Revolutionizing Agrochemical Intermediates: Atmospheric One-Pot Synthesis of Chloromethylphosphonic Acid Monosodium Salt
Revolutionizing Agrochemical Intermediates: Atmospheric One-Pot Synthesis of Chloromethylphosphonic Acid Monosodium Salt
The landscape of fine chemical manufacturing is constantly evolving, driven by the need for safer, more efficient, and cost-effective synthetic routes. A pivotal advancement in this domain is detailed in Chinese Patent CN100360546C, which discloses a novel one-pot preparation process for chloromethylphosphonic acid monosodium salt. This compound serves as a critical precursor in the synthesis of N-substituted aminomethylphosphonic acids, including the widely used herbicide glyphosate. Unlike traditional methods that rely on dangerous high-pressure conditions, this innovation utilizes an atmospheric pressure catalytic system that significantly simplifies the production workflow. By integrating the synthesis, hydrolysis, and purification stages into a streamlined sequence, the technology addresses long-standing challenges regarding catalyst separation and product crystallization. For a reliable agrochemical intermediate supplier, adopting such methodologies represents a strategic leap towards enhancing operational safety while maintaining rigorous quality standards.
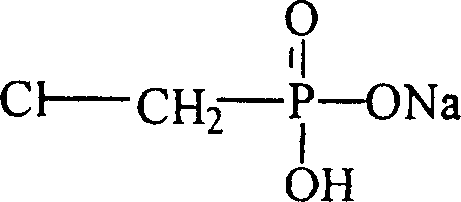
The structural integrity of the final product is paramount for downstream applications, particularly in the formulation of water-based lubricants and cellulose ion exchangers where chelating properties are exploited. The patent elucidates a pathway that not only secures the P-C bond structure essential for biological activity but also ensures the product exists in its stable monosodium salt form, which is the active species under alkaline application conditions. This direct synthesis approach bypasses the need for converting free acid to salt in a separate step, thereby reducing unit operations and potential yield losses. As the industry seeks cost reduction in pharmaceutical intermediates manufacturing, such integrated processes offer a compelling value proposition by minimizing solvent usage and energy consumption associated with multi-step isolations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chloromethylphosphonic acid has been plagued by severe engineering and safety constraints, primarily due to the reliance on high-pressure reactors. Early methodologies, such as those described in U.S. Patent 2874184, necessitated reaction pressures reaching as high as 9.3 MPa, requiring robust and expensive pressure vessels that increase capital expenditure significantly. Furthermore, these processes often utilized anhydrous hydrogen chloride as a catalyst, which is typically supplied in high-pressure cylinders, introducing a substantial risk of gas leakage and operator exposure to corrosive hazards. Even improved pressure methods operating at 1.7 to 3.5 MPa, while slightly safer, still demand specialized equipment and rigorous safety protocols that can bottleneck production capacity. Additionally, conventional atmospheric catalytic methods reported in literature, such as European Patent 1147179, frequently suffered from inadequate catalyst separation, leading to contaminated products that were difficult to crystallize and purify to acceptable commercial standards.
The Novel Approach
In stark contrast, the one-pot method disclosed in CN100360546C operates entirely under normal atmospheric pressure, effectively eliminating the need for high-pressure infrastructure and the associated safety risks. This approach leverages phosphorus trichloride and paraformaldehyde as readily available raw materials, catalyzed by anhydrous aluminum trichloride in a controlled thermal environment. The brilliance of this method lies in its post-reaction workup; by adjusting the pH of the hydrolyzed solution to a specific range of 4.5 to 5 using sodium hydroxide, the aluminum catalyst is converted into insoluble aluminum hydroxide, which precipitates out completely. This ingenious step resolves the persistent issue of catalyst removal that hindered previous atmospheric techniques, allowing for the isolation of high-purity crude products through simple filtration. Consequently, this facilitates the commercial scale-up of complex agrochemical intermediates by enabling the use of standard glass-lined or stainless steel reactors without pressure ratings.
Mechanistic Insights into AlCl3-Catalyzed Phosphonylation
The core of this synthetic breakthrough relies on the efficient activation of the phosphorus-carbon bond formation under mild thermal conditions. The reaction initiates with the interaction between phosphorus trichloride and paraformaldehyde, where the Lewis acid catalyst, anhydrous aluminum trichloride, plays a crucial role in facilitating the electrophilic attack. The process involves a gradual temperature ramping strategy, initially heating the mixture to 80°C to ensure the complete dissolution of paraformaldehyde, followed by a sustained reaction at 100°C. Subsequently, the addition of the catalyst and further heating to a range of 160°C to 180°C drives the formation of the key intermediate, chloromethylphosphonyl dichloride. This precise thermal control is vital to minimize the formation of by-products such as dichloromethane and dichloromethyl ether, which are distilled off during the process, thereby shifting the equilibrium towards the desired phosphonyl dichloride and enhancing the overall atom economy of the transformation.
Following the formation of the dichloride intermediate, the mechanism shifts to a hydrolysis and neutralization phase that is critical for impurity control. Upon the addition of water at 70°C, the P-Cl bonds are hydrolyzed to form the corresponding phosphonic acid species. The subsequent addition of sodium hydroxide serves a dual purpose: it neutralizes the acidic protons to form the monosodium salt and simultaneously triggers the precipitation of the aluminum catalyst as aluminum hydroxide. This selective precipitation is the key to achieving high purity, as it physically removes the metal catalyst from the organic phase without requiring complex solvent extractions that could lead to product loss. The resulting solution, now free of heavy metal contaminants, undergoes evaporation and cooling crystallization, followed by recrystallization in an ethanol-water system, ensuring that the final product meets stringent purity specifications exceeding 98% as confirmed by potentiometric titration and NMR analysis.
How to Synthesize Chloromethylphosphonic Acid Monosodium Salt Efficiently
Implementing this synthesis route requires strict adherence to moisture-free conditions during the initial stages to prevent premature hydrolysis of the reactive phosphorus trichloride. The protocol dictates the use of re-distilled phosphorus trichloride and dry apparatus to maximize yield, which has been demonstrated to reach over 80% in optimized examples. The detailed standardized synthesis steps involve precise temperature ramping and pH control to ensure the complete conversion of raw materials and the effective removal of the catalyst. For R&D teams looking to replicate or scale this process, understanding the nuances of the crystallization step is essential, as the choice of the 4:1 ethanol-water solvent system is critical for obtaining the white powdery crystal form with the correct melting point of 200-202°C.
- React phosphorus trichloride with excess paraformaldehyde under reflux, heating gradually to 100°C to form the initial adduct.
- Add anhydrous aluminum trichloride catalyst and heat to 160-180°C under atmospheric pressure to generate chloromethylphosphonyl dichloride.
- Hydrolyze the intermediate with water, adjust pH to 4.5-5 using sodium hydroxide to precipitate aluminum catalyst, then recrystallize from ethanol-water.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the transition to this atmospheric one-pot process offers profound strategic benefits that extend beyond mere technical feasibility. The elimination of high-pressure reactors significantly lowers the barrier to entry for manufacturing, allowing for a broader base of qualified suppliers and reducing the dependency on specialized, high-cost infrastructure. This democratization of production capability enhances supply chain reliability by mitigating the risk of bottlenecks associated with limited high-pressure processing capacity. Furthermore, the use of common raw materials like paraformaldehyde and phosphorus trichloride, which are commodity chemicals with stable global supply chains, ensures consistent availability and protects against raw material volatility. The simplified workup procedure also translates to reduced cycle times, enabling faster turnaround from batch initiation to finished goods, which is crucial for meeting just-in-time delivery schedules in the fast-paced agrochemical sector.
- Cost Reduction in Manufacturing: The economic implications of this process are substantial, primarily driven by the removal of expensive high-pressure equipment and the associated maintenance and safety compliance costs. By operating at atmospheric pressure, manufacturers can utilize standard reaction vessels, which depreciate slower and require less rigorous inspection regimes compared to pressure-rated systems. Additionally, the efficient catalyst removal via precipitation eliminates the need for costly purification resins or complex extraction solvents, directly lowering the variable cost per kilogram of the produced intermediate. The high yield reported in the patent examples suggests a maximized utilization of raw materials, further contributing to a leaner cost structure that can be passed down as competitive pricing for bulk purchasers seeking cost reduction in agrochemical intermediates manufacturing.
- Enhanced Supply Chain Reliability: Safety is a cornerstone of supply chain continuity, and this process markedly reduces operational risks by avoiding the handling of high-pressure hydrogen chloride gas. The reduced hazard profile minimizes the likelihood of unplanned shutdowns due to safety incidents or regulatory inspections, ensuring a more predictable production schedule. Moreover, the ability to produce the stable monosodium salt directly simplifies storage and logistics, as the salt form is generally less hygroscopic and more stable than the free acid, reducing degradation during transit. This stability allows for larger batch sizes and longer inventory holding periods without quality compromise, providing a buffer against market fluctuations and ensuring reducing lead time for high-purity agrochemical intermediates for downstream formulators.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces unforeseen challenges, but the linear nature of this one-pot synthesis facilitates a smoother transition from pilot plant to full commercial production. The absence of high-pressure constraints means that reactor volume can be increased without exponential increases in engineering complexity or safety risks. Environmentally, the process generates fewer hazardous waste streams; the aluminum catalyst is recovered as a solid hydroxide sludge which is easier to handle and dispose of compared to liquid acidic waste containing dissolved metals. The efficient distillation of by-products like dichloromethane allows for their recovery and reuse, aligning with green chemistry principles and helping manufacturers meet increasingly stringent environmental regulations without sacrificing throughput or efficiency.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of chloromethylphosphonic acid monosodium salt based on the patented technology. These insights are derived directly from the experimental data and process descriptions found in the intellectual property documentation, providing a transparent view of the method's capabilities. Understanding these details is crucial for technical buyers evaluating the feasibility of integrating this intermediate into their own synthesis pipelines for herbicides or chelating agents.
Q: How does the atmospheric method improve safety compared to traditional high-pressure synthesis?
A: Traditional methods often require pressures up to 9.3 MPa and involve hazardous anhydrous hydrogen chloride gas, posing significant leakage risks. The novel atmospheric process operates at normal pressure, eliminating the need for specialized high-pressure vessels and drastically reducing operational hazards associated with high-pressure gas handling.
Q: What is the key innovation regarding catalyst removal in this patent?
A: Previous atmospheric methods struggled with separating Lewis acid catalysts. This process utilizes a specific pH adjustment step (pH 4.5-5) using sodium hydroxide, which converts the soluble aluminum catalyst into insoluble aluminum hydroxide precipitate, allowing for easy filtration and ensuring high product purity without complex extraction steps.
Q: What purity levels can be achieved with this one-pot recrystallization technique?
A: By employing a specific recrystallization protocol using a 4:1 volume ratio of ethanol to water, the process consistently yields white powdery crystals with a purity exceeding 98%, as verified by potentiometric titration and NMR spectroscopy, making it suitable for sensitive agrochemical applications like glyphosate synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chloromethylphosphonic Acid Monosodium Salt Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of your final formulations. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering products that meet stringent purity specifications, leveraging advanced analytical capabilities in our rigorous QC labs to verify every batch against the highest industry standards. Our expertise in phosphorus chemistry allows us to optimize the one-pot synthesis described in CN100360546C, guaranteeing a consistent supply of chloromethylphosphonic acid monosodium salt that supports your production timelines and quality goals.
We invite you to collaborate with us to explore how this advanced manufacturing route can benefit your specific application needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized processes can drive value and efficiency in your supply chain, positioning you for success in the competitive global market.
