Scalable Synthesis Of 4-(1-Methyl-1H-Pyrazol-5-Yl)Piperidine For Commercial API Production
Introduction To The Novel Synthetic Breakthrough
The pharmaceutical industry constantly seeks robust and scalable pathways for critical heterocyclic intermediates, particularly those serving as core scaffolds for neuroactive drugs. Patent CN115073422A, published on September 20, 2022, introduces a highly efficient method for preparing 4-(1-methyl-1H-pyrazol-5-yl)piperidine and its salts, a vital intermediate for muscarinic M4 receptor agonists used in treating schizophrenia and Alzheimer's disease. This innovation addresses the historical lack of reported industrial methods for this specific scaffold, offering a streamlined four-step sequence that begins with commercially abundant 1-methylpyrazole and N-Boc-4-piperidone. By leveraging a strategic combination of organolithium chemistry, acid-mediated dehydration, and catalytic hydrogenation, the disclosed process achieves remarkable total yields while maintaining stringent purity profiles essential for downstream API synthesis. For R&D directors and procurement specialists, this patent represents a pivotal shift towards more accessible and cost-effective sourcing of this high-value building block.
The Limitations Of Conventional Methods Vs. The Novel Approach
The Limitations Of Conventional Methods
Historically, the synthesis of complex pyrazol-piperidine hybrids has been plagued by inefficient multi-step sequences that rely on scarce starting materials or harsh reaction conditions which compromise safety and yield. Traditional approaches often necessitate the use of expensive transition metal cross-coupling reactions that require rigorous purification to remove trace metal residues, a significant bottleneck for pharmaceutical grade intermediates. Furthermore, existing literature prior to this patent indicated a distinct absence of a direct, high-yielding route to 4-(1-methyl-1H-pyrazol-5-yl)piperidine, forcing manufacturers to rely on convoluted custom syntheses that drive up lead times and costs. The lack of a standardized protocol meant that impurity profiles were difficult to control, leading to batch-to-batch variability that is unacceptable for GMP manufacturing environments. These conventional limitations create substantial supply chain risks, making the development of a concise, reproducible methodology not just a technical improvement but a commercial necessity for reliable pharmaceutical intermediate supplier networks.
The Novel Approach
The methodology disclosed in CN115073422A revolutionizes the production landscape by condensing the synthesis into four high-efficiency steps that utilize readily available raw materials and standard laboratory equipment. As illustrated in the comprehensive reaction scheme below, the process initiates with a lithiation-addition sequence to form a tertiary alcohol, followed by a seamless dehydration to generate a tetrahydropyridine intermediate, which is then hydrogenated and deprotected to yield the final salt. 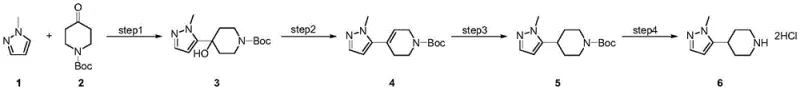 This novel approach eliminates the need for exotic catalysts or complex protecting group manipulations beyond the initial Boc strategy, drastically simplifying the operational workflow. The use of N-Boc-4-piperidone serves a dual purpose, acting as both an electrophile for carbon-carbon bond formation and a protected amine source, which streamlines the atom economy. By achieving yields of 90% in the initial coupling and 100% in the hydrogenation step, this route offers a level of efficiency that significantly lowers the cost of goods sold, making it an ideal candidate for cost reduction in API manufacturing on a commercial scale.
This novel approach eliminates the need for exotic catalysts or complex protecting group manipulations beyond the initial Boc strategy, drastically simplifying the operational workflow. The use of N-Boc-4-piperidone serves a dual purpose, acting as both an electrophile for carbon-carbon bond formation and a protected amine source, which streamlines the atom economy. By achieving yields of 90% in the initial coupling and 100% in the hydrogenation step, this route offers a level of efficiency that significantly lowers the cost of goods sold, making it an ideal candidate for cost reduction in API manufacturing on a commercial scale.
Mechanistic Insights Into Lithiation-Dehydration-Hydrogenation Cascade
The core of this synthetic success lies in the precise control of organometallic reactivity during the first step, where 1-methylpyrazole is treated with n-butyllithium at cryogenic temperatures ranging from -90°C to -50°C. This low-temperature environment is critical for generating the 5-lithio-1-methylpyrazole species selectively without inducing decomposition or side reactions on the sensitive heterocyclic ring. Upon addition of N-Boc-4-piperidone, the nucleophilic attack occurs rapidly to form the tertiary alcohol intermediate, 1-tert-butoxycarbonyl-4-hydroxy-4-(1-methyl-1H-pyrazol-5-yl)piperidine, with exceptional regioselectivity. The subsequent dehydration step utilizes concentrated hydrochloric acid at room temperature to facilitate an E1 elimination mechanism, converting the sterically hindered alcohol into the corresponding 1,2,3,6-tetrahydropyridine derivative. 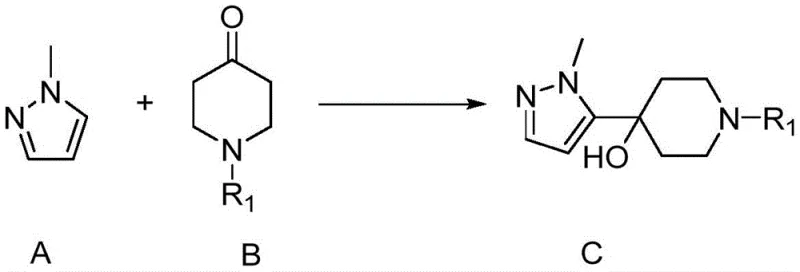 This acid-mediated transformation is particularly elegant as it proceeds under mild thermal conditions, avoiding the high energy inputs typically associated with dehydration reactions. Finally, the saturation of the double bond via Pd/C catalyzed hydrogenation at 50°C and 2MPa pressure ensures complete conversion to the piperidine ring system without affecting the pyrazole moiety, demonstrating the chemoselectivity required for complex molecule synthesis.
This acid-mediated transformation is particularly elegant as it proceeds under mild thermal conditions, avoiding the high energy inputs typically associated with dehydration reactions. Finally, the saturation of the double bond via Pd/C catalyzed hydrogenation at 50°C and 2MPa pressure ensures complete conversion to the piperidine ring system without affecting the pyrazole moiety, demonstrating the chemoselectivity required for complex molecule synthesis.
Impurity control is inherently built into this mechanism through the strategic use of the tert-butoxycarbonyl (Boc) protecting group, which shields the piperidine nitrogen from unwanted side reactions during the lithiation and dehydration phases. By masking the amine functionality, the process prevents polymerization or oligomerization that could occur if the free amine were present during the acidic dehydration step. Furthermore, the final deprotection step using hydrochloric acid in ethanol not only removes the Boc group but simultaneously converts the free base into the stable hydrochloride salt, which precipitates out of the solution, facilitating easy isolation and purification. This crystallization-driven purification strategy ensures that the final product meets the rigorous 98% HPLC purity specification mentioned in the patent examples. For quality assurance teams, this mechanistic design minimizes the formation of hard-to-remove byproducts, thereby reducing the burden on downstream purification processes and ensuring a consistent, high-quality output suitable for sensitive neurological drug applications.
How To Synthesize 4-(1-Methyl-1H-Pyrazol-5-Yl)Piperidine Efficiently
Implementing this synthesis requires strict adherence to the temperature profiles and stoichiometric ratios defined in the patent to maximize yield and safety. The process begins with the careful addition of n-butyllithium to a solution of 1-methylpyrazole in tetrahydrofuran at -70°C, followed by the introduction of the ketone substrate to form the alcohol intermediate in 90% yield. Detailed standard operating procedures for each reaction stage, including workup and purification parameters, are essential for replicating the high success rates observed in the patent examples. The following guide outlines the critical operational milestones necessary to achieve the reported efficiency and purity levels in a production setting.
- Perform lithiation of 1-methylpyrazole with n-butyllithium at -70°C followed by addition of N-Boc-4-piperidone to form the tertiary alcohol intermediate.
- Conduct acid-catalyzed dehydration using concentrated hydrochloric acid at room temperature to generate the tetrahydropyridine derivative.
- Execute catalytic hydrogenation using Pd/C under 2MPa hydrogen pressure at 50°C to saturate the double bond.
- Finalize the process by removing the Boc protecting group with hydrochloric acid in ethanol to obtain the target hydrochloride salt.
Commercial Advantages For Procurement And Supply Chain Teams
From a procurement perspective, this synthetic route offers transformative advantages by utilizing commodity chemicals that are widely available in the global market, thereby mitigating supply chain risks associated with specialized reagents. The reliance on 1-methylpyrazole and N-Boc-4-piperidone as starting materials ensures that raw material sourcing is stable and cost-effective, as these are produced at scale by multiple vendors worldwide. This accessibility translates directly into enhanced supply chain reliability, allowing manufacturers to secure long-term contracts without fear of single-source bottlenecks or geopolitical supply disruptions. Moreover, the elimination of precious metal catalysts like palladium in the early stages, reserving Pd/C only for the final hydrogenation where it can be easily recovered, significantly reduces the material cost profile. The process avoids the need for expensive chromatography columns in favor of crystallization and extraction techniques, which are far more scalable and economically viable for tonnage production.
- Cost Reduction In Manufacturing: The streamlined four-step sequence drastically reduces the number of unit operations required, leading to substantial savings in labor, energy, and solvent consumption compared to longer, more convoluted synthetic alternatives. By achieving high yields in each step, particularly the quantitative hydrogenation, the overall material throughput is maximized, meaning less raw material is wasted to side products. The use of common solvents like tetrahydrofuran and ethanol further lowers operational expenditures, as these can be readily recycled and reused within the plant infrastructure. Additionally, the avoidance of complex purification steps such as preparative HPLC in favor of simple filtration and drying reduces the capital investment required for processing equipment. These factors combine to create a highly competitive cost structure that allows for significant margin improvement in the final API pricing.
- Enhanced Supply Chain Reliability: The robustness of this chemistry ensures consistent batch-to-batch performance, which is critical for maintaining uninterrupted supply to downstream API manufacturers. Because the reaction conditions are well-defined and utilize standard reagents, the risk of batch failure due to reagent variability is minimized, fostering trust between the supplier and the client. The ability to produce the hydrochloride salt directly simplifies logistics, as the salt form is typically more stable and easier to handle than the free base during storage and transportation. This stability reduces the need for specialized cold-chain logistics, further lowering the total cost of ownership for the buyer. Consequently, partners can rely on a steady flow of high-quality intermediate to support their own clinical and commercial timelines without unexpected delays.
- Scalability And Environmental Compliance: The process is designed with green chemistry principles in mind, utilizing catalytic hydrogenation which produces water as the only byproduct in the reduction step, minimizing hazardous waste generation. The reaction temperatures, while requiring cooling for the first step, are otherwise moderate, reducing the energy load on the facility's HVAC and utility systems. The high atom economy of the coupling and dehydration steps ensures that the majority of the input mass ends up in the final product, aligning with modern sustainability goals and regulatory expectations for waste reduction. This environmental friendliness simplifies the permitting process for new manufacturing lines and enhances the corporate social responsibility profile of the supply chain. Scalability is further supported by the use of heterogeneous catalysts which are easily filtered, making the transition from kilogram to multi-ton production seamless and safe.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These answers are derived directly from the experimental data and beneficial effects described in CN115073422A, providing clarity on yield expectations, safety protocols, and purity standards. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this route for their specific supply chain needs.
Q: What is the overall yield and purity of the new synthesis method?
A: According to patent CN115073422A, the optimized process achieves a high total yield with the final step producing the hydrochloride salt at 85% yield and 98% HPLC purity, demonstrating excellent efficiency for industrial application.
Q: Are the reaction conditions safe for large-scale manufacturing?
A: Yes, the process utilizes standard reagents like n-butyllithium and Pd/C under controlled conditions. While low temperatures (-70°C) are required for lithiation, the subsequent steps operate at room temperature or moderate heating (50°C), ensuring operational safety and scalability.
Q: How does this method improve upon existing synthetic routes?
A: This method significantly shortens the synthetic sequence to just four steps from readily available starting materials, eliminating the need for complex multi-step protections or expensive transition metal catalysts often found in alternative pathways, thereby reducing production costs.
Partnering With NINGBO INNO PHARMCHEM: Your Reliable 4-(1-Methyl-1H-Pyrazol-5-Yl)Piperidine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of next-generation neurological therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is managed with precision and expertise. We are committed to delivering 4-(1-methyl-1H-pyrazol-5-yl)piperidine with stringent purity specifications, utilizing our rigorous QC labs to verify every batch against the highest industry standards. Our state-of-the-art facilities are equipped to handle the specific temperature and pressure requirements of this synthesis, guaranteeing a consistent supply that meets the demanding timelines of global pharmaceutical partners.
We invite you to engage with our technical procurement team to discuss how this optimized synthetic route can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of switching to this efficient manufacturing method. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments tailored to your volume needs. Let us collaborate to secure a stable, cost-effective, and high-quality supply chain for your vital API intermediates, driving your projects forward with confidence and reliability.
