Revolutionizing Halogenated Beta-Lactam Production: A Solvent-Free Aqueous Slurry Approach for Commercial Scale
Introduction to Advanced Halogenated Beta-Lactam Manufacturing
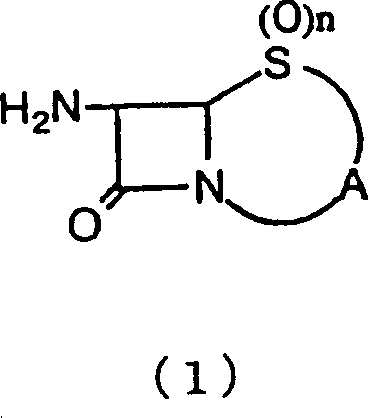
The pharmaceutical industry continuously seeks robust synthetic routes that balance high purity with environmental sustainability, particularly for critical antibiotic intermediates. Patent CN1125825C introduces a transformative process for producing halogenated beta-lactam compounds, specifically targeting the synthesis of key precursors like 6,6-dibromopenicillanic acid, an essential intermediate for the beta-lactamase inhibitor sulbactam. This technology represents a significant departure from legacy methods by utilizing an aqueous slurry system that completely avoids the use of hazardous halogenated organic solvents such as carbon tetrachloride. By reacting a beta-lactam-derived amino compound with nitrous acid or nitrites in the presence of halogen molecules under acidic conditions, the process achieves exceptional efficiency while adhering to modern green chemistry principles. The innovation lies in maintaining the reactants in a specific slurry dispersion state within water, which facilitates the reaction kinetics without the need for solubilizing agents that complicate downstream processing. This approach not only addresses the regulatory pressures surrounding volatile organic compounds (VOCs) but also provides a commercially viable pathway for the large-scale production of high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of halogenated beta-lactam derivatives has been plagued by significant operational and environmental challenges associated with solvent selection. Early methodologies, such as those described by Volkmann, relied heavily on chlorinated organic solvents like methylene chloride or carbon tetrachloride to facilitate the reaction between amino precursors and halogenating agents. While these solvents provided adequate solubility for the reactants, they introduced severe safety hazards and environmental liabilities, with carbon tetrachloride now being largely banned or strictly restricted due to its ozone-depleting potential and toxicity. Furthermore, alternative attempts to move towards greener solvents often resulted in drastic reductions in process efficiency; for instance, previous water-based methods reported yields as low as 34%, rendering them economically unfeasible for industrial application. The reliance on organic solvents also necessitates complex recovery systems and extensive waste treatment protocols to remove residual halogenated hydrocarbons from the final API intermediates, adding substantial cost and time to the manufacturing cycle. Consequently, there has been a persistent and urgent demand for a manufacturing protocol that can deliver high yields without compromising on safety or environmental compliance.
The Novel Approach
The patented process overcomes these historical barriers by establishing a unique reaction environment where water serves as the primary medium in a slurry dispersion state rather than a true solution. This novel approach leverages the insolubility of the beta-lactam amino compound in water to create a heterogeneous reaction system that surprisingly enhances the selectivity and yield of the halogenation reaction. By carefully controlling the addition of the substrate slurry into an acidic aqueous mixture containing halogen molecules and nitrites, the method achieves yields exceeding 90% in specific embodiments, a dramatic improvement over prior art aqueous methods. The elimination of organic solvents simplifies the work-up procedure significantly, as the product often precipitates directly from the reaction mixture upon completion, allowing for straightforward filtration and washing. This shift not only removes the cost burden associated with purchasing and disposing of tons of hazardous solvents but also mitigates the risk of solvent-related impurities contaminating the sensitive beta-lactam ring structure. Ultimately, this methodology aligns perfectly with the goals of a reliable pharmaceutical intermediates supplier aiming to deliver cost reduction in antibiotic manufacturing through process intensification and waste minimization.
Mechanistic Insights into Aqueous Phase Diazotization-Halogenation
The core chemical transformation in this process involves the in situ generation of reactive nitrosonium species from nitrites under acidic conditions, which subsequently interact with the primary amino group on the beta-lactam scaffold. In the presence of halogen molecules such as bromine or iodine, the reaction proceeds through a diazonium intermediate that is rapidly displaced by the halogen nucleophile to form the stable carbon-halogen bond at the alpha-position relative to the carbonyl. The use of strong acids like hydrobromic acid or sulfuric acid is critical not only for generating the nitrosating agent but also for maintaining the protonation state of the beta-lactam nitrogen, thereby protecting the strained four-membered ring from acid-catalyzed hydrolysis during the reaction window. The slurry state plays a pivotal mechanistic role by limiting the local concentration of the organic substrate in the aqueous phase, which helps to suppress side reactions such as over-halogenation or ring-opening that might occur in a homogeneous solution. This controlled heterogeneity ensures that the reaction occurs primarily at the interface or within the micro-environment of the suspended particles, leading to a cleaner impurity profile and higher overall material throughput. Understanding this interplay between phase transfer dynamics and chemical reactivity is essential for R&D teams looking to optimize the commercial scale-up of complex pharmaceutical intermediates.
Impurity control in this system is predominantly managed through precise thermal regulation and stoichiometric balancing of the halogenating agents. The patent specifies a narrow operating temperature range, preferably between -5°C and 6°C, which is vital for stabilizing the transient diazonium species and preventing thermal degradation of the sensitive beta-lactam core. Deviations above this range can lead to the formation of unidentified byproducts and a decrease in the stereochemical integrity of the final molecule, which is critical for biological activity. Additionally, the stepwise addition of the substrate slurry over an extended period, typically 12 to 24 hours, prevents the accumulation of excess heat and maintains a consistent ratio of reagents at the reaction site. Post-reaction treatment with reducing agents like sodium bisulfite effectively quenches any unreacted halogen, preventing oxidative damage to the product during isolation. These rigorous control parameters ensure that the resulting halogenated beta-lactam compounds meet the stringent purity specifications required for downstream coupling reactions in the synthesis of final drug substances.
How to Synthesize Halogenated Beta-Lactam Compounds Efficiently
The synthesis of these high-value intermediates requires strict adherence to the patented protocol to ensure reproducibility and safety on a manufacturing scale. The process begins with the preparation of a cooled acidic aqueous solution containing the necessary halogen and nitrite sources, followed by the controlled addition of the beta-lactam amino compound as a water slurry. Detailed operational parameters regarding reagent equivalents, stirring rates, and quenching procedures are critical for achieving the reported high yields and minimizing waste generation. For a comprehensive breakdown of the standardized operating procedures and specific equipment requirements, please refer to the technical guide below.
- Prepare an acidic aqueous mixture containing halogen molecules (e.g., bromine) and nitrite salts at low temperatures (-10 to 15°C).
- Create a slurry of the beta-lactam amino compound (Formula 1) in water and add it portion-wise to the reaction mixture while maintaining strict temperature control.
- Quench excess halogen with bisulfite, filter the precipitated product, and purify via recrystallization to obtain the target halogenated derivative.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, this manufacturing technology offers profound benefits that extend beyond simple yield improvements, directly impacting the total cost of ownership for pharmaceutical buyers. By transitioning away from regulated and expensive halogenated solvents, manufacturers can significantly reduce raw material costs and eliminate the logistical complexities associated with hazardous waste disposal. The reliance on water as the primary reaction medium drastically simplifies the effluent treatment process, lowering the environmental compliance burden and reducing the risk of supply chain disruptions caused by regulatory changes on solvent usage. Furthermore, the use of commodity chemicals such as sodium nitrite and hydrobromic acid ensures a stable and resilient supply chain, as these reagents are readily available from multiple global vendors, reducing dependency on single-source specialty chemical suppliers. This robustness makes the process highly attractive for long-term procurement contracts where supply continuity is paramount.
- Cost Reduction in Manufacturing: The elimination of organic solvents like carbon tetrachloride and methylene chloride removes a major cost center associated with solvent purchase, recovery, and incineration. Without the need for complex solvent recycling infrastructure, capital expenditure for new production lines is substantially lowered, and operational expenses are reduced due to simpler utility requirements. The high yield achieved in the aqueous slurry system means that less starting material is wasted, directly improving the atom economy and reducing the cost per kilogram of the final active pharmaceutical ingredient intermediate. Additionally, the simplified work-up procedure involving direct filtration reduces labor hours and energy consumption compared to multi-step extraction and distillation processes required in organic solvent methods.
- Enhanced Supply Chain Reliability: Utilizing water as the main solvent mitigates risks associated with the volatility of petrochemical-derived solvent markets, ensuring more predictable pricing and availability. The process operates under mild pressure and standard atmospheric conditions, allowing for flexible manufacturing in existing multipurpose facilities without requiring specialized high-pressure reactors. This flexibility enables faster response times to market demand fluctuations, as production batches can be initiated quickly without lengthy solvent qualification or drying steps. Moreover, the stability of the reagents used allows for safer storage and handling, reducing the likelihood of accidents or shutdowns that could interrupt the supply of critical antibiotic intermediates to downstream customers.
- Scalability and Environmental Compliance: The aqueous nature of the reaction inherently aligns with green chemistry initiatives, making it easier to obtain environmental permits for capacity expansion in regions with strict emission standards. Waste streams generated are primarily aqueous and saline, which are significantly easier and cheaper to treat than mixed organic-halogen waste streams that require specialized incineration. The process demonstrates excellent scalability, as heat transfer and mixing in slurry systems are well-understood engineering challenges that can be managed effectively from pilot plant to multi-ton commercial production. This scalability ensures that the technology can support the growing global demand for beta-lactam antibiotics without hitting bottlenecks related to solvent availability or waste treatment capacity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this aqueous slurry halogenation technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this route into their existing supply chains.
Q: Why is the aqueous slurry method superior to traditional organic solvent methods for beta-lactam halogenation?
A: Traditional methods rely on toxic solvents like carbon tetrachloride or methylene chloride, which pose severe environmental and safety risks. The aqueous slurry method eliminates these hazards while achieving significantly higher yields compared to previous water-based attempts.
Q: What are the critical reaction conditions for maximizing yield in this process?
A: Maintaining a low reaction temperature between -5°C and 6°C is crucial to prevent decomposition. Additionally, keeping the reactants in a slurry dispersion state ensures efficient contact without requiring solubilizing organic co-solvents.
Q: Can this process be scaled for industrial production of sulbactam intermediates?
A: Yes, the process is designed for industrial viability. By using water as the main solvent and commodity reagents like sodium nitrite and hydrobromic acid, it offers a cost-effective and scalable route for manufacturing key antibiotic intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Halogenated Beta-Lactam Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting sustainable and efficient synthetic routes to maintain competitiveness in the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and robust. We are committed to delivering high-purity halogenated beta-lactam intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. By leveraging advanced manufacturing technologies like the aqueous slurry process, we provide our partners with a secure supply of essential building blocks for next-generation antibiotics.
We invite you to engage with our technical procurement team to discuss how our capabilities can align with your specific project requirements. Contact us today to request a Customized Cost-Saving Analysis tailored to your volume needs, and let us provide you with specific COA data and route feasibility assessments. Together, we can drive efficiency and innovation in the production of life-saving medicines.
