Advanced High-Temperature Synthesis of Alkylidene 1,4-Diketones for Industrial Catalyst Applications
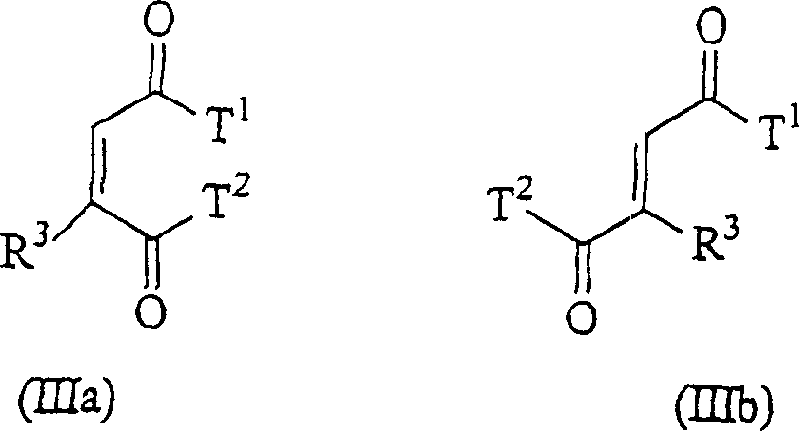
The chemical industry continuously seeks more efficient pathways for synthesizing critical intermediates, particularly those serving as electron donors in Ziegler-Natta heterogeneous catalysts for olefin polymerization. Patent CN1585737A introduces a transformative process for the preparation of alkylidene substituted 1,4-diketone derivatives, specifically addressing the limitations of traditional Stobbe condensations and room-temperature base promotions. This innovation centers on reacting nitroalkanes with 2-ene-1,4-diones in the presence of inorganic base salts at elevated temperatures exceeding 70°C. Unlike prior art that relied on expensive organic bases or complex multi-step workups, this method leverages thermal energy to drive high-yield conversions with remarkable simplicity. The resulting compounds, characterized by the general structures shown below, are pivotal precursors for generating alkyl-substituted derivatives used extensively in polyolefin manufacturing.

For procurement specialists and supply chain managers, the implications of this technology are profound. By shifting from stoichiometric excesses of costly reagents to catalytic or near-stoichiometric amounts of inexpensive inorganic salts, the process offers a reliable polymer additive supplier pathway that significantly lowers the cost of goods sold. The ability to produce these high-purity electron donor compounds with minimal downstream processing translates directly into enhanced operational efficiency and reduced environmental footprint, making it an attractive option for large-scale industrial adoption.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical methods for synthesizing monoalkylidene-substituted succinates, such as those described by Overberg and Roberts, utilized the Stobbe reaction with potassium tert-butoxide in tert-butanol. While effective on a small scale, these processes are economically burdensome for industrial application due to the requirement for large excesses of starting materials and base. Specifically, the starting diethyl succinate was often used in 25% excess, and the base in 10% excess relative to the ketone. More critically, the isolation of the final product demanded a laborious and hazardous workup sequence involving solvent distillation, acidification to pH 3, multiple ether extractions, and anhydrification. Similarly, methods employing DBU at room temperature, while faster, often resulted in lower yields relative to the succinate and left behind difficult-to-remove impurities, complicating the supply chain for high-purity intermediates.
The Novel Approach
The patented process fundamentally alters the reaction landscape by introducing thermal activation. By conducting the reaction between nitroalkanes and 2-ene-1,4-diones at temperatures ranging from 70°C to 300°C, preferably between 150°C and 200°C, the reaction kinetics are vastly improved. This thermal drive allows for the use of cheap, readily available inorganic bases like potassium carbonate or sodium bicarbonate instead of expensive organic amines. The reaction scheme below illustrates the core transformation where a 2-ene-1,4-dione reacts with a nitroalkane under these heated conditions to form the target alkylidene derivative.
This approach eliminates the need for the complex acid-base extractions typical of the Stobbe reaction. In most cases, the workup is reduced to a simple dilution with water followed by organic solvent extraction. This drastic simplification not only reduces solvent consumption and waste generation but also shortens the production cycle time, offering substantial cost reduction in polyolefin catalyst manufacturing. The robustness of this method allows for the use of various solvents, including high-boiling aprotic solvents like DMF or even solvent-free conditions if reactants are liquid, providing flexibility for process engineers optimizing for green chemistry metrics.
Mechanistic Insights into High-Temperature Base-Catalyzed Condensation
The mechanistic advantage of this process lies in the synergistic effect of temperature and base strength. At room temperature, the deprotonation of the nitroalkane by weaker inorganic bases is often insufficient to drive the condensation to completion efficiently, leading to equilibrium mixtures or stalled reactions. However, at elevated temperatures (>70°C), the kinetic barrier for the initial Michael addition of the nitronate anion to the activated double bond of the 2-ene-1,4-dione is overcome rapidly. Furthermore, the subsequent elimination of nitrous acid (or its equivalent) to form the exocyclic double bond is thermodynamically favored at higher temperatures. This ensures that the reaction proceeds to near-quantitative conversion without the need for the strong, non-nucleophilic organic bases like DBU that are typically required at ambient conditions.
From an impurity control perspective, the high-temperature regime is equally beneficial. Lower temperature reactions involving nitro compounds and amines (like diethylamine in older methods) are notorious for generating carcinogenic N-nitrosamines. By utilizing inorganic carbonate salts and avoiding amine bases, this process inherently mitigates the risk of forming such toxic by-products. Additionally, the thermal conditions promote the isomerization of any maleate starting material to the more stable fumarate or directly to the product, ensuring that the final crude mixture is clean. This purity profile is critical for R&D directors focusing on the downstream hydrogenation step, where catalyst poisoning by nitrogenous impurities must be strictly avoided to maintain the activity of palladium or platinum hydrogenation catalysts.
How to Synthesize Alkylidene Substituted 1,4-Diketone Efficiently
To implement this synthesis effectively, one must carefully control the stoichiometry and thermal profile. The process involves mixing the 2-ene-1,4-dione (such as diethyl maleate) with the nitroalkane in a suitable high-boiling solvent. A base salt, typically potassium carbonate, is added in an amount of at least one equivalent relative to the nitroalkane. The mixture is then heated rapidly to reflux or a specific target temperature between 150°C and 200°C. Monitoring via GC is recommended to determine the endpoint, which is typically reached within minutes to a few hours depending on the specific substrates. Upon completion, the reaction mass is cooled, diluted with water, and the product extracted, yielding a high-purity intermediate ready for further functionalization or hydrogenation.
- Mix a compound of formula (II) (e.g., diethyl maleate) with a compound of formula (IIIa) or (IIIb) (e.g., nitroalkane) in a suitable solvent.
- Add at least one equivalent of a base salt (such as potassium carbonate) relative to the nitroalkane compound.
- Heat the reaction mixture to a temperature higher than 70°C, preferably between 150°C and 200°C, until completion, followed by simple aqueous workup.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this high-temperature inorganic base process represents a strategic opportunity to optimize the supply of critical polymer additives. The shift away from proprietary or expensive organic bases to commodity chemicals like potassium carbonate creates a more resilient and cost-effective supply chain. This is particularly relevant for manufacturers of Ziegler-Natta catalysts where the internal electron donor is a key performance determinant. The simplified workflow reduces the dependency on specialized waste treatment facilities required for handling complex acidic and basic effluents generated by older methods.
- Cost Reduction in Manufacturing: The replacement of expensive organic bases like DBU or potassium tert-butoxide with inexpensive inorganic salts such as potassium carbonate results in direct raw material cost savings. Furthermore, the elimination of complex workup steps—specifically the removal of solvent distillation and multiple acid/base extraction cycles—significantly reduces utility costs associated with heating, cooling, and solvent recovery. The high yields reported (up to 98%) minimize raw material waste, ensuring that every kilogram of input generates maximum output value.
- Enhanced Supply Chain Reliability: The reagents required for this process, including diethyl maleate, nitroalkanes, and alkali metal carbonates, are globally available commodity chemicals with stable pricing and supply. This contrasts with specialized organic bases that may have longer lead times or supply volatility. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in reagent quality, reducing the risk of batch failures and ensuring consistent delivery schedules for downstream catalyst producers.
- Scalability and Environmental Compliance: The process is inherently scalable due to its simplicity and the use of thermally stable conditions. The absence of toxic N-nitrosamine by-products simplifies environmental compliance and waste disposal, lowering the regulatory burden on manufacturing sites. The ability to run the reaction in high-boiling solvents or even solvent-free conditions allows for higher throughput in existing reactor vessels, facilitating the commercial scale-up of complex polymer additives without significant capital expenditure on new equipment.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation of this patented synthesis route. These answers are derived directly from the experimental data and claims within the patent documentation, providing clarity on reaction conditions, safety, and product quality. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this method for their specific production lines.
Q: Why is high temperature critical for this synthesis compared to conventional DBU methods?
A: Conventional methods using DBU at room temperature often suffer from lower yields and complex impurity profiles. The patented high-temperature process (>70°C) significantly accelerates the reaction kinetics and drives the equilibrium towards the desired alkylidene product, achieving yields up to 98% while eliminating the need for expensive organic bases.
Q: What are the advantages of using inorganic base salts like K2CO3 over organic bases?
A: Using inorganic salts like potassium carbonate drastically reduces raw material costs compared to organic bases like DBU. Furthermore, inorganic bases facilitate a much simpler workup procedure involving basic aqueous dilution and extraction, avoiding the complex acidification and distillation steps required by older Stobbe condensation methods.
Q: How does this process impact the purity of the final electron donor compound?
A: The process minimizes the formation of harmful by-products such as N-nitrosodiethylamine, which is a common issue in nitroalkane reactions at lower temperatures. The high-temperature conditions ensure cleaner conversion, resulting in high-purity intermediates suitable for sensitive Ziegler-Natta catalyst preparation without extensive purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alkylidene 1,4-Diketone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity electron donor compounds play in the performance of modern polyolefin catalysts. Our technical team has extensively analyzed the process described in CN1585737A and possesses the expertise to translate this laboratory-scale innovation into robust commercial manufacturing. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our rigorous QC labs and stringent purity specifications guarantee that every batch of alkylidene 1,4-diketone meets the exacting standards required for Ziegler-Natta catalyst preparation.
We invite you to collaborate with us to leverage this advanced synthesis technology for your specific applications. Whether you require custom modifications to the alkyl groups or specific ester functionalities, our R&D team is ready to assist. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your current volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our optimized manufacturing capabilities can enhance your supply chain efficiency and product quality.
