Scalable Production of High-Purity Thymopentapeptide via Optimized Solid-Phase Synthesis
Scalable Production of High-Purity Thymopentapeptide via Optimized Solid-Phase Synthesis
The pharmaceutical industry continuously seeks robust manufacturing pathways for immunomodulatory agents, and the preparation method disclosed in patent CN1534042A represents a significant technological leap in the synthesis of Thymopentapeptide (TP-5). This specific intellectual property outlines a refined solid-phase peptide synthesis (SPPS) protocol that strategically replaces hazardous reagents with safer, more efficient alternatives, directly addressing the critical bottlenecks of traditional manufacturing. By utilizing 4-hydroxymethyl phenoxymethyl polystyrene resin, commonly known as Wang resin, in conjunction with fluorenylmethyloxycarbonyl (Fmoc) protected amino acids, the process achieves a simultaneous cleavage of the peptide from the solid support and removal of side-chain protecting groups. This innovation not only streamlines the operational workflow but also drastically enhances the safety profile of the production environment, making it an ideal candidate for reliable API intermediate supplier partnerships aiming for large-scale commercialization without compromising on quality or regulatory compliance standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of Thymopentapeptide relied heavily on the classical Merrifield resin methodology, which, while technically mature, presents severe limitations for modern large-scale manufacturing. The primary drawback of the traditional approach lies in its reliance on hydrogen fluoride (HF) for the final cleavage step, a reagent known for its extreme corrosivity and high toxicity, necessitating specialized gas-generating units and rigorous safety containment systems that significantly inflate capital expenditure. Furthermore, the conventional process often involves a multi-step workup where the peptide must be extracted and lyophilized before refining, leading to extended production cycles and increased opportunities for product degradation or contamination. Alternative methods described in prior art, such as those utilizing specific bromoacetamide resins, introduce additional supply chain vulnerabilities due to the difficulty in sourcing these non-standard materials at a competitive cost, rendering them economically unviable for high-volume enterprise production despite their theoretical benefits in preventing racemization.
The Novel Approach
The novel approach detailed in the patent data fundamentally re-engineers the synthesis pathway by adopting an acid-labile Wang resin system coupled with Fmoc chemistry, effectively bypassing the need for hazardous HF cleavage. This methodology allows for the use of trifluoroacetic acid (TFA) to achieve both side-chain deprotection and resin cleavage in a single, unified operation, thereby collapsing multiple processing stages into one efficient step. By eliminating the requirement for specialized HF infrastructure, the new process lowers the barrier to entry for manufacturing facilities and reduces the operational complexity associated with handling dangerous gases. Additionally, the integration of diisopropylcarbodiimide (DIC) as the condensing agent ensures that the urea byproducts generated during coupling remain soluble in the reaction medium, facilitating easier washing procedures and minimizing the risk of impurity entrapment within the resin matrix, which ultimately translates to a higher quality crude product ready for final purification.
Mechanistic Insights into Fmoc/Wang Solid-Phase Synthesis
The core of this technological advancement lies in the strategic selection of the solid support and the orthogonal protection strategy employed throughout the peptide chain assembly. The Wang resin functions as an acid-labile linker, stable under the basic conditions required for Fmoc deprotection but readily cleavable under moderately acidic conditions provided by TFA. This chemical orthogonality is crucial because it allows the iterative addition of amino acids—Tyrosine, Valine, Aspartic Acid, Lysine, and Arginine—without prematurely releasing the growing peptide chain from the solid support. The use of Fmoc for alpha-amino protection ensures that deprotection can be achieved rapidly using mild bases like piperidine, avoiding the harsh acidic conditions of Boc chemistry that would otherwise compromise the acid-sensitive Wang linker before the synthesis is complete. This delicate balance of stability and lability is what enables the high output rate and simplified purification process cited in the patent documentation.
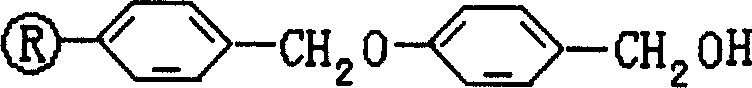
Furthermore, the mechanism of impurity control is significantly enhanced by the choice of coupling reagents and the cleavage cocktail composition. The utilization of DIC in the presence of additives like HOBt and DMAP promotes efficient amide bond formation while suppressing racemization, a common pitfall in peptide synthesis that can render the final API inactive or toxic. Unlike traditional carbodiimides that form insoluble precipitates, the soluble nature of the DIC-derived urea byproduct ensures that the resin beads remain accessible to reagents, preventing the formation of deletion sequences or incomplete couplings that are difficult to separate later. During the final acidolysis step, the inclusion of phenol in the TFA mixture acts as a scavenger, trapping reactive cationic species generated during the cleavage of side-chain protecting groups, thereby preventing alkylation side reactions on the sensitive aromatic residues of the thymopentapeptide sequence and ensuring a cleaner crude profile for downstream HPLC refinement.
How to Synthesize Thymopentapeptide Efficiently
The synthesis of Thymopentapeptide via this optimized route involves a logical sequence of resin loading, iterative coupling, and final cleavage, designed to maximize yield while minimizing operational hazards. The process begins with the preparation of the Fmoc-Tyr(tBu)-Wang resin, where the first amino acid is anchored to the solid support under controlled pH and temperature conditions to ensure high loading efficiency without steric hindrance. Following this initialization, the peptide chain is elongated through successive cycles of Fmoc deprotection and amino acid coupling, utilizing a molar excess of activated amino acids to drive the reactions to completion. The detailed standardized synthetic steps, including specific reagent ratios, reaction times, and washing protocols necessary to replicate this high-efficiency pathway, are outlined in the comprehensive guide below for technical teams evaluating process feasibility.
- Preparation of Fmoc-Tyr(tBu)-Wang Resin: React Wang resin with Fmoc-Tyr(tBu)-OH using DIC, HOBt, and DMAP in DMF at controlled pH and temperature to form the initial loaded resin.
- Sequential Peptide Coupling: Iteratively remove the Fmoc group using piperidine/DMF and couple subsequent protected amino acids (Val, Asp, Lys, Arg) using DIC/HOBt to build the pentapeptide chain.
- Acidolytic Cleavage and Purification: Treat the protected resin with a TFA/phenol mixture to simultaneously cleave the peptide from the resin and remove side-chain protecting groups, followed by ether precipitation and HPLC purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this Wang resin-based synthesis protocol offers substantial strategic benefits that extend far beyond simple chemical yield improvements. By eliminating the dependency on hydrogen fluoride, organizations can drastically reduce the capital investment required for safety infrastructure and ongoing maintenance of specialized gas handling systems, leading to significant cost reduction in pharmaceutical intermediates manufacturing. The simplified workflow, which merges cleavage and deprotection into a single step, shortens the overall production cycle time, allowing for faster turnover of batches and improved responsiveness to market demand fluctuations without the need for complex inventory buffering strategies. Moreover, the use of commercially available Wang resin and standard Fmoc-amino acids ensures a robust and diversified supply base, mitigating the risk of raw material shortages that often plague processes relying on proprietary or custom-synthesized solid supports.
- Cost Reduction in Manufacturing: The elimination of hazardous HF gas removes the need for expensive corrosion-resistant reactors and specialized waste treatment facilities, resulting in substantial operational expenditure savings. Additionally, the use of DIC as a coupling agent avoids the formation of insoluble byproducts, reducing solvent consumption during washing steps and lowering the overall cost of goods sold through improved material efficiency and reduced labor hours associated with filtration and cleaning.
- Enhanced Supply Chain Reliability: Sourcing standard Wang resin and common Fmoc-protected amino acids is significantly more reliable than procuring specialized bromoacetamide resins or managing the logistics of hazardous HF transport. This shift to commodity-grade raw materials enhances supply chain resilience, ensuring continuous production capabilities even during periods of global raw material volatility, and reduces the lead time for high-purity peptide intermediates by streamlining the procurement approval process.
- Scalability and Environmental Compliance: The milder reaction conditions and the absence of highly toxic HF gas make this process inherently safer and easier to scale from pilot plant to commercial production volumes. The reduced environmental footprint, characterized by simpler waste streams and lower energy requirements for safety containment, aligns perfectly with modern green chemistry initiatives and stringent environmental regulations, facilitating smoother regulatory approvals and long-term operational sustainability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthesis route, derived directly from the comparative analysis of the patent's background and beneficial effects. These insights are intended to clarify the operational advantages and safety improvements offered by the Wang resin methodology for stakeholders evaluating the technology for potential adoption or outsourcing. Understanding these nuances is critical for making informed decisions about process validation and supplier qualification in the competitive landscape of peptide manufacturing.
Q: Why is Wang resin preferred over Merrifield resin for TP-5 synthesis?
A: Wang resin is acid-labile, allowing for cleavage using trifluoroacetic acid (TFA) under mild conditions. In contrast, Merrifield resin requires hazardous hydrogen fluoride (HF) for cleavage, which poses severe safety risks and requires specialized industrial equipment.
Q: What are the advantages of using DIC as a condensing agent in this process?
A: Diisopropylcarbodiimide (DIC) forms a urea byproduct that is soluble in organic solvents, unlike the insoluble dicyclohexylurea (DCU) formed by DCC. This eliminates the need for difficult filtration steps, simplifies resin washing, and reduces impurity entrapment.
Q: How does the Fmoc strategy improve the purification of Thymopentapeptide?
A: The Fmoc protecting group is removed under mild alkaline conditions (piperidine), which is orthogonal to the acid-labile side-chain protections. This orthogonality, combined with the single-step TFA cleavage, minimizes side reactions and simplifies the final crude product profile for HPLC purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Thymopentapeptide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting safe, efficient, and scalable synthesis routes for complex immunomodulatory peptides like Thymopentapeptide. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory optimization to industrial manufacturing is seamless and compliant with global regulatory standards. We are committed to delivering products with stringent purity specifications, leveraging our rigorous QC labs to verify that every batch meets the exacting requirements of the pharmaceutical industry, thereby providing our partners with a secure and high-quality supply of essential API intermediates.
We invite you to engage with our technical procurement team to discuss how this optimized Wang resin synthesis can be tailored to your specific production needs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits of switching to this safer protocol. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of technical excellence and commercial reliability.
