Advanced Synthesis of Coupled Double-BODIPY Near-Infrared Dyes for Commercial Scale-Up
Advanced Synthesis of Coupled Double-BODIPY Near-Infrared Dyes for Commercial Scale-Up
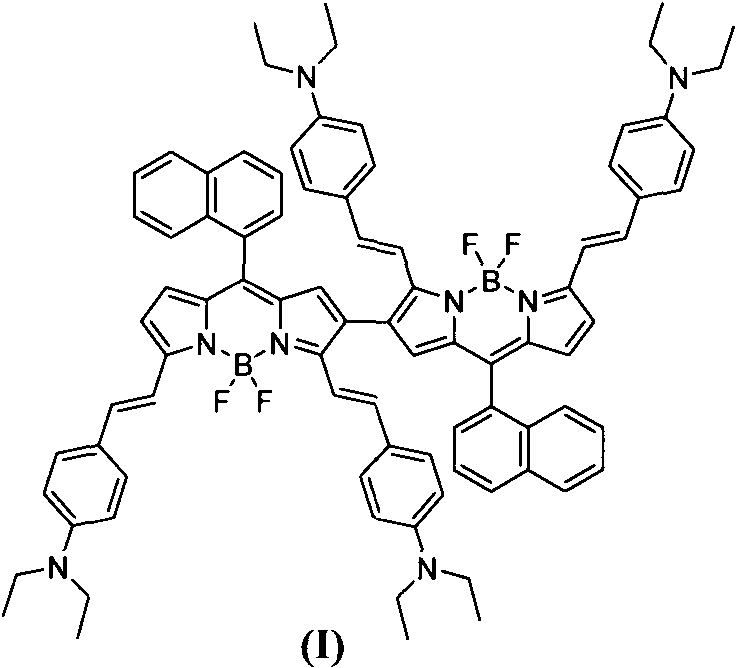
The rapid evolution of optoelectronic materials and biomedical imaging technologies has created an urgent demand for high-performance near-infrared (NIR) absorption dyes that offer superior stability and tunable spectral properties. Patent CN113150017A introduces a groundbreaking methodology for the preparation of coupled double-BODIPY near-infrared absorption dyes, utilizing a direct oxidative coupling strategy that bypasses the limitations of traditional cross-coupling reactions. This technical breakthrough enables the formation of C-C covalent bonds at the beta position of the BODIPY parent body under the catalytic action of FeCl3 and CH3NO2, resulting in a dimeric structure with exceptional photophysical characteristics. The resulting Compound I demonstrates a maximum absorption wavelength red-shifted to 796nm, positioning it as a critical component for next-generation organic solar cells and deep-tissue biological fluorescence imaging. As a reliable electronic chemical supplier, understanding the nuances of this synthetic route is essential for integrating these advanced materials into commercial supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the expansion of the pi-conjugated system in BODIPY molecules to achieve near-infrared absorption has relied heavily on palladium-catalyzed cross-coupling reactions, such as Suzuki or Sonogashira couplings. These conventional pathways typically require the prior introduction of halogen atoms at the 2,6-positions of the BODIPY core, followed by coupling with aromatic groups, which inherently increases the number of synthetic steps and overall process complexity. Furthermore, the reliance on precious metal catalysts like palladium introduces significant cost burdens and necessitates rigorous downstream purification processes to remove trace heavy metals, which is particularly critical for biomedical applications. The toxicity associated with certain organometallic reagents and the sensitivity of these reactions to oxygen and moisture often result in lower yields and inconsistent batch-to-batch reproducibility, creating bottlenecks for cost reduction in electronic chemical manufacturing. Additionally, the structural modifications achievable through these methods often struggle to push the absorption wavelength sufficiently into the deep near-infrared region above 750nm without compromising the stability of the fluorophore.
The Novel Approach
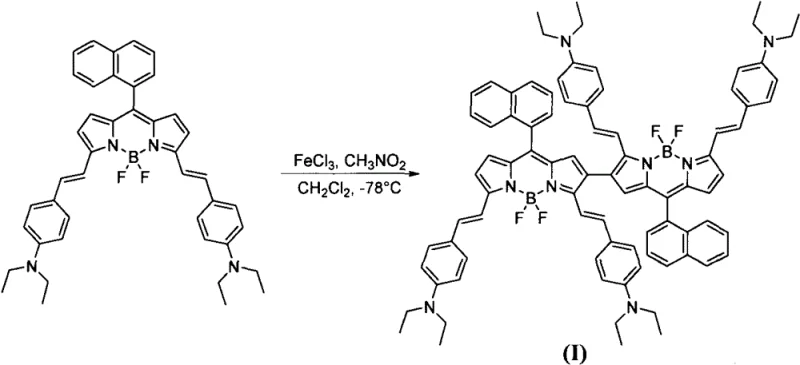
In stark contrast, the novel approach detailed in the patent data utilizes a direct oxidative coupling mechanism mediated by ferric chloride (FeCl3) in a nitromethane and dichloromethane solvent system. This method allows for the direct formation of C-C covalent bonds between two monomer BODIPY units at their beta positions, effectively doubling the conjugated system in a single synthetic operation. By operating at cryogenic temperatures initially (-78°C) and warming to room temperature, the reaction achieves high selectivity for the desired dimer while minimizing side reactions. This streamlined process eliminates the need for pre-functionalization with halogens and avoids the use of expensive transition metal catalysts, thereby drastically simplifying the workflow. The ability to synthesize these complex dimeric structures with good selectivity and under relatively mild conditions represents a paradigm shift in how high-performance NIR dyes can be produced commercially.
Mechanistic Insights into FeCl3-Catalyzed Oxidative Coupling
The core of this innovation lies in the Lewis acid and oxidizing properties of iron(III) chloride, which facilitates the generation of radical cation intermediates on the electron-rich BODIPY monomer. When the monomer BODIPY derivative, specifically 3,5-((4-N,N-diethyl)-divinylbenzene)-8-(1-naphthalene)-BODIPY, is exposed to FeCl3 in nitromethane, single electron transfer occurs, generating a reactive species that undergoes dimerization at the most nucleophilic beta-position of the pyrrole ring. The use of nitromethane as a co-solvent is crucial, as it stabilizes the charged intermediates and enhances the solubility of the inorganic catalyst in the organic phase, ensuring a homogeneous reaction environment. The molar ratio of the monomer to FeCl3 is carefully controlled between 1:10 and 1:15 to drive the equilibrium towards the coupled product while preventing over-oxidation or polymerization. This mechanistic pathway not only extends the conjugation length significantly but also rigidifies the molecular structure, which contributes to the observed narrowing of the absorption peak and the enhancement of the molar extinction coefficient.
From an impurity control perspective, this oxidative coupling mechanism offers distinct advantages over step-wise condensation reactions. Because the reaction proceeds through a specific radical coupling pathway rather than a multi-step substitution, the impurity profile is generally cleaner, dominated primarily by unreacted starting material or minor oligomers that can be easily separated. The subsequent workup involves a simple quench with saturated sodium bicarbonate solution, which neutralizes the acidic byproducts and precipitates iron salts, allowing for easy separation of the organic layer. Purification via silica gel column chromatography using 100% dichloromethane as the eluent effectively isolates the black-brown solid product (Compound I) with high purity. This robustness in impurity management is vital for R&D directors who require materials with consistent optical properties for sensitive applications like biological sensing, where background fluorescence from impurities can compromise data integrity.
How to Synthesize Coupled Double-BODIPY Efficiently
The synthesis of this high-value near-infrared dye is designed to be operationally straightforward, leveraging standard laboratory equipment and readily available reagents to ensure ease of technology transfer from lab to pilot plant. The process begins with the rigorous exclusion of water and oxygen, utilizing dried dichloromethane and an inert atmosphere to protect the sensitive radical intermediates during the initial low-temperature phase. Precise control of the stoichiometry, particularly the excess of the iron catalyst and the nitromethane solvent, is key to maximizing the conversion of the monomer into the desired dimeric structure. While the patent examples demonstrate yields ranging from 18% to 29% on a small scale, these figures serve as a baseline for further optimization during commercial scale-up, where mixing efficiency and heat transfer can be improved to enhance throughput. The detailed standardized synthesis steps for replicating this high-purity electronic chemical are outlined in the guide below.
- Prepare anhydrous dichloromethane and monomer BODIPY derivative in a round-bottom flask under inert atmosphere, cooling the system to -78°C.
- Dropwise add a solution of FeCl3 in nitromethane to the reaction mixture, maintaining the molar ratio of monomer to catalyst between 1: 10 and 1:15.
- Warm the reaction to room temperature, quench with saturated sodium bicarbonate, and purify the resulting black-brown solid via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition from palladium-catalyzed methods to this iron-mediated oxidative coupling offers substantial strategic benefits regarding cost stability and sourcing reliability. The elimination of precious metal catalysts removes exposure to the volatile pricing markets associated with platinum group metals, leading to more predictable raw material costs over the long term. Furthermore, the simplified synthetic route reduces the total number of processing steps, which directly correlates to lower labor costs, reduced energy consumption, and decreased solvent usage per kilogram of final product. This efficiency gain is critical for achieving cost reduction in display material manufacturing, allowing end-users to integrate advanced NIR capabilities into their products without prohibitive expense. The use of common industrial solvents like dichloromethane and nitromethane ensures that supply chains remain resilient, as these chemicals are produced at massive scales globally and are not subject to the same geopolitical supply risks as specialized ligands or catalysts.
- Cost Reduction in Manufacturing: The replacement of expensive palladium catalysts with abundant iron salts fundamentally alters the cost structure of the synthesis, removing the need for costly scavengers to remove heavy metal residues from the final product. This simplification of the downstream processing significantly lowers the operational expenditure required to produce high-purity batches, as fewer purification columns and less specialized waste treatment are needed. Additionally, the high atom economy of the direct coupling reaction means that a larger proportion of the starting material ends up in the final valuable product, minimizing waste disposal costs. These factors combine to create a manufacturing process that is inherently more economical and sustainable, providing a competitive edge in price-sensitive markets.
- Enhanced Supply Chain Reliability: By relying on commodity chemicals such as FeCl3 and standard organic solvents, the production of this coupled double-BODIPY dye is decoupled from the supply constraints often faced by specialty catalytic reagents. This ensures a continuous and stable supply of raw materials, reducing the risk of production delays caused by vendor shortages or logistics bottlenecks. The robustness of the reaction conditions, which tolerate a range of temperatures during the warm-up phase, also makes the process more forgiving and easier to manage in large-scale reactors, further enhancing supply continuity. For supply chain planners, this translates to shorter lead times for high-purity electronic chemicals and greater confidence in meeting delivery schedules for downstream clients.
- Scalability and Environmental Compliance: The mild reaction conditions and the absence of toxic heavy metals make this process highly scalable and environmentally compliant, aligning with increasingly stringent global regulations on chemical manufacturing. The waste stream generated is primarily composed of iron salts and organic solvents, which are well-understood and easier to treat compared to the complex hazardous waste associated with palladium chemistry. This environmental advantage facilitates faster regulatory approvals for new facilities and reduces the liability associated with long-term waste management. Consequently, the commercial scale-up of complex optoelectronic materials becomes more feasible, allowing manufacturers to rapidly increase capacity from pilot batches to multi-ton production to meet growing market demand.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this novel near-infrared dye, based on the specific data provided in the patent documentation. Understanding these details helps stakeholders evaluate the feasibility of integrating this material into their specific product lines, whether for solar energy harvesting or medical diagnostics. The answers reflect the balance between high-performance specifications and practical manufacturing considerations inherent in this new synthetic route.
Q: What are the primary advantages of the FeCl3-catalyzed method over traditional palladium coupling?
A: The FeCl3-catalyzed oxidative coupling eliminates the need for expensive palladium catalysts and halogenated precursors, significantly reducing raw material costs and simplifying the purification process by avoiding heavy metal removal steps.
Q: What is the absorption range of the synthesized coupled double-BODIPY dye?
A: The synthesized dye exhibits a strongest electronic absorption spectrum red-shifted to 796nm, with a narrow absorption peak in the near-infrared region and a molar extinction coefficient greater than 2.5 x 10^5 cm^-1 mol^-1.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the method utilizes mild reaction conditions (-78°C to room temperature) and common solvents like dichloromethane and nitromethane, making it highly adaptable for commercial scale-up with robust safety and environmental profiles.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Coupled Double-BODIPY Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the coupled double-BODIPY near-infrared absorption dye in advancing technologies ranging from organic photovoltaics to deep-tissue imaging. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including precise analysis of absorption maxima and molar extinction coefficients to guarantee batch-to-batch consistency. We understand that for applications in biological fluorescence imaging and solar cells, even minor impurities can degrade performance, which is why our quality assurance protocols are designed to exceed industry standards.
We invite R&D directors and procurement specialists to collaborate with us to leverage this innovative FeCl3-catalyzed synthesis for their next-generation projects. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis that details how switching to this iron-mediated route can optimize your specific bill of materials. We encourage you to contact us directly to obtain specific COA data for Compound I and to discuss route feasibility assessments tailored to your volume requirements, ensuring a secure and cost-effective supply of this critical electronic material.
