Advanced Synthesis of 13C-Mexaxetine: A Breakthrough in High-Purity Diagnostic Reagent Manufacturing
Introduction to Next-Generation 13C-Labeling Technology
The landscape of diagnostic reagent manufacturing is undergoing a significant transformation driven by the demand for higher isotopic purity and more efficient synthetic routes. Patent CN115073252B introduces a groundbreaking methodology for the preparation of 13C-Mexaxetine and 13C-Phenacetin, two critical substrates used in non-invasive liver function detection and breath tests. Unlike traditional approaches that struggle with purification bottlenecks, this invention leverages a modified Mitsunobu reaction strategy utilizing Boc-protected precursors. By shifting the labeling step to an earlier stage of the synthesis where the molecular polarity is more favorable for separation, the process overcomes the historical challenges associated with triphenylphosphine oxide removal. This technical advancement not only ensures product purity exceeding 99% but also establishes a robust framework for the reliable supply of high-performance diagnostic intermediates.
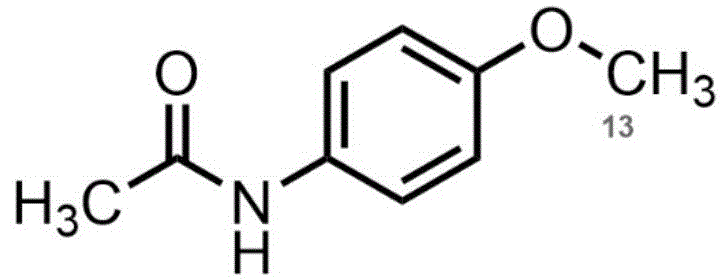
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 13C-labeled acetaminophen derivatives relied on the direct alkylation of acetaminophen using 13C-methyl iodide or similar activated species. While conceptually straightforward, this conventional pathway presents severe downstream processing challenges that hinder commercial viability. The primary obstacle lies in the physicochemical similarity between the desired 13C-Mexaxetine product and the stoichiometric byproduct, triphenylphosphine oxide, generated during the coupling reaction. Both molecules exhibit comparable polarity and hydrogen-bonding capabilities, rendering standard extraction and crystallization techniques ineffective. Consequently, manufacturers are often forced to rely on repetitive and costly chromatographic separations, which drastically reduce overall yield and increase the cost of goods sold. Furthermore, the harsh conditions sometimes required to drive the direct reaction can lead to isotopic scrambling or degradation, compromising the stringent abundance requirements necessary for clinical breath testing applications.
The Novel Approach
The innovative strategy disclosed in the patent fundamentally re-engineers the synthetic timeline by introducing a protecting group strategy that decouples the labeling event from the final pharmacophore assembly. By employing tert-butoxycarbonyl (Boc) protected 4-hydroxyaniline as the starting nucleophile, the resulting ether intermediate possesses distinct solubility characteristics compared to the phosphine oxide waste. This polarity divergence allows for the efficient removal of impurities via standard column chromatography or extraction prior to the final deprotection steps. The process utilizes a photo-delayed reaction system involving diisopropyl azodicarboxylate (DIAD) and triphenylphosphine to activate the 13C-alcohol, ensuring high regioselectivity for the phenolic oxygen. This approach not only simplifies the purification workflow but also protects the amine functionality from unwanted side reactions during the harsh alkylation conditions, thereby preserving the integrity of the nitrogen center for subsequent acetylation.
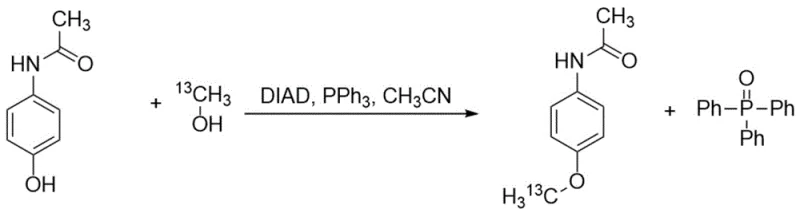
Mechanistic Insights into the Modified Mitsunobu Alkylation
The core of this synthetic breakthrough relies on the precise orchestration of the Mitsunobu reaction mechanism, which facilitates the formation of the carbon-oxygen bond between the phenolic ring and the 13C-labeled methyl group. In this catalytic cycle, triphenylphosphine acts as a potent oxygen acceptor, attacking the azo compound (DIAD) to form a highly reactive phosphonium intermediate. This species subsequently activates the hydroxyl group of the 13C-methanol, converting it into an excellent leaving group. Simultaneously, the phenolic oxygen of the Boc-protected aniline is deprotonated, generating a nucleophilic phenoxide anion. The subsequent SN2-type attack by the phenoxide on the activated 13C-methyl species results in the inversion of configuration (though irrelevant for a methyl group) and the formation of the ether linkage. Crucially, the reaction conditions are maintained at 0°C during the addition of reagents to suppress competing side reactions and control the exotherm, ensuring that the delicate 13C-label remains intact and exclusively positioned on the methoxy group.
From an impurity control perspective, the mechanism offers inherent advantages that translate directly to process robustness. The formation of triphenylphosphine oxide is inevitable in Mitsunobu chemistry; however, the presence of the bulky, lipophilic Boc group on the aniline nitrogen significantly alters the partition coefficient of the organic product. While the phosphine oxide byproduct remains highly polar and prone to retaining in aqueous washes or specific silica phases, the Boc-protected intermediate exhibits enhanced organic solubility. This differential behavior is exploited during the workup phase, where sequential washing with saturated sodium carbonate and brine effectively strips away acidic and polar impurities. The result is a crude intermediate that is already of high purity before any chromatographic polishing, minimizing the load on purification columns and reducing solvent consumption. This mechanistic understanding allows for tighter control over the impurity profile, ensuring that the final API intermediate meets the rigorous specifications required for in vivo diagnostic use.
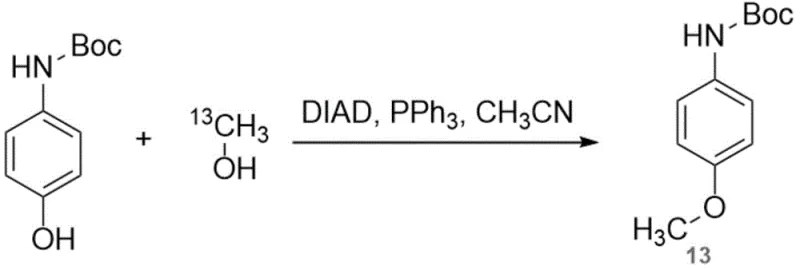
How to Synthesize 13C-Mexaxetine Efficiently
The operational protocol for this synthesis is designed to balance high yield with ease of execution, making it suitable for both laboratory scale-up and industrial production. The process begins with the dissolution of the Boc-protected precursor and 13C-methanol in an anhydrous solvent such as acetonitrile, followed by the controlled addition of the phosphine and azo reagents. Following the coupling reaction, the mixture undergoes a specialized quenching and extraction sequence to isolate the protected intermediate. The critical transformation occurs in the final stages, where the protecting group is removed, and the amine is acetylated to restore the acetaminophen scaffold. This two-step finishing sequence—deprotection followed by acylation—is highly efficient and avoids the need for isolating the unstable free amine, thereby maximizing overall throughput. For a comprehensive breakdown of the specific molar ratios, temperature profiles, and workup procedures, please refer to the standardized guide below.
- Perform a Mitsunobu reaction between tert-butoxycarbonyl-4-hydroxyaniline and 13C-methanol using triphenylphosphine and DIAD in acetonitrile at 0°C.
- Quench the reaction with saturated sodium carbonate, extract with ethyl acetate, and purify the intermediate via column chromatography.
- Remove the Boc protecting group with trifluoroacetic acid, followed by acetylation with acetyl chloride and triethylamine to yield the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers tangible strategic benefits that extend beyond mere technical elegance. The primary value proposition lies in the drastic simplification of the purification train. By eliminating the need for complex, multi-pass chromatography to separate the final product from phosphine oxides, the process significantly reduces solvent usage and processing time. This streamlining translates directly into lower manufacturing costs and a reduced environmental footprint, aligning with modern green chemistry initiatives. Furthermore, the reliance on commodity chemicals like acetyl chloride, triethylamine, and standard protecting group reagents ensures that the supply chain is resilient against raw material shortages. The robustness of the intermediate isolation step means that production batches are less likely to fail quality control due to trace impurities, thereby enhancing supply continuity and reliability for downstream diagnostic kit manufacturers.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the elimination of expensive and time-consuming purification steps associated with conventional methods. By shifting the difficulty of separation to an intermediate stage where polarity differences are maximized, the process avoids the yield losses typically incurred during the final purification of sensitive labeled compounds. The ability to use standard crystallization techniques for the final product, rather than preparative HPLC, represents a massive reduction in operational expenditure. Additionally, the high atom economy of the Mitsunobu reaction, combined with the recovery potential of solvents like acetonitrile and ethyl acetate, further drives down the cost per gram of the active ingredient, making high-purity 13C-diagnostic reagents more accessible for widespread clinical screening programs.
- Enhanced Supply Chain Reliability: Supply security is paramount in the pharmaceutical intermediate sector, and this synthesis route bolsters reliability through the use of stable, commercially available starting materials. The Boc-protected aniline precursor is a stable solid that can be sourced from multiple global suppliers, mitigating the risk of single-source dependency. Moreover, the reaction conditions are mild and do not require exotic catalysts or extreme pressures, allowing for production in standard stainless steel reactors found in most fine chemical facilities. This flexibility means that manufacturing can be easily distributed across different geographic locations to mitigate logistical risks. The high yield and reproducibility of the reaction ensure that production schedules can be met consistently, reducing the lead time for delivering critical diagnostic substrates to healthcare providers.
- Scalability and Environmental Compliance: Scaling isotopic labeling processes often presents unique challenges due to the high cost of the labeled starting materials; however, this method optimizes the incorporation efficiency of the expensive 13C-methanol. The high conversion rates ensure minimal waste of the valuable isotope, which is a significant cost driver. From an environmental standpoint, the process generates waste streams that are well-characterized and manageable, primarily consisting of phosphine oxides and organic salts that can be treated using standard effluent protocols. The reduction in solvent volume required for purification, coupled with the potential for solvent recycling, supports compliance with increasingly stringent environmental regulations. This scalability ensures that the technology can grow from kilogram-scale R&D batches to multi-ton commercial production without requiring fundamental process re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on performance metrics and operational feasibility. Understanding these nuances is essential for technical teams evaluating the integration of this route into their existing manufacturing portfolios.
Q: Why is the Boc-protected route superior to direct acetaminophen labeling?
A: Direct labeling of acetaminophen results in a product with polarity similar to the triphenylphosphine oxide byproduct, making separation extremely difficult. The Boc-protected intermediate has significantly different polarity, allowing for easy removal of impurities before the final deprotection step.
Q: What isotopic abundance can be achieved with this method?
A: The patented process consistently achieves a 13C isotopic abundance greater than 99% at the labeling position, which is critical for the sensitivity and accuracy of liver function breath tests.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the method replaces difficult chromatographic separations of the final product with a crystallization step. The use of standard reagents like acetyl chloride and TFA, along with robust intermediate purification, makes the process highly scalable for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 13C-Mexaxetine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent to production requires a partner with deep technical expertise and a commitment to quality. Our team specializes in the commercialization of complex isotopically labeled compounds, leveraging advanced process chemistry to bridge the gap between laboratory innovation and industrial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying isotopic abundance and chemical purity to the highest international standards, guaranteeing that every batch of 13C-Mexaxetine or 13C-Phenacetin performs flawlessly in clinical diagnostic applications.
We invite you to collaborate with us to optimize your supply chain for liver function detection reagents. Our technical sales team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how our streamlined synthesis route can reduce your total landed cost. We encourage you to contact our technical procurement team today to request specific COA data, route feasibility assessments, and sample availability. Let us help you secure a reliable, high-quality supply of these critical diagnostic intermediates, ensuring the success of your next-generation medical testing kits.
