Scalable Synthesis of Bis-(p-carboxyanilino) Phenylphosphine Oxide for Industrial Flame Retardants
Scalable Synthesis of Bis-(p-carboxyanilino) Phenylphosphine Oxide for Industrial Flame Retardants
The development of high-performance, halogen-free flame retardants is critical for the advancement of sustainable polymer materials, particularly in polyester and polyamide applications. Patent CN112538091B introduces a groundbreaking synthesis method for bis-(p-carboxyanilino) phenylphosphine oxide, a reactive organophosphorus compound that offers superior thermal stability and fire resistance. This technical insight report analyzes the proprietary two-step pathway detailed in the patent, which achieves a remarkable intermediate yield of 84% and final product purity exceeding 98.4%. By shifting from traditional acid-based routes to a novel ester aminolysis strategy, this process resolves long-standing issues regarding side reactions and low conversion rates. For R&D directors and procurement specialists seeking a reliable flame retardant intermediate supplier, understanding the mechanistic advantages of this route is essential for securing a stable supply chain of high-purity polymer additives.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of bis-(p-carboxyanilino) phenylphosphine oxide has been plagued by significant chemical inefficiencies that hinder industrial scalability. Prior art, such as US2882294, relied on the reaction of phenylphosphoryl dichloride with ethyl p-aminobenzoate using triethylamine as an acid scavenger. However, this approach suffered from a critically low yield of only 42% due to the formation of unstable phosphoramide isomers under basic conditions. Furthermore, alternative methods utilizing p-aminobenzoic acid directly often resulted in complex impurity profiles. In these acid-based routes, phenylphosphoryl dichloride reacts indiscriminately with the carboxylic acid group to form acid chlorides, triggering uncontrolled self-condensation and acetylation side reactions. These impurities not only reduce the overall mass balance but also compromise the thermal performance of the final flame retardant, making purification costly and technically challenging for large-scale manufacturing.
The Novel Approach
The methodology disclosed in CN112538091B represents a paradigm shift in organophosphorus chemistry by eliminating external bases and problematic acid functionalities during the coupling phase. Instead of triethylamine, the process utilizes a four-fold molar excess of ethyl p-aminobenzoate, which serves simultaneously as the nucleophile and the acid binding agent. This strategic modification suppresses the isomerization of the P-N bond, thereby stabilizing the intermediate and driving the reaction yield to approximately 84%. Additionally, the subsequent hydrolysis step employs a ternary solvent system of ethanol, water, and 1,4-dioxane. This specific solvent mixture ensures complete solubility of the diester intermediate, creating a homogeneous reaction environment that prevents localized high alkalinity which could otherwise degrade the phosphoramide linkage. 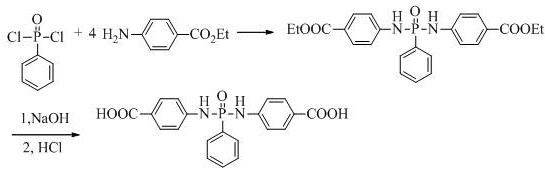
Mechanistic Insights into Ester Aminolysis and Controlled Hydrolysis
The core innovation of this synthesis lies in the precise control of nucleophilic substitution at the phosphorus center. In the first step, phenylphosphoryl dichloride undergoes aminolysis with the amine group of ethyl p-aminobenzoate. By maintaining a molar ratio between 4.00:1 and 4.20:1 at a controlled temperature of 50-60°C, the reaction kinetics favor the formation of the P-N bond while the excess amine neutralizes the generated HCl in situ. This avoids the introduction of tertiary amine salts that are difficult to remove and often catalyze degradation. The absence of strong external bases prevents the rearrangement of the ArNHP=O structure into the less stable ArN=P-OH tautomer, which is the primary cause of yield loss in conventional methods. Consequently, the diester intermediate precipitates with high purity, requiring only simple acid washing to remove the amine hydrochloride salt before proceeding to hydrolysis.
The second stage involves the alkaline saponification of the ester groups to generate the free carboxylic acids required for polymer reactivity. A critical mechanistic challenge in this step is preserving the integrity of the phosphoramide bond, which is susceptible to hydrolysis under harsh alkaline conditions. The patent specifies the use of 1,4-dioxane as a co-solvent alongside ethanol and water. This addition is vital because it maintains a single-phase system throughout the addition of sodium hydroxide. In binary ethanol-water systems, the intermediate often remains partially insoluble, leading to heterogeneous reaction zones where local pH spikes can exceed 13, causing cleavage of the P-N bond. By ensuring homogeneity, the pH can be strictly maintained between 9 and 13, allowing for selective ester hydrolysis while leaving the phosphoramide backbone intact. This results in a hydrolysis yield of approximately 80%, significantly higher than the 45-57% observed in solvent systems lacking the co-solvent.
How to Synthesize Bis-(p-carboxyanilino) Phenylphosphine Oxide Efficiently
Implementing this synthesis route requires careful attention to stoichiometry and solvent management to maximize the economic and technical benefits described in the patent. The process is designed for batch production, starting with the dissolution of the amine ester in 1,4-dioxane followed by the controlled addition of the phosphoryl chloride. Post-reaction workup involves a unique recycling protocol where the filtrate containing excess starting material can be recovered and reused, further enhancing process efficiency. The standardized synthetic steps below outline the critical parameters for temperature, pH control, and isolation that ensure the production of high-purity material suitable for reactive flame retardant applications. Detailed operational procedures follow the established safety and quality protocols for handling phosphorus halides and organic solvents.
- React phenylphosphoryl dichloride with a 4-fold molar excess of ethyl p-aminobenzoate in 1,4-dioxane at 50-60°C to form the diester intermediate.
- Isolate the intermediate via filtration and wash with dilute hydrochloric acid to remove excess amine salts.
- Perform alkaline saponification using NaOH in an ethanol-1,4-dioxane-water mixture, followed by acidification to precipitate the pure acid product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthesis route offers substantial strategic advantages beyond mere technical performance. The elimination of triethylamine removes the need for expensive quenching and wastewater treatment processes associated with amine salts, leading to significant cost reduction in polymer additive manufacturing. Furthermore, the ability to recover and recycle the excess ethyl p-aminobenzoate from the mother liquor creates a closed-loop material flow that minimizes raw material waste. This efficiency translates directly into a more competitive pricing structure for the final flame retardant intermediate, allowing downstream polymer producers to manage their bill of materials more effectively without sacrificing quality.
- Cost Reduction in Manufacturing: The process achieves cost optimization primarily through the dual function of the amine ester as both reactant and acid scavenger. By removing the requirement for external bases like triethylamine, the process eliminates the cost of purchasing additional reagents and the downstream costs associated with separating and disposing of amine hydrochloride salts. Additionally, the high yield of 84% for the intermediate and 80% for the final hydrolysis step means that less raw material is required per kilogram of output. The patent explicitly describes a recovery method for the filtrate, allowing the excess starting material to be recycled one to two times, which drastically reduces the net consumption of ethyl p-aminobenzoate and lowers the overall variable cost of production.
- Enhanced Supply Chain Reliability: Sourcing high-purity organophosphorus intermediates can often be a bottleneck due to the complexity of purification. This method simplifies the supply chain by producing a crude intermediate that is easily purified through crystallization and washing, rather than requiring complex chromatography or distillation. The raw materials, specifically ethyl p-aminobenzoate and phenylphosphoryl dichloride, are commodity chemicals with robust global availability, reducing the risk of supply disruptions. The robustness of the reaction conditions, which tolerate slight variations in temperature and time without significant yield loss, ensures consistent batch-to-batch quality, providing reliability for long-term contracts and just-in-time delivery schedules.
- Scalability and Environmental Compliance: From an environmental and scaling perspective, the use of 1,4-dioxane and ethanol allows for efficient solvent recovery via vacuum distillation, minimizing volatile organic compound (VOC) emissions. The aqueous waste streams generated during the washing steps contain recoverable amine salts that can be neutralized and extracted, reducing the environmental load compared to processes generating complex organic sludge. The homogeneous nature of the hydrolysis step facilitates heat transfer and mixing in large reactors, making the scale-up from laboratory to commercial tonnage straightforward. This scalability ensures that suppliers can meet increasing demand for halogen-free flame retardants driven by stricter global fire safety regulations without requiring prohibitive capital investment in new reactor types.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of bis-(p-carboxyanilino) phenylphosphine oxide. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation. Understanding these details helps stakeholders evaluate the feasibility of integrating this intermediate into their specific polymer formulations and manufacturing workflows.
Q: Why is ethyl p-aminobenzoate preferred over p-aminobenzoic acid in this synthesis?
A: Using the ester prevents the formation of carboxylic acid chlorides and subsequent self-condensation by-products, significantly improving purity compared to direct acid methods.
Q: How does the new method improve yield compared to US2882294?
A: By using the amine ester itself as the acid scavenger instead of triethylamine, the process avoids phosphoramide isomerization side reactions, boosting yield from 42% to over 84%.
Q: What is the role of 1,4-dioxane in the hydrolysis step?
A: 1,4-dioxane acts as a co-solvent to ensure homogeneity during saponification, preventing localized high pH that could cleave the sensitive phosphoramide bond.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bis-(p-carboxyanilino) Phenylphosphine Oxide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-performance flame retardants play in the next generation of engineering plastics. Our technical team has thoroughly analyzed the pathway described in CN112538091B and is fully equipped to translate this laboratory-scale innovation into commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the stringent purity specifications required for reactive monomers are met consistently. Our rigorous QC labs utilize advanced HPLC and NMR spectroscopy to verify that every batch meets the >98.4% purity standard, guaranteeing optimal performance in your polyester or polyamide matrices.
We invite you to collaborate with us to optimize your supply chain for halogen-free flame retardants. By leveraging our process engineering expertise, we can offer a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you secure a sustainable, high-quality supply of bis-(p-carboxyanilino) phenylphosphine oxide that drives both performance and profitability in your manufacturing operations.
