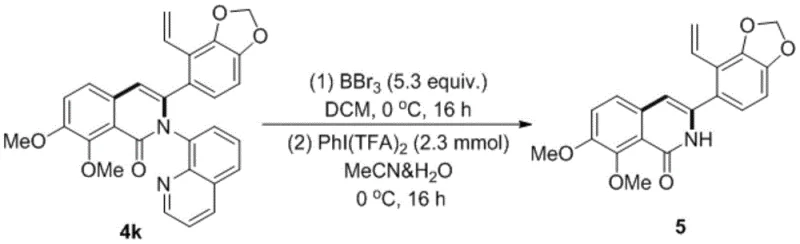
Advanced Cobalt-Catalyzed Synthesis of Polysubstituted Isoquinolinone Derivatives for Commercial Scale-Up
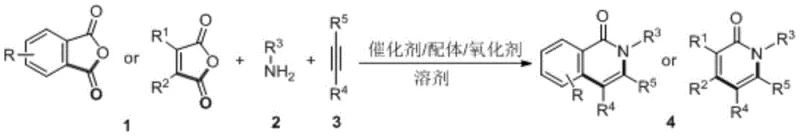
The pharmaceutical and agrochemical industries are constantly seeking more efficient pathways to access complex heterocyclic scaffolds, particularly the isoquinolinone core, which serves as a critical pharmacophore in numerous antifungal, antiviral, and antitumor agents. Patent CN113321639B introduces a groundbreaking one-pot methodology for the preparation of polysubstituted isoquinolinone derivatives, utilizing a cobalt-catalyzed three-component condensation and cyclization reaction. This innovative approach starts from simple, commercially available acid anhydrides, amines, and alkynes, effectively bypassing the need for pre-synthesized intermediates that traditionally plague multi-step syntheses. By leveraging a robust cobalt catalytic system, this technology not only streamlines the synthetic route but also significantly enhances the economic viability of producing these high-value pharmaceutical intermediates. For R&D directors and procurement managers alike, this represents a pivotal shift towards more sustainable and cost-effective API manufacturing processes that do not compromise on yield or purity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the isoquinolinone skeleton has relied heavily on multi-step sequences that often require the pre-functionalization of starting materials, leading to increased waste generation and higher overall production costs. Traditional methods frequently employ precious metal catalysts such as rhodium, palladium, or nickel, which are not only expensive but also sensitive to air and moisture, necessitating stringent inert atmosphere conditions that complicate scale-up operations. Furthermore, the reliance on niche starting materials often creates supply chain bottlenecks, as these precursors may not be available in bulk quantities, thereby increasing lead times and exposing manufacturers to price volatility. The cumulative effect of these factors is a synthetic route that is fragile, costly, and difficult to translate from the laboratory bench to commercial scale-up of complex pharmaceutical intermediates, ultimately hindering the rapid development of new drug candidates.
The Novel Approach
In stark contrast, the novel cobalt-catalyzed protocol described in the patent offers a streamlined, one-pot solution that directly assembles the target molecule from three readily accessible building blocks. This method eliminates the need for intermediate isolation, drastically reducing solvent consumption and processing time while improving the overall atom economy of the transformation. The use of earth-abundant cobalt catalysts, specifically cobalt iodide, replaces expensive noble metals, offering a substantial reduction in raw material costs without sacrificing catalytic efficiency. Moreover, the reaction conditions are remarkably robust, tolerating temperatures between 80°C and 160°C and functioning effectively in common organic solvents like 1,2-dichlorobenzene. This resilience allows for greater flexibility in process engineering, making it an ideal candidate for cost reduction in pharmaceutical intermediate manufacturing while ensuring consistent quality and high yields across a diverse range of substrates.
Mechanistic Insights into Cobalt-Catalyzed Cyclization
The core of this transformative chemistry lies in the unique ability of the cobalt catalyst to facilitate C-H activation and subsequent annulation under oxidative conditions. The reaction initiates with the coordination of the cobalt species to the nitrogen atom of the amine and the carbonyl oxygen of the anhydride, forming a reactive metallacycle intermediate. Upon the addition of the alkyne component, the cobalt center promotes the insertion of the unsaturated bond into the metal-carbon framework, followed by reductive elimination to forge the new C-C and C-N bonds essential for the isoquinolinone ring system. The presence of an oxidant, such as silver carbonate or copper sulfate, is crucial for regenerating the active cobalt species, thereby sustaining the catalytic cycle and driving the reaction to completion. This mechanistic pathway ensures high regioselectivity and minimizes the formation of side products, which is critical for maintaining the high-purity standards required in drug substance production.
From an impurity control perspective, the one-pot nature of the reaction inherently limits the accumulation of intermediate by-products that typically arise in stepwise syntheses. The specific choice of ligands, such as o-methoxytriphenylphosphine or triphenylphosphine, further tunes the electronic environment around the cobalt center, enhancing the reaction rate and selectivity for the desired polysubstituted product. Experimental data indicates that varying the molar ratios of reactants, specifically maintaining an anhydride to amine ratio of 1:1.3 and an anhydride to alkyne ratio of 1:1.5, optimizes the conversion efficiency. This precise control over reaction parameters allows manufacturers to achieve yields as high as 95%, as demonstrated in specific embodiments, ensuring that the final pharmaceutical intermediates meet rigorous quality specifications with minimal downstream purification efforts.
How to Synthesize Polysubstituted Isoquinolinone Derivatives Efficiently
To implement this cutting-edge synthesis in a production environment, operators must adhere to a specific sequence of addition and thermal management to maximize yield and safety. The process begins with the pre-stirring of the acid anhydride and amine components to establish the initial coordination complex before introducing the alkyne and catalytic system. Detailed standard operating procedures regarding stoichiometry, solvent selection, and workup protocols are essential for reproducibility. For a comprehensive guide on the exact experimental steps and conditions validated in the patent, please refer to the standardized synthesis protocol below.
- Pre-stir acid anhydride and amine in a reactor for 1-12 hours to ensure complete interaction before cyclization.
- Add alkyne, cobalt catalyst (e.g., cobalt iodide), ligand, oxidant, and solvent to the mixture.
- Heat the reaction mixture to 80°C-160°C for 0.5-36 hours, then isolate the product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this cobalt-catalyzed methodology translates into tangible strategic benefits that extend beyond simple chemistry. By shifting away from precious metal catalysts and complex multi-step sequences, companies can significantly de-risk their supply chains and improve their bottom line. The reliance on bulk commodity chemicals like acid anhydrides and alkynes ensures a stable supply of raw materials, mitigating the risks associated with sourcing specialized reagents. Furthermore, the simplified workflow reduces the burden on manufacturing facilities, allowing for faster turnaround times and more efficient utilization of reactor capacity. These factors collectively contribute to a more resilient and agile production model capable of meeting the dynamic demands of the global pharmaceutical market.
- Cost Reduction in Manufacturing: The substitution of expensive nickel or palladium catalysts with inexpensive cobalt salts represents a direct and significant saving in raw material costs. Additionally, the elimination of intermediate isolation steps reduces solvent usage, energy consumption, and labor hours associated with purification processes. This streamlined approach lowers the overall cost of goods sold (COGS), enabling more competitive pricing for the final pharmaceutical intermediates while maintaining healthy profit margins for manufacturers.
- Enhanced Supply Chain Reliability: Since the starting materials—acid anhydrides, amines, and alkynes—are widely available bulk chemicals, the risk of supply disruption is minimized compared to routes requiring custom-synthesized precursors. The robustness of the cobalt catalyst, which is insensitive to air and moisture, further simplifies logistics and storage requirements, removing the need for specialized handling equipment. This reliability ensures consistent production schedules and reducing lead time for high-purity pharmaceutical intermediates, allowing customers to bring their drugs to market faster.
- Scalability and Environmental Compliance: The reaction conditions are well-suited for large-scale operations, with temperatures and pressures that are easily manageable in standard industrial reactors. The high atom economy and reduced waste generation align with green chemistry principles, simplifying waste treatment and regulatory compliance. This scalability ensures that the process can seamlessly transition from pilot plant trials to commercial scale-up, supporting the growing demand for isoquinolinone-based therapeutics without environmental compromise.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These answers are derived directly from the experimental data and beneficial effects outlined in the patent documentation, providing clarity on the feasibility and advantages of adopting this technology for your specific production needs. Understanding these details is crucial for making informed decisions about process integration and supplier selection.
Q: What are the primary advantages of using cobalt catalysts over nickel in this synthesis?
A: Cobalt catalysts, such as cobalt iodide, offer significant cost advantages over nickel-based systems and exhibit superior stability against water and oxygen, simplifying operational requirements and reducing raw material costs.
Q: Can this one-pot method be applied to both aryl and alkyl alkynes?
A: Yes, the patented process demonstrates broad substrate scope, successfully accommodating both aryl-substituted alkynes and alkyl-substituted alkynes with high yields ranging from 70% to 95%.
Q: Is this synthetic route suitable for large-scale commercial production?
A: Absolutely. The use of bulk chemical raw materials, robust reaction conditions (up to 160°C), and inexpensive catalysts makes this method highly scalable for industrial manufacturing of pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polysubstituted Isoquinolinone Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this cobalt-catalyzed technology in accelerating the development of next-generation therapeutics. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project moves smoothly from concept to market. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of polysubstituted isoquinolinone derivatives delivered meets the highest industry standards for safety and efficacy.
We invite you to collaborate with us to leverage this advanced synthetic route for your upcoming projects. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this method can optimize your budget. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a reliable supply of these critical building blocks for your drug discovery pipeline.