Scalable Synthesis of Soluble D-A-D Organic Photothermal Small Molecules for Advanced Applications
Scalable Synthesis of Soluble D-A-D Organic Photothermal Small Molecules for Advanced Applications
The rapid evolution of photothermal therapy and optoelectronic devices has created an urgent demand for materials that combine high efficiency with processability. Patent CN110950894A introduces a breakthrough D-A-D (Donor-Acceptor-Donor) type organic photo-thermal micromolecule material that addresses the critical limitations of traditional conjugated polymers. By strategically integrating an electron-withdrawing 2,1,3-benzothiadiazole core with electron-donating 3,6-di(thiophen-2-yl)-2,5-dihydropyrrolo[3,4-c]pyrrole-1,4-dione (DPP) units, this technology delivers a material with a definite molecular structure and superior solubility. For R&D directors and procurement specialists in the electronic chemical and biomedical sectors, this patent represents a viable pathway to high-performance materials that eliminate the batch-to-batch variability inherent in polymeric systems.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, organic photothermal materials have relied heavily on conjugated polymer nanoparticles. While these polymers possess extensive pi-conjugation systems capable of absorbing light, they suffer from severe structural rigidity which leads to poor solubility in common organic solvents. This insolubility creates significant downstream processing challenges, often requiring harsh conditions or specialized surfactants to achieve dispersion, which can introduce impurities detrimental to biological applications. Furthermore, the polydisperse nature of polymers means that molecular weight distribution varies between batches, leading to inconsistent optical properties and unpredictable photothermal conversion efficiencies. For supply chain managers, this variability translates into quality control nightmares and potential delays in product validation.
The Novel Approach
The methodology outlined in CN110950894A circumvents these issues by employing a small-molecule design strategy. The introduction of branched alkyl chains, specifically 2-ethylhexyl groups, onto the nitrogen atoms of the DPP donor units acts as a solubilizing handle. This structural feature effectively breaks up strong intermolecular pi-pi stacking interactions without compromising the electronic communication across the D-A-D backbone. The result is a material that maintains the desirable rigid planar structure for light absorption while gaining the fluidity required for solution processing. This approach ensures a monodisperse product with a precise molecular weight, offering R&D teams the reproducibility necessary for rigorous clinical or device testing.
Mechanistic Insights into Suzuki-Miyaura Cross-Coupling and Heterocyclic Condensation
The synthesis of this advanced material relies on two pivotal chemical transformations: the formation of the DPP core and the final assembly via palladium-catalyzed cross-coupling. The initial step involves the condensation of 2-cyanothiophene with diisopropyl succinate in the presence of sodium tert-amylate. This base-mediated reaction facilitates the nucleophilic attack and subsequent cyclization to form the pyrrolo-pyrrole dione scaffold. The choice of tert-amyl alcohol as a solvent is critical here, as it provides the necessary thermal stability for reflux at 90°C while solubilizing the reactive intermediates. Following this, the N-alkylation step utilizes cesium carbonate to deprotonate the lactam nitrogens, allowing for the attachment of the solubilizing bromoisooctane chains.
The culmination of the synthesis is the Suzuki-Miyaura coupling reaction, which links the brominated DPP donor units to the central benzothiadiazole acceptor. This reaction is performed under inert atmosphere using a palladium catalyst, specifically [1,1'-bis(diphenylphosphino)ferrocene]palladium(II) dichloride (PdCl2(dppf)), and cesium carbonate as the base in 1,4-dioxane. The mechanism proceeds through the oxidative addition of the aryl bromide to the Pd(0) species, followed by transmetallation with the boronic acid ester and reductive elimination to form the new carbon-carbon bond. This step is highly sensitive to oxygen and moisture, necessitating the vacuum-nitrogen purge cycles described in the patent to maintain catalyst activity and ensure high yields.
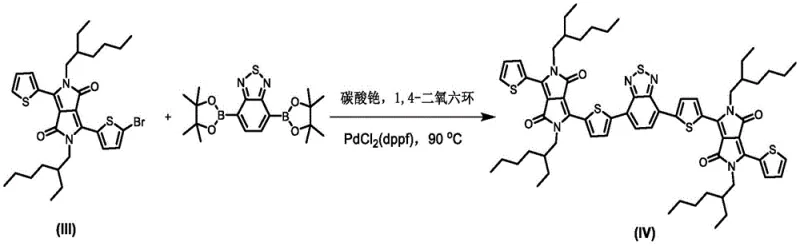
Impurity control in this sequence is managed through careful stoichiometry and purification techniques. In the bromination step, using a slight excess of N-bromosuccinimide (1:1.2 molar ratio) ensures mono-substitution while minimizing dibrominated byproducts. The subsequent column chromatography using dichloromethane and petroleum ether mixtures effectively separates the desired mono-brominated intermediate from unreacted starting material and over-brominated species. This rigorous purification at the intermediate stage prevents the propagation of impurities into the final coupling step, which is essential for achieving the high purity required for electronic and biomedical applications.
How to Synthesize D-A-D Organic Photothermal Material Efficiently
The preparation of this high-value material follows a logical four-step sequence that balances yield with purity. The process begins with the construction of the heterocyclic core, followed by solubility enhancement, functionalization, and final assembly. Each step has been optimized in the patent examples to provide reproducible results, with the initial condensation yielding up to 87% and the final coupling achieving nearly 50% yield on a laboratory scale. The detailed standardized synthesis steps for commercial production are provided in the guide below.
- Condense 2-cyanothiophene with diisopropyl succinate using sodium tert-amylate in tert-amyl alcohol at 90°C to form the DPP core.
- Perform N-alkylation of the DPP core with bromoisooctane using cesium carbonate in acetonitrile to enhance solubility.
- Execute selective monobromination of the thiophene ring using N-bromosuccinimide (NBS) in chloroform at room temperature.
- Complete the D-A-D assembly via Suzuki coupling with 2,1,3-benzothiadiazole bis-boronate ester using a palladium catalyst.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition from polymeric to small-molecule photothermal materials offers distinct logistical and economic benefits. The primary advantage lies in the definable nature of the chemical structure. Unlike polymers, which require complex characterization methods like GPC to determine molecular weight distribution, small molecules can be fully characterized using standard NMR and Mass Spectrometry. This simplifies the incoming quality control (IQC) process, reducing the time and cost associated with raw material verification. Additionally, the improved solubility eliminates the need for expensive and potentially toxic surfactants or high-energy sonication processes often required to disperse insoluble polymers.
- Cost Reduction in Manufacturing: The synthetic route utilizes commercially available starting materials such as 2-cyanothiophene and diisopropyl succinate, which are produced at scale for various industries. The avoidance of exotic monomers reduces raw material costs significantly. Furthermore, the reaction conditions are relatively mild, with most steps occurring at or below 90°C, which lowers energy consumption compared to high-temperature polymerization processes. The ability to purify intermediates via standard column chromatography or recrystallization avoids the need for specialized fractionation equipment.
- Enhanced Supply Chain Reliability: The reliance on robust, well-understood reactions like the Suzuki coupling ensures that the manufacturing process is less prone to failure. The reagents involved, including palladium catalysts and boronic esters, are commodity chemicals with stable global supply chains. This reduces the risk of production stoppages due to raw material shortages. Moreover, the solid-state stability of the intermediates allows for safer storage and transportation, mitigating risks associated with hazardous liquid monomers used in other polymer syntheses.
- Scalability and Environmental Compliance: The stepwise nature of the synthesis allows for easy scale-up from gram to kilogram scales without fundamental changes to the chemistry. The use of solvents like acetonitrile and 1,4-dioxane, while requiring proper handling, allows for efficient recovery and recycling through distillation, aligning with green chemistry principles. The elimination of heavy metal contaminants is streamlined, as the palladium catalyst can be removed using standard scavengers or filtration techniques, ensuring the final product meets stringent regulatory limits for biomedical use.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this D-A-D photothermal material. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy for decision-makers evaluating this technology for integration into their product pipelines.
Q: How does this small molecule overcome the solubility issues of conjugated polymers?
A: Unlike rigid conjugated polymers, this D-A-D small molecule incorporates flexible 2-ethylhexyl alkyl chains on the donor units. This structural modification significantly disrupts molecular stacking, thereby drastically improving solubility in common organic solvents like chloroform and THF, facilitating solution processing.
Q: What is the photostability profile of the synthesized nanoparticles?
A: The patent data indicates that nanoparticles derived from this material exhibit excellent photostability under continuous 808 nm laser irradiation. The defined molecular structure prevents the photodegradation often seen in less stable polymeric systems, ensuring consistent photothermal conversion over multiple cycles.
Q: Is the Suzuki coupling step scalable for industrial production?
A: Yes, the Suzuki-Miyaura coupling utilized in the final step is a robust, widely established industrial reaction. Using standard catalysts like PdCl2(dppf) and mild bases like cesium carbonate in 1,4-dioxane allows for reliable scale-up from gram to kilogram quantities with established purification protocols.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable D-A-D Organic Photothermal Material Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the D-A-D organic photothermal material described in CN110950894A for next-generation biomedical and electronic applications. As a leading CDMO partner, we possess the technical expertise to translate this laboratory-scale protocol into a robust commercial manufacturing process. Our facilities are equipped to handle complex multi-step syntheses involving air-sensitive palladium catalysis and rigorous purification requirements. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our stringent purity specifications and rigorous QC labs guarantee that every batch meets the exacting standards required for high-performance optoelectronic and therapeutic applications.
We invite you to collaborate with us to optimize this synthesis for your specific volume requirements. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your project goals, identifying opportunities to further streamline the process. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a reliable supply of this cutting-edge photothermal material.
