Advanced Synthesis of Asymmetric Diphenyl Sulfone Dianhydride for High-Performance Polyimides
Advanced Synthesis of Asymmetric Diphenyl Sulfone Dianhydride for High-Performance Polyimides
The rapid evolution of the microelectronics and aerospace sectors demands polyimide materials with superior thermal stability and mechanical properties, driving the need for innovative monomer synthesis strategies. Patent CN113754621A introduces a groundbreaking method for synthesizing 2,3',3,4'-diphenyl sulfone tetracarboxylic dianhydride, an asymmetric monomer crucial for disrupting molecular chain regularity and enhancing solubility. Unlike traditional symmetric dianhydrides, this asymmetric structure introduces non-coplanar twists that significantly improve melt stability and processability without sacrificing thermal performance. The disclosed technology leverages a unique thioether oxidation pathway that achieves exceptional selectivity, addressing long-standing challenges in the production of high-performance electronic chemical intermediates. For R&D directors and procurement specialists, this patent represents a pivotal shift towards more efficient, scalable, and cost-effective manufacturing protocols for next-generation polyimides.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of diphenyl sulfone tetracarboxylic dianhydrides has relied heavily on the reaction of 4-chlorobenzoic anhydride with carbon disulfide in the presence of sulfur and basic catalysts. This conventional route, while established, suffers from severe drawbacks when applied to asymmetric targets, primarily generating complex mixtures of three different dianhydride isomers that are notoriously difficult to separate. Furthermore, prior art methods utilizing elemental sulfur often require harsh reaction conditions and result in low yields and poor purity, necessitating extensive downstream purification that drives up operational costs. Another existing approach involves reacting N-substituted chlorophthalimides with sodium hydrosulfide, but this pathway is plagued by expensive raw materials, high energy consumption due to elevated reaction temperatures, and poor selectivity during the critical thioether oxidation step. These inefficiencies create significant bottlenecks for supply chain heads seeking reliable sources of high-purity electronic chemical intermediates.
The Novel Approach
The methodology outlined in CN113754621A fundamentally reengineers the synthetic route by employing dimethyl thiophenol as a key starting material, which serves as a highly reactive precursor for the thioether linkage. This strategic choice enables the formation of a specific thioether intermediate containing two methyl groups and two carboxyl groups, which exhibits markedly higher reaction activity during the subsequent oxidation phase. By utilizing this activated intermediate, the process achieves a thioether oxidation yield exceeding 90%, a substantial improvement over the low selectivity observed in traditional sulfur-based methods. The entire sequence operates under mild conditions, with condensation temperatures capped at 125°C, thereby reducing energy loads and enhancing safety profiles for industrial scale-up. This novel approach not only simplifies the purification landscape by minimizing isomer formation but also aligns perfectly with the rigorous quality standards required for reliable electronic chemical suppliers.
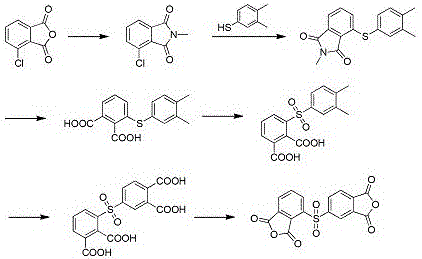
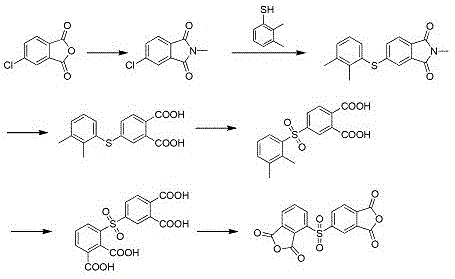
Mechanistic Insights into Selective Thioether Oxidation
The core innovation of this synthesis lies in the precise control of the oxidation states, particularly the conversion of the thioether linkage to a sulfone group using a hydrogen peroxide and sodium tungstate system. Mechanistically, the presence of electron-donating methyl groups on the phenyl ring adjacent to the sulfur atom increases the electron density, thereby facilitating the nucleophilic attack by the peroxotungstate species. This electronic activation allows the oxidation to proceed rapidly and selectively at moderate temperatures between 25-50°C, preventing over-oxidation or degradation of the sensitive phthalimide backbone. The use of sodium tungstate as a phase-transfer catalyst ensures that the oxidant effectively interacts with the organic substrate in the toluene phase, maximizing atom economy and minimizing waste generation. For technical teams, understanding this catalytic cycle is vital, as it demonstrates how subtle structural modifications in the starting material can drastically alter reaction kinetics and thermodynamic favorability.
Following the sulfone formation, the process employs a robust methyl oxidation step using potassium permanganate under reflux conditions in water to convert the methyl groups into carboxylic acids. This step is critical for establishing the tetracarboxylic acid framework necessary for the final cyclization into the dianhydride. The hydrolysis of the N-methyl imide prior to oxidation ensures that the nitrogen functionality is removed cleanly, preventing interference with the oxidation catalysts. The final dehydration step, typically achieved using acetic anhydride, closes the anhydride rings to yield the target 2,3',3,4'-diphenyl sulfone tetracarboxylic dianhydride with purity levels reaching 99.2%. This meticulous control over each mechanistic stage ensures that the final impurity profile meets the stringent specifications demanded by high-end polymer applications.
How to Synthesize 2,3',3,4'-Diphenyl Sulfone Tetracarboxylic Dianhydride Efficiently
The synthesis protocol described in the patent offers a clear, six-step pathway that transforms inexpensive chlorophthalic anhydrides into high-value asymmetric dianhydrides through a series of well-defined chemical transformations. The process begins with an N-alkylation reaction in a mixed solvent system of acetic acid and toluene, followed by a base-catalyzed condensation with dimethyl thiophenol in polar aprotic solvents like DMF or DMAc. Subsequent steps involve alkaline hydrolysis, selective oxidation of the sulfur bridge, oxidation of the methyl substituents, and finally, thermal or chemical dehydration. Each stage is optimized for yield and purity, with specific attention paid to temperature control and stoichiometric ratios to prevent side reactions. The detailed standardized synthesis steps below provide a comprehensive guide for replicating this high-efficiency route in a pilot or commercial setting.
- Perform N-alkylation of chlorophthalic anhydride with methylamine in acetic acid and toluene under reflux to form N-methyl-chlorophthalimide.
- Conduct condensation between N-methyl-chlorophthalimide and dimethyl thiophenol using a basic catalyst (e.g., K2CO3/KF) in DMF at 110-125°C.
- Execute sequential hydrolysis, thioether oxidation using hydrogen peroxide/sodium tungstate, and methyl oxidation with potassium permanganate, followed by final dehydration.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthesis route offers profound advantages for procurement managers and supply chain leaders focused on cost reduction in electronic chemical manufacturing. By replacing expensive and hazardous reagents like elemental sulfur or sodium hydrosulfide with stable, commodity-grade dimethyl thiophenol, the process significantly lowers the raw material cost baseline. The mild reaction conditions, particularly the condensation step operating below 125°C, reduce the energy intensity of the production cycle, leading to substantial operational expenditure savings over time. Furthermore, the high selectivity of the oxidation steps minimizes the formation of by-products, which simplifies downstream purification and reduces the consumption of solvents and adsorbents typically required for chromatography or recrystallization. These factors collectively contribute to a more resilient and economically viable supply chain for complex polymer additives and monomers.
- Cost Reduction in Manufacturing: The elimination of complex isomer separation processes, which are common in traditional sulfur-based syntheses, drastically reduces processing time and resource allocation. By achieving high yields in the critical oxidation steps, the overall material throughput is maximized, ensuring that less feedstock is wasted as off-spec material. The use of common inorganic oxidants like hydrogen peroxide and potassium permanganate further stabilizes the cost structure, shielding the production process from volatility in specialty reagent markets. Consequently, manufacturers can achieve a lower cost of goods sold (COGS) while maintaining competitive pricing for their high-purity OLED material or polyimide precursors.
- Enhanced Supply Chain Reliability: The reliance on widely available starting materials such as chlorophthalic anhydride and dimethyl thiophenol mitigates the risk of supply disruptions associated with niche or regulated chemicals. The robustness of the reaction conditions allows for flexible scheduling and easier scale-up, enabling suppliers to respond rapidly to fluctuating market demands without compromising product quality. Additionally, the reduced hazard profile of the reagents simplifies logistics and storage requirements, facilitating smoother transportation and inventory management. This reliability is essential for maintaining continuous production lines in the fast-paced semiconductor and display industries.
- Scalability and Environmental Compliance: The process is inherently designed for commercial scale-up, with reaction parameters that translate effectively from laboratory glassware to large-scale stainless steel reactors. The aqueous workup procedures and the ability to recover solvents like toluene and DMF support sustainable manufacturing practices and reduce the environmental footprint of the facility. By avoiding heavy metal catalysts and minimizing toxic waste streams, the method aligns with increasingly stringent global environmental regulations, ensuring long-term operational compliance. This scalability ensures that the commercial scale-up of complex polymer additives can proceed without encountering the typical bottlenecks of process intensification.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of this asymmetric dianhydride, based on the specific advantages detailed in the patent literature. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this monomer into their existing polymer formulations. The answers reflect the practical realities of implementing this chemistry in an industrial environment, focusing on yield optimization and purity control.
Q: Why is dimethyl thiophenol preferred over elemental sulfur in this synthesis?
A: Using dimethyl thiophenol allows for the formation of a thioether intermediate with higher reaction activity during oxidation. This significantly improves selectivity and yields over 90%, avoiding the complex isomer mixtures generated by elemental sulfur methods.
Q: What are the critical temperature controls for the oxidation steps?
A: The thioether oxidation must be maintained between 25-50°C (optimally 32-38°C) to ensure safety and selectivity, while the subsequent methyl oxidation requires reflux conditions in water with potassium permanganate.
Q: How does this method improve supply chain reliability for polyimide manufacturers?
A: The process utilizes cheap, readily available raw materials like chlorophthalic anhydride and operates under mild conditions (≤125°C), reducing energy consumption and equipment stress, which ensures consistent, scalable production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,3',3,4'-Diphenyl Sulfone Tetracarboxylic Dianhydride Supplier
As the demand for advanced polyimide materials continues to surge in the aerospace and microelectronics sectors, securing a dependable source of high-quality monomers is paramount for maintaining product performance and market competitiveness. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to deliver consistent supply. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced analytical instrumentation to ensure that every batch meets stringent purity specifications, including the precise isomer ratios required for optimal polymer properties. We understand that the transition from lab-scale discovery to full-scale manufacturing requires a partner who can navigate complex regulatory landscapes and technical challenges with precision.
We invite R&D directors and procurement professionals to collaborate with us to explore how this innovative synthesis route can enhance your material portfolio and drive down production costs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and application needs. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to excellence can support your long-term strategic goals in the field of high-performance electronic materials.
