Advanced Synthesis of Large Stokes Shift NIR Fluorescent Molecules for Bio-Imaging
The landscape of fluorescent imaging technology is undergoing a significant transformation driven by the demand for deeper tissue penetration and higher signal-to-noise ratios in biological assays. Patent CN113416203B introduces a groundbreaking class of near-infrared emitting thiadiazoloquinoxaline fluorescent molecules that address the critical limitations of conventional dyes. Unlike traditional fluorophores such as fluorescein or rhodamine, which suffer from small Stokes shifts and severe spectral overlap, this novel chemical architecture leverages a sophisticated intramolecular proton hydrogen transfer mechanism combined with an electron push-pull system. 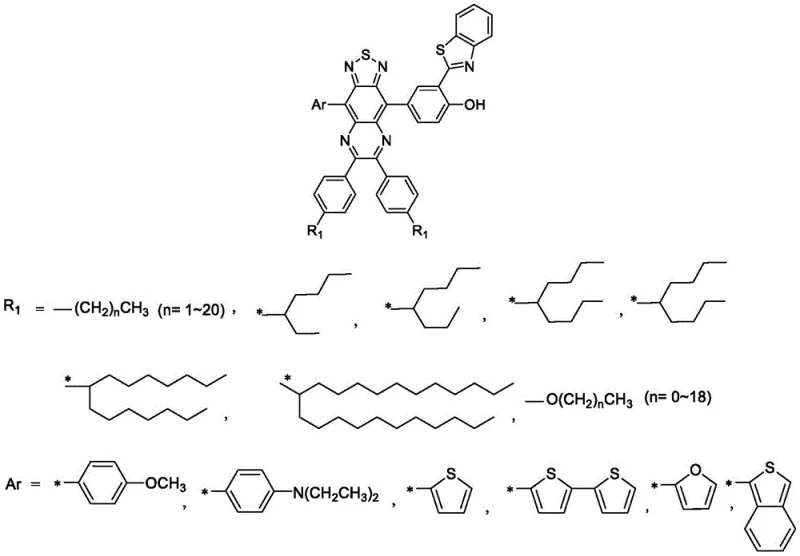 . This strategic molecular design results in a maximum fluorescence emission wavelength exceeding 724 nm and a Stokes shift greater than 138 nm, effectively separating excitation and emission spectra to minimize self-quenching. For research directors and procurement specialists seeking a reliable pharma intermediates supplier, this technology represents a pivotal advancement in the development of high-performance bioluminescent probes and bio-imaging agents.
. This strategic molecular design results in a maximum fluorescence emission wavelength exceeding 724 nm and a Stokes shift greater than 138 nm, effectively separating excitation and emission spectra to minimize self-quenching. For research directors and procurement specialists seeking a reliable pharma intermediates supplier, this technology represents a pivotal advancement in the development of high-performance bioluminescent probes and bio-imaging agents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the field of organic dye synthesis has been constrained by the inherent physical properties of established fluorophore families like oxazines and cyanines. These conventional dyes typically exhibit Stokes shifts of less than 30 nm, leading to substantial overlap between their excitation and emission spectra which drastically lowers the signal-to-noise ratio during imaging processes. Furthermore, existing methods to improve these metrics, such as expanding the conjugated plane of xanthene dyes, often result in multi-step synthetic routes with low total yields and limited improvements in Stokes displacement. The complexity of purifying these traditional dyes to the stringent purity specifications required for in-vivo applications often introduces significant bottlenecks in the supply chain. Additionally, the reliance on complex structural modifications to achieve red-shifted emission frequently compromises the photostability and quantum yield of the final product, limiting their utility in long-term biological tracking and real-time nondestructive detection scenarios.
The Novel Approach
The patented methodology overcomes these historical barriers by utilizing a thiadiazoloquinoxaline nucleus, which acts as a robust electron-deficient core capable of forming a highly efficient electron push-pull system when paired with electron-donating units. This approach not only facilitates a significant red-shift in fluorescence emission into the near-infrared region but also integrates an Excited State Intramolecular Proton Transfer (ESIPT) component via a 2-benzothiazolyl-phenol moiety. This dual-mechanism strategy ensures that the final product possesses both a large Stokes shift and superior light stability, effectively solving the self-quenching issues prevalent in prior art. By employing a modular two-step Suzuki coupling reaction, the synthesis allows for the convenient regulation of fluorescence properties simply by varying the electron-donating aryl group, offering unparalleled flexibility for customizing optical characteristics without redesigning the entire synthetic pathway.
Mechanistic Insights into Suzuki-Catalyzed Cyclization and Functionalization
The core of this innovative synthesis lies in the precise application of palladium-catalyzed cross-coupling reactions to construct the complex conjugated system required for near-infrared emission. The process initiates with the activation of the 4,9-dibromo-thiadiazoloquinoxaline precursor, where the electron-withdrawing nature of the thiadiazole ring enhances the reactivity of the bromine positions towards oxidative addition. 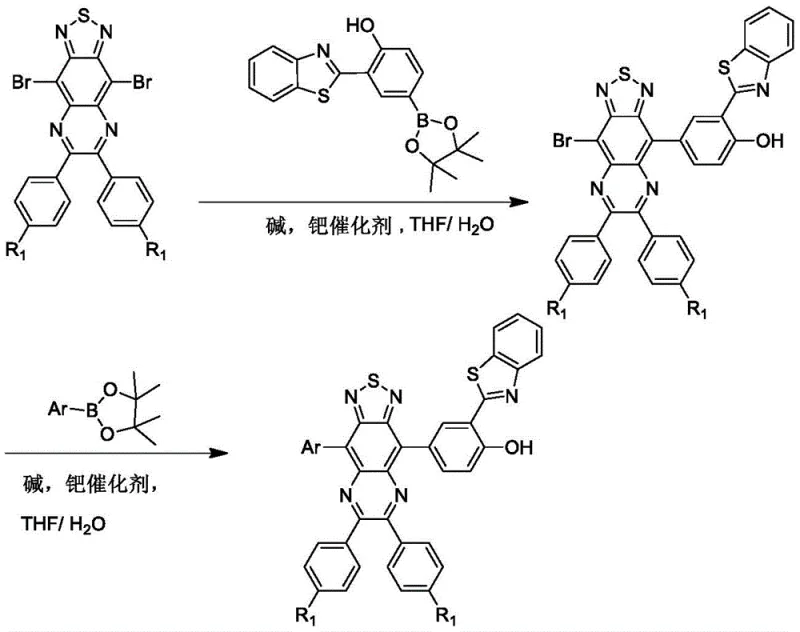 . In the first stage, the reaction with 2-benzothiazolyl-4-boronate-phenol establishes the critical ESIPT unit, creating an intermediate that retains a reactive bromine handle for further functionalization. The use of mild alkaline aqueous solutions and tetrahydrofuran as a co-solvent ensures that the sensitive boronic ester groups remain stable while facilitating the transmetallation step essential for carbon-carbon bond formation.
. In the first stage, the reaction with 2-benzothiazolyl-4-boronate-phenol establishes the critical ESIPT unit, creating an intermediate that retains a reactive bromine handle for further functionalization. The use of mild alkaline aqueous solutions and tetrahydrofuran as a co-solvent ensures that the sensitive boronic ester groups remain stable while facilitating the transmetallation step essential for carbon-carbon bond formation.
Impurity control in this synthesis is managed through the high selectivity of the palladium catalyst system, which minimizes homocoupling side reactions often seen in complex biaryl syntheses. The second coupling step introduces the final electron-donating aryl group, completing the push-pull electronic circuit that drives the red-shifted emission. By carefully selecting ligands such as Pd(PPh3)4 or Pd(dppf)Cl2, the reaction maintains high efficiency even with sterically hindered substrates, ensuring a clean impurity profile that simplifies downstream purification. This mechanistic robustness is crucial for maintaining the batch-to-batch consistency required for clinical-grade imaging agents, as it prevents the formation of non-fluorescent byproducts that could interfere with biological assays or complicate regulatory approval processes for new diagnostic tools.
How to Synthesize Near-Infrared Thiadiazoloquinoxaline Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for producing these advanced fluorescent materials with high reproducibility and yield. The procedure relies on standard laboratory equipment and readily available reagents, making it accessible for both pilot-scale development and full commercial manufacturing. The key to success lies in maintaining strict inert gas protection throughout the reaction to prevent oxidation of the palladium catalyst and the sensitive boronic acid intermediates. Detailed standardized synthesis steps for this process are provided in the guide below, ensuring that technical teams can replicate the high-purity specifications necessary for sensitive bio-imaging applications.
- Perform the first Suzuki coupling between 4,9-dibromo-thiadiazoloquinoxaline and 2-benzothiazolyl-4-boronate-phenol using a palladium catalyst in THF/water.
- Execute the second Suzuki coupling by reacting the intermediate bromo-compound with an electron-donating aryl boronate ester under inert gas protection.
- Purify the final near-infrared emission product via column chromatography to achieve high purity specifications suitable for bio-imaging applications.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel thiadiazoloquinoxaline synthesis route offers substantial strategic benefits regarding cost structure and supply reliability. The elimination of complex, multi-step sequences found in traditional xanthene or rhodamine synthesis significantly streamlines the manufacturing process, reducing the overall consumption of solvents and energy. By utilizing a convergent two-step strategy, the production timeline is drastically shortened, allowing for faster response times to market demands for specialized bio-imaging reagents. Furthermore, the modularity of the second coupling step means that a single intermediate can be diversified into a library of products with varying emission profiles, maximizing the utility of inventory and reducing the risk of obsolescence for specific SKUs.
- Cost Reduction in Manufacturing: The synthesis avoids the use of exotic or prohibitively expensive reagents, relying instead on commodity chemicals like tetrahydrofuran, water, and standard palladium catalysts which are widely available in the global chemical market. The mild reaction conditions, operating between 50°C and 80°C, eliminate the need for energy-intensive heating or cryogenic cooling, leading to significant operational expenditure savings. Additionally, the high selectivity of the Suzuki coupling reduces the burden on purification resources, as less chromatographic separation is required to achieve the necessary purity levels compared to older dye synthesis methods.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, including dibromo-thiadiazoloquinoxaline derivatives and various aryl boronic esters, are sourced from stable and mature supply chains, mitigating the risk of raw material shortages. The robustness of the reaction conditions ensures high batch consistency, which is critical for maintaining long-term contracts with pharmaceutical and diagnostic clients who require rigorous quality assurance. This stability allows for better forecasting and inventory planning, ensuring that critical imaging agents are available without interruption even during periods of high market volatility.
- Scalability and Environmental Compliance: The use of aqueous alkaline solutions and common organic solvents facilitates easier waste treatment and compliance with increasingly stringent environmental regulations regarding hazardous waste disposal. The process is inherently scalable from gram-scale laboratory synthesis to multi-ton commercial production without requiring fundamental changes to the reaction engineering, supporting seamless capacity expansion. This scalability ensures that the supply chain can adapt quickly to surging demand for near-infrared probes in emerging fields like intraoperative imaging and deep-tissue diagnostics.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these advanced fluorescent molecules. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation to ensure accuracy and relevance for industry stakeholders. Understanding these details is essential for evaluating the feasibility of integrating this technology into existing product lines or research pipelines.
Q: What is the primary advantage of this thiadiazoloquinoxaline derivative?
A: The molecule exhibits a large Stokes shift exceeding 138 nm and near-infrared emission over 724 nm, achieved through intramolecular proton transfer and electron push-pull systems, which significantly reduces self-quenching in biological imaging.
Q: How is the fluorescence emission range regulated in this synthesis?
A: The emission range is conveniently adjusted by modifying the electron-donating aryl group introduced in the second step, allowing for tunable optical properties without altering the core synthetic pathway.
Q: Is this synthesis route scalable for industrial production?
A: Yes, the process utilizes standard Suzuki coupling conditions with mild temperatures (50-80°C) and common solvents like THF and water, making it highly amenable to commercial scale-up and consistent batch production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Near-Infrared Thiadiazoloquinoxaline Supplier
As the demand for high-performance bio-imaging agents continues to grow, partnering with an experienced CDMO partner is essential for navigating the complexities of commercializing novel fluorescent materials. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, guaranteeing that every batch of thiadiazoloquinoxaline derivative meets the exacting standards required for sensitive biological applications and regulatory submissions.
We invite you to contact our technical procurement team to discuss how we can support your specific requirements for cost reduction in fluorescent probe manufacturing. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our optimized synthesis routes can lower your overall cost of goods. We encourage you to reach out today to obtain specific COA data and route feasibility assessments tailored to your project's unique challenges and timelines.
