Revolutionizing Polyimide Monomer Production: Advanced Synthesis of 4,4'-Oxydiphthalic Anhydride
Revolutionizing Polyimide Monomer Production: Advanced Synthesis of 4,4'-Oxydiphthalic Anhydride
The global demand for high-performance polyimide materials continues to surge, driven by applications in flexible electronics, aerospace composites, and advanced insulation systems. At the heart of these materials lies the critical monomer 4,4'-oxydiphthalic anhydride (ODPA), which imparts essential flexibility and thermal stability to the polymer chain. A groundbreaking patent, CN111187240A, disclosed on May 22, 2020, introduces a transformative preparation method that addresses long-standing inefficiencies in ODPA manufacturing. This novel approach leverages a direct dehydration coupling reaction to achieve superior yields and purity levels, marking a significant leap forward for the reliable ODPA supplier market. By streamlining the synthetic route, this technology not only enhances product quality but also aligns with modern green chemistry principles, offering a robust solution for the commercial scale-up of complex electronic chemicals.
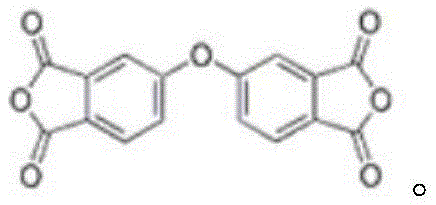
The structural integrity of ODPA is paramount for its performance in polyimide applications. As illustrated in the molecular diagram, the central ether linkage serves as a flexible pivot point within the rigid aromatic backbone. This unique architecture reduces intermolecular forces and lowers the glass transition temperature, thereby improving the processability of the resulting polyimide without compromising its thermal resistance. The ability to synthesize this specific architecture with high precision is crucial for manufacturers aiming to produce next-generation display substrates and flexible circuit boards. The patent highlights that controlling the formation of this ether bond is the key to unlocking higher purity grades, which are increasingly demanded by the semiconductor and optoelectronic industries for high-purity electronic chemical applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of 4,4'-oxydiphthalic anhydride has been plagued by convoluted synthetic pathways that hinder efficiency and increase costs. Traditional methods often involve the condensation of 3,4-dimethoxyphenol with 4-bromo-o-xylene followed by harsh oxidation steps using potassium permanganate in pyridine media. Alternatively, other routes rely on the nitration of methylated phthalic anhydride, followed by bimolecular condensation and subsequent hydrolysis. These legacy processes suffer from severe drawbacks, including low reaction selectivity and the generation of substantial quantities of hazardous byproducts. The presence of these impurities necessitates rigorous and costly purification steps, often involving multiple recrystallizations that further erode the overall yield. Furthermore, the use of strong oxidants and nitrating agents poses significant safety risks and environmental challenges, complicating waste treatment and regulatory compliance for cost reduction in polyimide manufacturing.
The Novel Approach
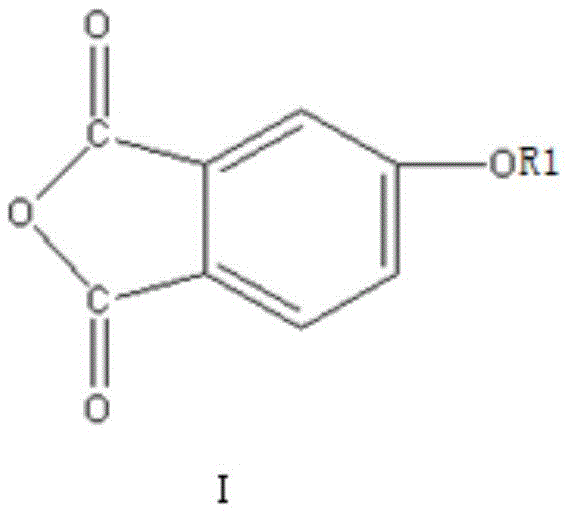
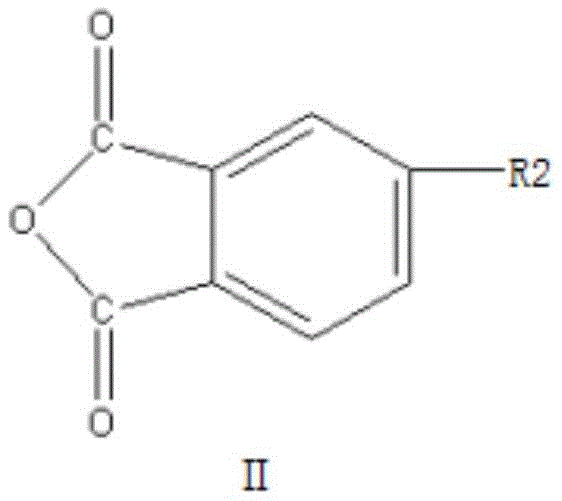
In stark contrast to these cumbersome legacy techniques, the method described in patent CN111187240A employs a streamlined one-step dehydration coupling reaction. This innovative strategy directly couples an alkali metal salt of 4-hydroxyphthalic anhydride (Compound I) with a 4-halogenated phthalic anhydride (Compound II). By bypassing the need for intermediate oxidation or nitration stages, the process drastically simplifies the operational workflow. The reaction proceeds efficiently in a mixture of polar aprotic solvents and water-carrying agents at temperatures ranging from 110°C to 155°C. This direct approach not only minimizes the formation of side products but also ensures a reaction yield exceeding 97%. The simplicity of the route allows for easier separation of the product, where the solvent and water-carrying agent can be recovered and reused, significantly lowering the environmental footprint and operational expenditure associated with reducing lead time for high-purity monomers.
Mechanistic Insights into Dehydration Coupling Reaction
The core of this technological advancement lies in the nucleophilic aromatic substitution mechanism facilitated by the specific reactivity of the reactants. The reaction initiates with the nucleophilic attack of the phenoxide oxygen from Compound I on the electron-deficient aromatic ring of Compound II. The presence of the anhydride moiety in Compound II acts as a powerful electron-withdrawing group, activating the position ortho or para to the carbonyl groups for nucleophilic attack, particularly when a good leaving group such as chlorine, fluorine, bromine, or iodine is present at the 4-position. This electronic activation allows the coupling to occur under relatively mild thermal conditions compared to traditional Ullmann-type couplings which often require copper catalysts and higher temperatures. The use of alkali metals like sodium or potassium in Compound I ensures the formation of a highly reactive phenoxide species, driving the equilibrium towards the formation of the diphenyl ether bond with high specificity.
Controlling impurity profiles in this reaction is achieved through precise management of solvent water content and cooling kinetics. The patent specifies that the water content in the solvent must be maintained below 500ppm. This stringent control is critical because excess water can hydrolyze the anhydride rings prematurely or interfere with the dehydration equilibrium, leading to open-ring acid impurities that are difficult to remove. Furthermore, the cooling crystallization process is optimized with a controlled rate of 6-8°C/h. This slow cooling promotes the growth of large, well-defined crystals while excluding impurities from the crystal lattice. A hot filtration step at 80-100°C prior to final cooling effectively removes insoluble byproducts or unreacted starting materials, ensuring that the final precipitate is of exceptional purity. This mechanistic understanding allows for the consistent production of ODPA with purity levels reaching 99.99%, meeting the rigorous standards required for advanced material applications.
How to Synthesize 4,4'-Oxydiphthalic Anhydride Efficiently
The implementation of this synthesis route requires careful attention to reagent stoichiometry and thermal management to maximize efficiency. The process begins by mixing Compound I and Compound II in a molar ratio of approximately 1:1 to 1:1.2, along with a suitable solvent system comprising a polar aprotic solvent and a water-carrying agent like toluene or xylene. The reaction mixture is heated to initiate the dehydration coupling, typically maintained between 110°C and 155°C for a duration of 6 to 10 hours. Following the reaction, the mixture undergoes a staged cooling process to induce crystallization.
- Mix Compound I (alkali metal salt of 4-hydroxyphthalic anhydride) and Compound II (4-halogenated phthalic anhydride) with a polar aprotic solvent and a water-carrying agent.
- Conduct a dehydration coupling reaction at temperatures between 110°C and 155°C for 6 to 10 hours to form the ether linkage.
- Cool the reaction mixture gradually to crystallize the product, filter to remove impurities, and refine via recrystallization to achieve >99.9% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis method presents compelling economic and logistical benefits. The shift from multi-step oxidative processes to a direct coupling reaction fundamentally alters the cost structure of ODPA production. By eliminating the need for expensive oxidizing agents like potassium permanganate and complex nitration reagents, the raw material costs are significantly reduced. Additionally, the ability to recycle solvents and water-carrying agents after the reaction creates a closed-loop system that minimizes waste disposal costs and reduces the consumption of fresh solvents. This efficiency translates into a more stable pricing model for buyers, shielding them from the volatility often associated with the supply of specialty chemical intermediates.
- Cost Reduction in Manufacturing: The streamlined one-step process eliminates several unit operations found in traditional methods, such as separate oxidation, hydrolysis, and acidification stages. This reduction in processing steps directly lowers energy consumption and labor requirements. Furthermore, the high reaction selectivity means that less raw material is wasted on byproduct formation, maximizing the atom economy of the process. The absence of transition metal catalysts also removes the need for costly metal scavenging steps, which are often required to meet ppm-level metal specifications in electronic grade materials. These factors combine to deliver substantial cost savings without compromising on the quality of the final product.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, specifically 4-halogenated phthalic anhydrides and hydroxyphthalic anhydride salts, are commodity chemicals with robust global supply chains. Unlike specialized reagents that may face sourcing bottlenecks, these precursors are readily available from multiple vendors. The simplicity of the reaction conditions also means that the process can be easily scaled up in standard stainless steel reactors without requiring exotic equipment or extreme pressure conditions. This ease of scale-up ensures that suppliers can rapidly ramp up production to meet surges in demand, providing buyers with greater confidence in supply continuity and shorter lead times for order fulfillment.
- Scalability and Environmental Compliance: From an environmental perspective, this method offers a cleaner alternative to legacy processes. The reduction in hazardous byproducts and the elimination of heavy metal catalysts simplify wastewater treatment and reduce the burden on effluent processing facilities. The ability to recover and reuse organic solvents further diminishes the release of volatile organic compounds (VOCs) into the atmosphere. For manufacturers operating under strict environmental regulations, adopting this greener synthesis route facilitates compliance and reduces the risk of regulatory penalties. The process is inherently safer due to the avoidance of exothermic oxidation reactions, contributing to a safer working environment and lower insurance premiums for production facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 4,4'-oxydiphthalic anhydride synthesized via this advanced method. These insights are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy and relevance for industry professionals.
Q: What are the primary advantages of this new ODPA synthesis method over traditional routes?
A: The patented method utilizes a direct one-step dehydration coupling reaction, eliminating the complex multi-step oxidation and nitration processes found in conventional synthesis. This significantly reduces byproduct formation, simplifies purification, and improves overall reaction yield to over 97%.
Q: How does the choice of solvent impact the purity of the final 4,4'-oxydiphthalic anhydride crystal?
A: The process requires polar aprotic solvents like DMF or DMSO with strictly controlled water content (less than 500ppm). Low water content ensures orderly crystallization during the cooling phase, preventing oiling out and facilitating the removal of solid impurities through hot filtration.
Q: Is this synthesis method scalable for industrial production of electronic grade monomers?
A: Yes, the method is designed for scalability. It uses readily available raw materials, operates at moderate temperatures (110-155°C), and allows for the recycling of solvents and water-carrying agents, making it economically viable for large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4,4'-Oxydiphthalic Anhydride Supplier
As the demand for high-performance polyimides grows, securing a stable source of high-quality monomers is essential for maintaining competitive advantage. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging advanced synthetic methodologies like the one described in CN111187240A to deliver superior products. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements regardless of project stage. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of ODPA meets the exacting standards required for electronic and aerospace applications.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs. Our technical experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific production needs. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us partner with you to drive innovation and efficiency in your polyimide manufacturing processes.
