing Antiviral Intermediate Production via One-Pot Pyran-4-One Synthesis
The pharmaceutical industry continuously seeks robust synthetic pathways for critical intermediates that serve as the backbone for life-saving medications. A significant advancement in this domain is detailed in patent CN111606879A, which discloses a novel one-pot method for preparing 2-hydroxymethyl-3-alkoxy-4H-pyran-4-one. This specific chemical scaffold is not merely an academic curiosity; it serves as a pivotal precursor for the synthesis of 3-alkoxy-4-oxo-4H-pyran-2-carboxylic acid, a key building block for potent antiviral agents. The strategic importance of this molecule cannot be overstated, as it directly feeds into the supply chains for major therapeutics such as Dolutegravir, used in HIV treatment, and Baloxavir marboxil, a breakthrough influenza medication. 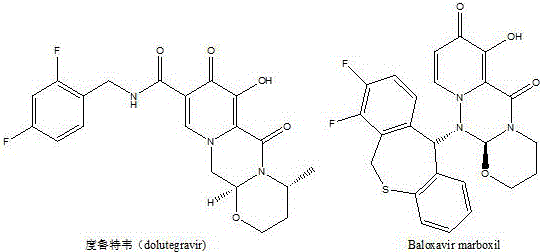 By optimizing the production of this intermediate, manufacturers can indirectly enhance the availability and affordability of these essential global health resources. The technical breakthrough lies in the ability to bypass traditional bottlenecks, offering a streamlined approach that aligns perfectly with the demands of modern, high-efficiency pharmaceutical manufacturing.
By optimizing the production of this intermediate, manufacturers can indirectly enhance the availability and affordability of these essential global health resources. The technical breakthrough lies in the ability to bypass traditional bottlenecks, offering a streamlined approach that aligns perfectly with the demands of modern, high-efficiency pharmaceutical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-hydroxymethyl-3-alkoxy-4H-pyran-4-one has been plagued by inefficiencies inherent in multi-step batch processing. Traditional protocols, as referenced in prior art such as U.S. Pat. No. 4,633,535 and others, typically mandate a disjointed two-step sequence. In the first stage, pyromeconic acid reacts with formaldehyde under alkaline conditions to generate an intermediate, which must then be laboriously isolated. This isolation involves acidification to precipitate the product, followed by filtration, washing, and often recrystallization to achieve necessary purity levels. 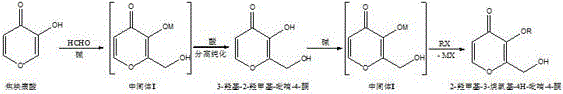 This conventional workflow introduces multiple points of failure and material loss. Each transfer and purification step inevitably leads to yield erosion, while the repeated cycling of pH from basic to acidic and back to basic consumes excessive amounts of reagents. Furthermore, the intermediate itself can be sensitive, and prolonged exposure to isolation conditions may lead to degradation or oxidation, compromising the quality of the final API intermediate. These operational complexities translate directly into higher production costs and extended lead times, creating friction in the supply chain for downstream drug manufacturers.
This conventional workflow introduces multiple points of failure and material loss. Each transfer and purification step inevitably leads to yield erosion, while the repeated cycling of pH from basic to acidic and back to basic consumes excessive amounts of reagents. Furthermore, the intermediate itself can be sensitive, and prolonged exposure to isolation conditions may lead to degradation or oxidation, compromising the quality of the final API intermediate. These operational complexities translate directly into higher production costs and extended lead times, creating friction in the supply chain for downstream drug manufacturers.
The Novel Approach
In stark contrast, the methodology outlined in CN111606879A introduces a paradigm shift through a seamless one-pot tandem reaction strategy. This innovative approach recognizes that both the initial hydroxymethylation and the subsequent alkylation steps fundamentally operate under basic conditions and proceed through similar anionic intermediates. By leveraging this chemical compatibility, the new process eliminates the need to isolate the 3-hydroxy-2-hydroxymethyl-pyran-4-one intermediate. Instead, once the initial reaction with formaldehyde is complete, the alkylating agent is introduced directly into the same reaction vessel. This continuity prevents the material loss associated with physical separation and avoids the wasteful consumption of acids and bases required for pH swinging. The result is a significantly shortened production cycle where the reaction proceeds from starting material to final product with minimal intervention. This simplification not only enhances the overall yield but also drastically reduces the environmental footprint by minimizing solvent usage and waste generation, representing a substantial leap forward in green chemistry principles for fine chemical synthesis.
Mechanistic Insights into Base-Catalyzed One-Pot Alkylation
To fully appreciate the technical elegance of this synthesis, one must delve into the mechanistic underpinnings that allow for such seamless integration. The reaction initiates with the deprotonation of pyromeconic acid by a strong base, such as sodium methoxide or sodium hydroxide, generating a reactive pyromeconic anion. This nucleophilic species attacks the electrophilic carbon of formaldehyde, facilitating the introduction of the hydroxymethyl group at the 2-position of the pyran ring. Crucially, the reaction environment remains alkaline throughout this phase, stabilizing the intermediate as an anionic species rather than allowing it to revert to the neutral, less reactive phenol form. In traditional methods, this anionic state is destroyed by acidification to isolate the solid intermediate. However, in this novel one-pot design, the basic environment is maintained, preserving the reactivity of the system. When the alkylating reagent, such as benzyl chloride or methyl iodide, is added, the anionic oxygen at the 3-position is perfectly poised for nucleophilic substitution. This direct transition avoids the kinetic penalty of re-dissolving and re-deprotonating the intermediate, ensuring a rapid and efficient conversion to the final 2-hydroxymethyl-3-alkoxy-4H-pyran-4-one structure.
From an impurity control perspective, this mechanism offers distinct advantages that are critical for R&D directors focused on product quality. By avoiding the isolation step, the process minimizes the exposure of the reactive intermediate to potential oxidants present in the air or during workup procedures. The patent notes that the hydroxymethyl group is susceptible to oxidation, which can lead to the formation of carboxylic acid byproducts. The one-pot method reduces the handling time and surface area exposure, thereby suppressing these oxidative side reactions. Additionally, the absence of acidification steps prevents the formation of acid-induced degradation products that might arise from the instability of the pyranone ring under low pH conditions. The consistent solvent system, whether it be methanol, ethanol, or a mixed aqueous-organic phase, ensures homogeneous reaction conditions that further promote selectivity. This controlled environment results in a cleaner crude profile, simplifying downstream purification and ensuring that the final intermediate meets the stringent purity specifications required for GMP pharmaceutical production.
How to Synthesize 2-Hydroxymethyl-3-Alkoxy-4H-Pyran-4-One Efficiently
Implementing this advanced synthetic route requires precise control over reaction parameters to maximize the benefits of the one-pot design. The process begins by dissolving pyromeconic acid in a suitable polar solvent system, which may include alcohols like methanol or ethanol, potentially mixed with water to optimize solubility and cost. The concentration of the starting material is typically maintained between 0.1 to 5 mol/L to balance reaction kinetics with heat management. Upon addition of the base, formaldehyde is introduced to effect the hydroxymethylation. Once this conversion is confirmed, the alkylating agent is added directly without any quenching or separation. The detailed standardized operating procedures, including specific temperature ramps, addition rates, and safety protocols for handling alkylating agents, are critical for successful scale-up. Adhering to these optimized conditions ensures that the theoretical advantages of the one-pot method are realized in practical manufacturing settings, delivering consistent quality and performance.
- Dissolve pyromeconic acid in a polar solvent system and react with formaldehyde under basic conditions to form the hydroxymethyl intermediate.
- Without isolation or acidification, directly introduce the alkylating reagent into the same reaction vessel.
- Maintain basic conditions to facilitate nucleophilic substitution, yielding the final 2-hydroxymethyl-3-alkoxy-4H-pyran-4-one product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this one-pot synthesis methodology represents a strategic opportunity to optimize cost structures and enhance supply reliability. The primary economic driver here is the drastic simplification of the unit operations. By removing the isolation, filtration, and drying steps associated with the intermediate, the process significantly reduces the demand for equipment time and labor hours. This streamlining translates into tangible cost reductions in manufacturing overheads without the need for complex capital investment. Furthermore, the qualitative reduction in solvent and reagent consumption directly lowers the variable cost per kilogram of the produced intermediate. Since the process avoids the use of mineral acids for neutralization and the subsequent need for base to re-adjust pH, the expenditure on bulk chemicals is substantially decreased. These efficiencies accumulate to offer a more competitive pricing structure for the final API intermediate, allowing downstream partners to manage their own cost of goods sold more effectively.
- Cost Reduction in Manufacturing: The elimination of intermediate isolation removes the need for extensive filtration and drying infrastructure, leading to significant savings in energy consumption and equipment maintenance. By avoiding the repeated dissolution and precipitation cycles, the process minimizes solvent losses and reduces the volume of waste streams requiring treatment. This leaner operational model ensures that resources are focused purely on value-added chemical transformation rather than mechanical separation, driving down the overall production cost significantly.
- Enhanced Supply Chain Reliability: Simplifying the synthesis route inherently reduces the number of potential failure points in the manufacturing process. Fewer steps mean fewer opportunities for deviations, equipment breakdowns, or quality hold-ups that can disrupt supply continuity. The robustness of the one-pot method allows for more predictable production scheduling and shorter cycle times, enabling suppliers to respond more agilely to fluctuations in market demand. This reliability is crucial for maintaining the steady flow of materials needed for continuous antiviral drug production.
- Scalability and Environmental Compliance: The reduced consumption of acids, bases, and organic solvents aligns perfectly with increasingly stringent environmental regulations. Lower waste generation simplifies effluent treatment and reduces the environmental compliance burden on manufacturing sites. Moreover, the straightforward nature of the reaction makes it highly amenable to scale-up from pilot plants to commercial tonnage production. The ability to run this process efficiently on a large scale ensures that supply can be expanded to meet global healthcare needs without proportionally increasing the environmental footprint.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is vital for stakeholders evaluating its adoption. The following questions address common inquiries regarding the practical implementation and benefits of this one-pot technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent literature, offering a clear picture of what partners can expect when integrating this intermediate into their supply chains. Engaging with these technical details helps clarify the operational advantages and ensures that all parties are aligned on the quality and efficiency standards of the new manufacturing route.
Q: How does the one-pot method improve yield compared to traditional two-step synthesis?
A: By eliminating the isolation and purification of the unstable 3-hydroxy-2-hydroxymethyl-pyran-4-one intermediate, material loss during filtration and recrystallization is avoided, significantly improving overall process efficiency.
Q: What are the primary cost drivers reduced in this new synthetic route?
A: The process drastically reduces the consumption of solvents, acids for neutralization, and alkalis for re-basification, while also lowering energy usage associated with multiple heating and cooling cycles.
Q: Is this intermediate scalable for commercial antiviral drug production?
A: Yes, the simplified operational steps and reduced sensitivity to intermediate handling make this route highly suitable for large-scale commercial manufacturing of key antiviral precursors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Hydroxymethyl-3-Alkoxy-4H-Pyran-4-One Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development and production of next-generation therapeutics. As a dedicated CDMO partner, we possess the technical expertise to translate complex laboratory innovations like the one-pot pyran-4-one synthesis into robust commercial realities. Our facilities are equipped to handle diverse synthetic pathways, with extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We understand that consistency is key, which is why our operations are governed by stringent purity specifications and supported by rigorous QC labs that ensure every batch meets the exacting standards required by the global pharmaceutical industry. Our commitment to technical excellence ensures that the advantages of this novel synthesis are fully preserved during scale-up.
We invite procurement leaders and R&D teams to collaborate with us to leverage these advancements for your specific projects. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your volume requirements and quality needs. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments for this and other critical intermediates. Together, we can optimize your supply chain, reduce costs, and accelerate the delivery of vital medications to patients worldwide.
