Advanced Catalytic Oxidation for 2,2'-Dithiodibenzothiazole: Scaling Green Chemistry for Global Markets
Advanced Catalytic Oxidation for 2,2'-Dithiodibenzothiazole: Scaling Green Chemistry for Global Markets
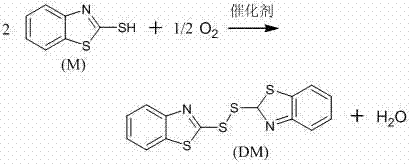
The global demand for high-purity heterocyclic intermediates is driving a significant shift towards greener, more sustainable manufacturing processes. Patent CN102167686A introduces a breakthrough methodology for the preparation of 2,2'-dithiodibenzothiazole (DM) through the catalytic oxidation of 2-mercaptobenzothiazole (M) using molecular oxygen. This technology represents a pivotal advancement over traditional chemical oxidation routes, addressing critical pain points regarding environmental compliance, waste management, and product quality. By utilizing a composite catalyst system comprising transition metal salts and organic ligands, this process operates efficiently under mild conditions, yielding a product with exceptional purity suitable for both rubber acceleration and pharmaceutical synthesis. For R&D directors and procurement leaders, understanding the mechanistic advantages of this aerobic oxidation pathway is essential for securing a competitive edge in the supply of reliable pharmaceutical intermediates and specialty chemicals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of 2,2'-dithiodibenzothiazole has relied heavily on stoichiometric oxidants such as sodium nitrite or chlorine gas. These conventional pathways are fraught with severe operational and environmental drawbacks that modern supply chains can ill afford. The use of inorganic oxidants inevitably generates substantial quantities of saline wastewater, which is notoriously difficult and expensive to treat, posing a significant liability under increasingly stringent global environmental regulations. Furthermore, these reactions often produce harmful gaseous by-products, including nitrogen oxides and hydrogen chloride, necessitating complex scrubbing systems and increasing capital expenditure. From a quality perspective, products derived from these harsh chemical oxidations frequently suffer from lower melting points and higher impurity profiles, rendering them unsuitable for high-value pharmaceutical applications where strict specification limits are mandatory.
The Novel Approach
In stark contrast, the technology disclosed in CN102167686A leverages molecular oxygen or air as the terminal oxidant, fundamentally altering the economic and ecological footprint of the synthesis. This catalytic aerobic oxidation proceeds cleanly, producing water as the sole by-product, thereby eliminating the generation of salty wastewater entirely. The process utilizes a sophisticated catalyst system formed in situ from transition metal salts and nitrogen or oxygen-containing ligands, which activates molecular oxygen with high efficiency. This approach not only simplifies the post-treatment workflow by removing the need for extensive washing to remove inorganic salts but also ensures a product profile with superior purity and melting point characteristics. The elimination of acidic or basic additives further streamlines the process, making it an ideal candidate for cost reduction in pharmaceutical intermediate manufacturing where impurity control is paramount.
Mechanistic Insights into Transition Metal-Ligand Catalyzed Aerobic Oxidation
The core innovation of this process lies in the synergistic interaction between the transition metal center and the organic ligand, which creates a highly active catalytic species capable of activating the relatively stable oxygen-oxygen bond. The patent specifies a range of transition metals, including Cobalt, Copper, Iron, and Manganese, paired with ligands such as acetylacetone, oxalic acid, or TEMPO derivatives. This coordination complex facilitates a redox cycle where the metal center shuttles electrons between the thiol substrate and molecular oxygen. Mechanistically, the thiol is likely deprotonated or coordinated to the metal center, forming a metal-thiolate intermediate that is subsequently oxidized by the activated oxygen species to form the disulfide bond. This controlled radical or ionic pathway ensures high atom economy and prevents the over-oxidation of the sulfur moiety to sulfones or sulfoxides, which are common impurities in less selective oxidation protocols.
From an impurity control perspective, the high selectivity of this catalytic system is a major advantage for R&D teams focused on developing robust synthetic routes. The absence of strong acids or bases prevents side reactions such as ring opening or hydrolysis of the benzothiazole scaffold, which can occur under the harsh conditions of traditional nitrite oxidation. Furthermore, the ability to tune the catalyst composition by adjusting the metal-to-ligand ratio allows for fine-tuning of the reaction kinetics to match specific reactor configurations. This tunability ensures that the formation of oligomeric sulfur by-products is minimized, resulting in a crude product that requires minimal purification. For manufacturers aiming for commercial scale-up of complex heterocyclic intermediates, this level of mechanistic control translates directly into consistent batch-to-batch quality and reduced downstream processing costs.
How to Synthesize 2,2'-Dithiodibenzothiazole Efficiently
Implementing this catalytic oxidation route requires careful attention to reaction parameters to maximize yield and catalyst longevity. The process typically involves dissolving the starting material, 2-mercaptobenzothiazole, in a polar organic solvent such as methanol, ethanol, or acetonitrile. The catalyst components are introduced either as a pre-formed complex or as separate salts and ligands that generate the active species in situ upon heating. The reaction is conducted under pressurized oxygen or air, with temperatures ranging from ambient to 120°C depending on the specific catalyst activity. A key operational feature highlighted in the patent is the recyclability of the reaction mother liquor; after filtering the solid product, the filtrate containing the dissolved catalyst can be reused for multiple subsequent batches. Detailed standardized synthesis steps and specific parameter optimizations for different metal systems are outlined below.
- Prepare the catalyst system by mixing a transition metal salt (such as Cobalt, Copper, or Iron salts) with an organic ligand containing nitrogen or oxygen in a suitable solvent like methanol or ethanol.
- Charge the reactor with 2-mercaptobenzothiazole (M) and the prepared catalyst solution, then heat the mixture to a temperature range of 20-120°C while maintaining oxygen or air pressure between 0.01-2.00 MPa.
- After the reaction completes (typically 1-30 hours), cool the mixture, filter to separate the solid product, and recycle the mother liquor for subsequent batches to maximize efficiency.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this catalytic technology offers transformative benefits that extend far beyond simple yield improvements. The shift from stoichiometric chemical oxidants to catalytic aerobic oxidation fundamentally restructures the cost basis of production by eliminating the purchase of expensive and hazardous oxidizing agents. Moreover, the drastic reduction in waste generation lowers the burden on effluent treatment plants, resulting in significant operational expenditure savings related to environmental compliance and waste disposal fees. The simplicity of the work-up procedure, which primarily involves filtration and solvent recovery, reduces labor intensity and shortens the overall production cycle time. These factors combine to create a more resilient and cost-effective supply chain capable of meeting the rigorous demands of global pharmaceutical and agrochemical markets.
- Cost Reduction in Manufacturing: The economic model of this process is driven by the elimination of costly raw materials and the maximization of resource efficiency. By replacing sodium nitrite or chlorine with air or oxygen, the direct material cost is drastically simplified, as oxygen is essentially free and abundant. Furthermore, the patent demonstrates that the catalyst system does not require precious metals, utilizing abundant transition metals like iron or copper instead, which stabilizes pricing against volatile noble metal markets. The ability to reuse the mother liquor multiple times without replenishing the catalyst means that the effective catalyst cost per kilogram of product approaches zero over a production campaign. Additionally, the high recovery rates of solvents like methanol and ethanol through distillation further compress the variable costs of production, ensuring a highly competitive price point for high-purity 2,2'-dithiodibenzothiazole.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the regulatory restrictions surrounding hazardous chemicals used in traditional synthesis. By removing the dependency on chlorine gas and sodium nitrite, manufacturers mitigate the risk of supply disruptions caused by transportation bans or stricter safety regulations on hazardous goods. The raw materials required for this catalytic process—transition metal salts and common organic solvents—are commodity chemicals with robust, multi-vendor global supply chains, ensuring consistent availability. The operational simplicity of the process also reduces the likelihood of unplanned downtime due to equipment corrosion or fouling, which are common issues in acidic or saline oxidation environments. This reliability is crucial for reducing lead time for high-purity rubber additives and pharmaceutical intermediates, allowing buyers to maintain leaner inventory levels with confidence.
- Scalability and Environmental Compliance: As environmental regulations tighten globally, the ability to scale production without increasing the environmental footprint is a critical strategic asset. This technology is inherently green, producing water as the only by-product and avoiding the generation of saline wastewater that requires energy-intensive evaporation or treatment. The absence of corrosive acids and bases extends the lifespan of reactor vessels and piping, reducing capital maintenance costs during scale-up from pilot to commercial tonnage. The process operates at moderate pressures and temperatures that are easily manageable in standard stainless steel reactors, facilitating seamless technology transfer to large-scale manufacturing facilities. This alignment with green chemistry principles not only future-proofs the supply chain against regulatory changes but also enhances the brand value of the end products in markets that prioritize sustainability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this catalytic oxidation technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for evaluating the feasibility of this route for your specific application needs. Understanding these nuances is vital for technical teams assessing the compatibility of this method with existing infrastructure and quality standards.
Q: How does this catalytic method improve environmental compliance compared to traditional oxidation?
A: Traditional methods often use sodium nitrite or chlorine gas, generating large amounts of salty wastewater and harmful gases like nitrogen oxides. This patented catalytic method uses molecular oxygen and produces water as the only byproduct, eliminating saline waste streams and significantly reducing environmental treatment costs.
Q: Can the catalyst and solvent be reused to lower production costs?
A: Yes, the patent explicitly demonstrates that the mother liquor containing the catalyst can be reused multiple times (up to 10 cycles in examples) without adding fresh catalyst. Additionally, solvents like methanol and ethanol can be recovered via distillation, leading to substantial reductions in raw material consumption.
Q: Is the product purity sufficient for pharmaceutical applications?
A: The method achieves high selectivity with minimal by-products. Experimental data in the patent shows product purity reaching 99.5% with melting points consistent with high-grade standards (180-182°C), making it suitable not just for rubber acceleration but also as a critical intermediate for beta-lactam and cephalosporin antibiotics.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,2'-Dithiodibenzothiazole Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced catalytic processes requires a partner with deep technical expertise and proven manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN102167686A are fully realized in practical, large-volume manufacturing. We operate stringent purity specifications and maintain rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of 2,2'-dithiodibenzothiazole meets the exacting standards required for pharmaceutical and high-performance rubber applications. Our commitment to process excellence ensures that we can deliver consistent quality while adhering to the highest environmental and safety protocols.
We invite you to collaborate with us to explore how this green oxidation technology can optimize your supply chain and reduce your overall cost of goods. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data from our pilot runs and comprehensive route feasibility assessments to demonstrate how our advanced manufacturing capabilities can support your long-term strategic goals in the fine chemicals sector.
