Advanced Palladium-Catalyzed Synthesis of 2-Trifluoromethylindenylmethylene Indole Derivatives for Commercial Scale-Up
Advanced Palladium-Catalyzed Synthesis of 2-Trifluoromethylindenylmethylene Indole Derivatives for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking efficient routes to complex heterocyclic scaffolds that possess high biological activity. Patent CN109320446B introduces a groundbreaking synthetic methodology for producing 2-trifluoromethylindenylmethylene indole derivatives, a class of compounds that merges the pharmacophoric properties of indenes and indoles with the metabolic stability conferred by trifluoromethyl groups. This innovation addresses a significant gap in organic synthesis, as prior art lacked direct methods to construct this specific polycyclic architecture in a single operational step. The disclosed technology leverages a palladium-catalyzed tandem cyclization strategy that operates under relatively mild thermal conditions, utilizing readily available starting materials to generate high-value intermediates. For R&D directors and process chemists, this represents a pivotal advancement in accessing novel chemical space for drug discovery programs focused on sulindac analogs or ellipticine derivatives. The robustness of this catalytic system suggests immediate applicability in the manufacturing of high-purity pharmaceutical intermediates where structural complexity often dictates cost and timeline.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the construction of fused indene-indole systems has been a formidable challenge requiring multi-step sequences that suffer from poor atom economy and low overall yields. Conventional approaches often involve the separate synthesis of the indene core followed by coupling with an indole moiety, necessitating harsh reaction conditions and the use of stoichiometric amounts of activating agents. These stepwise protocols frequently result in the accumulation of impurities that are difficult to remove, thereby complicating downstream purification and increasing the cost of goods sold. Furthermore, the introduction of fluorine atoms into such rigid frameworks typically requires specialized fluorinating reagents that are expensive, hazardous, and difficult to handle on a large scale. The cumulative effect of these inefficiencies is a prolonged development timeline and reduced supply chain reliability for critical API intermediates. Consequently, there has been a persistent demand for a more convergent strategy that can assemble these complex motifs with greater precision and operational simplicity.
The Novel Approach
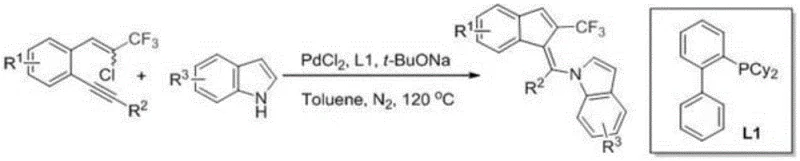
The methodology described in the patent data revolutionizes this landscape by employing a palladium-catalyzed cascade reaction that constructs the entire carbon skeleton in a single pot. By utilizing 1-(2-chloro-3,3,3-trifluoroprop-1-en-1-yl)-2-(phenylethynyl)benzene as a multifunctional substrate, the process enables simultaneous ring closure and C-N bond formation without the need for intermediate isolation. This tandem cyclization is driven by a carefully optimized catalytic system comprising PdCl2 and a bulky phosphine ligand, which facilitates the activation of the chloro-alkene moiety and subsequent intramolecular insertion. The use of sodium tert-butoxide as a base ensures efficient deprotonation and catalyst turnover, allowing the reaction to proceed smoothly at 120°C in toluene. This novel approach not only streamlines the synthetic route but also significantly enhances the safety profile by avoiding the handling of reactive intermediates. For procurement managers, this translates to a drastic reduction in raw material costs and waste disposal expenses, making the production of these specialized intermediates economically viable.
Mechanistic Insights into Pd-Catalyzed Tandem Cyclization
A deep understanding of the catalytic cycle is essential for optimizing this process for commercial manufacturing and ensuring consistent product quality. The reaction initiates with the in situ generation of active Pd(0) species from the Pd(II) precursor under the influence of the phosphine ligand, which then undergoes oxidative addition with the chloro-alkene substrate to form a key organopalladium intermediate. This intermediate subsequently engages in an intramolecular carbopalladation across the alkyne triple bond, effectively closing the five-membered indene ring and establishing the core steric framework. Following this cyclization event, the resulting vinyl-palladium species undergoes a ligand exchange with the indole nitrogen, facilitated by the base, to form a stable palladium-amido complex. The final step involves reductive elimination, which releases the target 2-trifluoromethylindenylmethylene indole product and regenerates the Pd(0) catalyst to enter the next turnover cycle. This elegant mechanism minimizes side reactions such as homocoupling or polymerization, which are common pitfalls in palladium chemistry, thereby ensuring high selectivity for the desired Z-isomer.
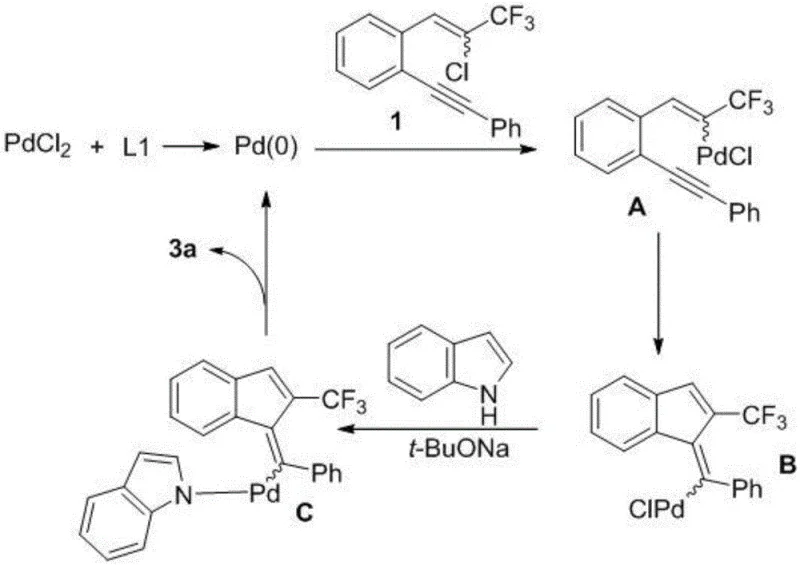
Controlling the impurity profile is paramount when synthesizing pharmaceutical intermediates, and this mechanistic pathway offers inherent advantages in that regard. The concerted nature of the tandem cyclization limits the lifetime of reactive intermediates, reducing the opportunity for decomposition or rearrangement into structurally related byproducts. The specific choice of the 2-dicyclohexylphosphino-biphenyl ligand creates a steric environment that favors the formation of the Z-isomer over the E-isomer, as evidenced by the high Z/E ratios observed in the experimental data. Additionally, the use of a non-polar solvent like toluene helps to solubilize the organic substrates while keeping inorganic salts precipitated, which simplifies the workup procedure and minimizes metal contamination in the final product. For quality control teams, this means that the crude product often requires minimal purification, typically achievable through standard flash column chromatography. The robustness of the catalytic cycle against variations in substrate electronics further ensures that a wide range of derivatives can be produced with consistent purity specifications, meeting the rigorous standards required for clinical supply.
How to Synthesize 2-Trifluoromethylindenylmethylene Indole Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific operational parameters to maximize yield and safety. The standard protocol involves charging a reaction vessel with the chloro-alkyne substrate, the indole coupling partner, palladium chloride, and the phosphine ligand under an inert nitrogen atmosphere to prevent catalyst oxidation. Sodium tert-butoxide is then added as a solid or solution, and the mixture is heated to reflux in toluene, maintaining a temperature of 120°C for approximately 24 hours to ensure complete conversion. Monitoring the reaction progress via TLC or HPLC is recommended to determine the optimal endpoint before proceeding to the aqueous workup. Detailed standardized operating procedures for scaling this reaction from gram to kilogram quantities are provided in the technical guide below.
- Combine 1-(2-chloro-3,3,3-trifluoroprop-1-en-1-yl)-2-(phenylethynyl)benzene and indole substrates with PdCl2 catalyst and phosphine ligand in toluene.
- Add sodium tert-butoxide as the base and heat the mixture to 120°C under a nitrogen atmosphere for 24 hours to facilitate tandem cyclization.
- Upon completion, perform aqueous workup with ethyl acetate and brine, dry over sodium sulfate, and purify via flash column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented technology offers substantial benefits that directly impact the bottom line and supply chain resilience for chemical manufacturers. The elimination of multiple synthetic steps and intermediate isolation procedures results in a significantly simplified process flow, which reduces labor costs and facility occupancy time. By consolidating the synthesis into a single reactor operation, companies can achieve higher throughput and better utilization of existing manufacturing assets without the need for capital-intensive equipment upgrades. The use of commodity chemicals such as toluene and sodium tert-butoxide ensures that raw material sourcing remains stable and cost-effective, mitigating the risks associated with supply chain disruptions of exotic reagents. Furthermore, the high atom economy of the tandem cyclization minimizes the generation of chemical waste, aligning with increasingly stringent environmental regulations and reducing disposal costs. These factors collectively contribute to a more sustainable and economically competitive manufacturing model for high-value fine chemicals.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the removal of unit operations associated with intermediate purification and solvent exchanges. Traditional multi-step syntheses often incur significant losses in yield at each stage, whereas this one-pot method preserves mass efficiency throughout the transformation. The ability to use a relatively low loading of palladium catalyst combined with a recyclable ligand system further lowers the direct material costs per kilogram of product. Additionally, the reduced energy consumption resulting from shorter overall processing times contributes to lower utility bills and a smaller carbon footprint. These cumulative savings allow suppliers to offer more competitive pricing structures to their pharmaceutical clients while maintaining healthy profit margins.
- Enhanced Supply Chain Reliability: Dependence on complex, multi-vendor supply chains for specialized intermediates introduces significant risk, which this streamlined methodology effectively mitigates. Since the starting materials are structurally simple and widely available from bulk chemical suppliers, the risk of raw material shortages is substantially diminished. The robustness of the reaction conditions means that production can be easily transferred between different manufacturing sites without extensive re-validation, ensuring business continuity. Moreover, the short lead time from raw material intake to finished product allows for a more agile response to fluctuating market demands. This reliability is crucial for pharmaceutical partners who require guaranteed supply continuity to support their own clinical trials and commercial launches.
- Scalability and Environmental Compliance: Scaling chemical processes often reveals hidden bottlenecks, but the exothermic profile and pressure requirements of this reaction are well-suited for large-scale batch reactors. The absence of high-pressure hydrogenation or cryogenic steps simplifies the engineering controls needed for safe operation at the 100 MT scale. From an environmental standpoint, the reduction in solvent volume and the avoidance of hazardous fluorinating agents make this process inherently greener than alternative routes. Waste streams are primarily composed of benign inorganic salts and recoverable organic solvents, facilitating easier treatment and compliance with local discharge regulations. This alignment with green chemistry principles not only reduces regulatory burden but also enhances the corporate sustainability profile of the manufacturing entity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and mechanistic understanding presented in the patent documentation to provide clarity for potential partners. Understanding these nuances is critical for evaluating the feasibility of integrating this route into your existing production portfolio. We encourage stakeholders to review these points carefully to assess the alignment with their specific project requirements.
Q: What are the key advantages of this Pd-catalyzed method over traditional synthesis?
A: This method utilizes a one-pot tandem cyclization strategy that eliminates the need for isolating unstable intermediates, significantly reducing solvent consumption and processing time while achieving yields up to 76%.
Q: Is this process scalable for industrial production of pharmaceutical intermediates?
A: Yes, the reaction employs standard conditions (120°C, atmospheric pressure) and commercially available reagents like PdCl2 and toluene, making it highly suitable for commercial scale-up without requiring specialized high-pressure equipment.
Q: How does the trifluoromethyl group impact the biological potential of these derivatives?
A: The introduction of the trifluoromethyl group enhances metabolic stability and lipophilicity, which are critical parameters for drug candidates targeting various therapeutic areas including oncology and inflammation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethylindenylmethylene Indole Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this palladium-catalyzed technology in accelerating the development of next-generation therapeutics. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to market is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 2-trifluoromethylindenylmethylene indole derivatives meets the highest industry standards. We are committed to leveraging our technical expertise to optimize this specific catalytic system for your unique needs, delivering high-quality intermediates that drive your drug discovery programs forward.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can benefit your specific project goals. By requesting a Customized Cost-Saving Analysis, you can gain detailed insights into the economic advantages of adopting this method for your supply chain. We are ready to provide specific COA data and comprehensive route feasibility assessments to support your decision-making process. Contact us today to explore a strategic partnership that combines cutting-edge chemistry with reliable commercial execution.
