Advanced Synthesis of 9,10-Dipyrrolyl Anthracene AIE Materials for Commercial Optoelectronics
Introduction to Next-Generation AIE Materials
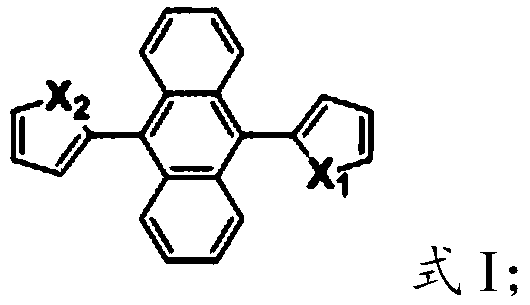
The landscape of organic optoelectronic materials is undergoing a significant transformation, driven by the limitations of traditional fluorophores that suffer from Aggregation-Caused Quenching (ACQ). Patent CN110143907B introduces a breakthrough class of 9,10-dipyrrolyl anthracene fluorescent compounds that exhibit superior Aggregation-Induced Emission (AIE) properties. These materials, including the symmetrical 9,10-di-1H-pyrrol-2-ylanthracene (DPA) and the asymmetric 9-(N-tert-butyloxycarbonyl-pyrrol-2-yl)-10-(1H-pyrrol-2-yl)anthracene (BPPA), represent a pivotal advancement for high-performance OLEDs and bio-imaging applications. Unlike conventional molecules that lose efficiency in solid films, these anthracene derivatives intensify their fluorescence upon aggregation, making them ideal candidates for solid-state lighting and display technologies where high concentration and film formation are critical requirements for device performance.
The strategic value of this technology lies in its dual capability to serve both biomedical and electronic sectors. The patent details how these compounds can effectively stain HeLa cells while maintaining high chemical and thermal stability, a rare combination that broadens their commercial utility. For procurement and R&D leaders in the electronic chemical sector, this represents an opportunity to diversify supply chains with materials that offer enhanced stability and simplified processing. The ability to tune fluorescence properties through simple structural modifications at the nitrogen position of the pyrrole ring allows for precise customization of emission wavelengths, particularly in the green light spectrum, which is essential for full-color display manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional organic light-emitting materials, particularly those based on planar aromatic systems, have long struggled with the ACQ effect, where intermolecular interactions in the solid state lead to non-radiative decay and reduced efficiency. Common AIEgens like tetraphenylethylene (TPE) derivatives often require complex multi-step syntheses involving harsh conditions and expensive catalysts, which drives up the cost of goods sold (COGS) for large-scale manufacturing. Furthermore, many existing anthracene-based emitters lack the necessary thermal stability for prolonged operation in high-brightness OLED devices, leading to premature device failure. The reliance on intricate molecular designs to prevent pi-pi stacking often compromises the charge transport properties, creating a trade-off between luminescence efficiency and electrical performance that limits commercial viability.
The Novel Approach
The methodology outlined in patent CN110143907B circumvents these issues by utilizing a robust 9,10-disubstituted anthracene core functionalized with pyrrole rings. This design inherently restricts intramolecular rotation in the aggregated state, triggering the AIE mechanism without the need for bulky steric groups that hinder packing. The synthesis strategy employs standard Suzuki-Miyaura cross-coupling reactions, which are well-understood and easily scalable in industrial settings. By introducing N-Boc protected pyrrole boronic acids, the process ensures high regioselectivity and yield, avoiding the formation of difficult-to-separate isomers. This streamlined approach not only reduces the number of purification steps but also utilizes readily available raw materials, positioning this novel compound class as a cost-effective alternative for next-generation electronic chemical manufacturing.
Mechanistic Insights into Suzuki-Miyaura Coupling and Deprotection
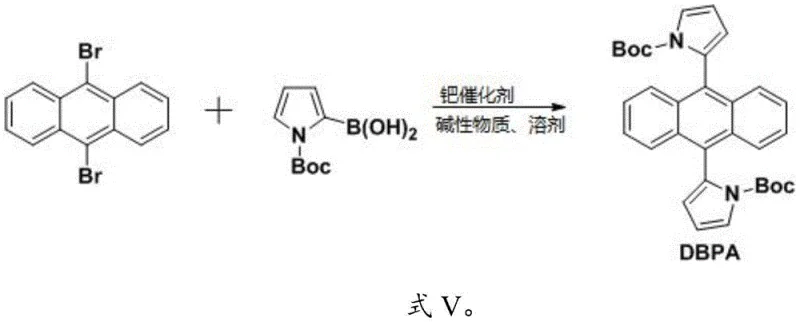
The core of this synthesis relies on the palladium-catalyzed cross-coupling between 9,10-dibromoanthracene and N-Boc-pyrrole-2-boronic acid. The reaction mechanism proceeds through the standard catalytic cycle of oxidative addition, transmetallation, and reductive elimination. In the oxidative addition step, the Pd(0) catalyst inserts into the carbon-bromine bond of the anthracene core, forming a reactive organopalladium intermediate. This is followed by transmetallation with the boronic acid species, activated by the base such as sodium carbonate or potassium phosphate. The choice of base and solvent system, typically THF or DMF with aqueous base, is critical for maintaining the stability of the boronic acid while facilitating the transfer of the pyrrole group to the metal center.
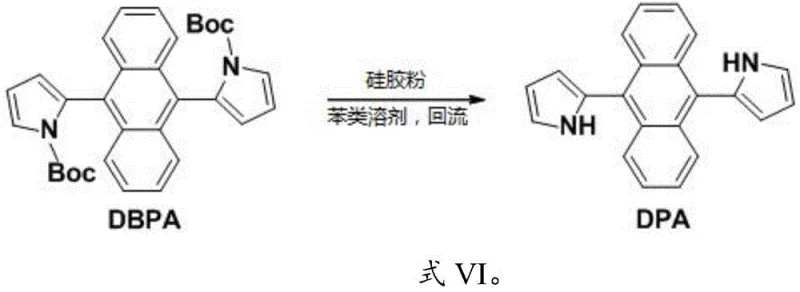
A distinct innovation in this patent is the deprotection strategy used to convert the N-Boc protected intermediates into the free NH pyrrole derivatives. Instead of using strong acids like trifluoroacetic acid which can degrade the sensitive anthracene backbone, the process utilizes silica gel powder in refluxing toluene. The weakly acidic surface of the silica acts as a solid-supported acid catalyst, cleaving the tert-butyloxycarbonyl group under mild thermal conditions. This heterogeneous approach simplifies the workup significantly, as the silica can be removed by simple filtration, eliminating the need for aqueous washes and reducing solvent consumption. This mechanistic nuance is crucial for maintaining high purity levels required for electronic grade materials.
How to Synthesize 9,10-Dipyrrolyl Anthracene Efficiently
The synthesis protocol described in the patent offers a versatile route to both symmetrical and asymmetrical derivatives, allowing manufacturers to tailor material properties for specific applications. The process begins with the double Suzuki coupling to form the fully protected DBPA intermediate, which serves as a stable precursor that can be stored or further modified. For the final DPA product, a simple reflux with silica gel achieves quantitative deprotection. For asymmetric variants like BPPA, stoichiometric control during the initial coupling allows for mono-substitution, followed by selective deprotection and a second coupling step. This modularity provides a significant advantage in R&D settings where rapid iteration of molecular structures is necessary to optimize device performance.
- Perform Suzuki coupling between 9,10-dibromoanthracene and N-Boc-pyrrole-2-boronic acid using a palladium catalyst and base in THF or DMF under reflux.
- For symmetrical DPA, subject the resulting DBPA intermediate to deprotection using silica gel powder in toluene under reflux conditions.
- For asymmetrical BPPA, control stoichiometry to mono-substitute, deprotect one side, and perform a second Suzuki coupling with protected boronic acid.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, the adoption of 9,10-dipyrrolyl anthracene synthesis offers tangible benefits in terms of raw material security and process robustness. The primary starting material, 9,10-dibromoanthracene, is a commodity chemical produced at scale for various dye and pigment applications, ensuring a stable and competitive supply base. Unlike specialized AIEgens that rely on bespoke building blocks with long lead times, this route leverages established global supply chains for anthracene derivatives and pyrrole boronic acids. This reduces the risk of supply disruptions and allows procurement managers to negotiate better pricing due to the availability of multiple qualified vendors for the key inputs.
- Cost Reduction in Manufacturing: The elimination of harsh acidic deprotection reagents and the use of silica gel significantly lowers the cost of waste treatment and disposal. Traditional acid deprotection generates large volumes of acidic wastewater requiring neutralization, whereas the silica method produces solid waste that is easier to handle. Additionally, the high yields reported in the patent, such as 96% for the deprotection step and 88% for the initial coupling, minimize raw material loss. This efficiency translates directly into lower production costs per kilogram, making high-purity OLED material more economically viable for mass-market display applications without compromising on quality standards.
- Enhanced Supply Chain Reliability: The synthetic route avoids the use of air-sensitive or pyrophoric reagents that often complicate logistics and storage. The palladium catalysts used, such as Pd(PPh3)4, are stable and commercially available in bulk quantities. The reaction conditions, primarily heating under reflux in common solvents like toluene or THF, are compatible with standard glass-lined or stainless steel reactors found in most fine chemical manufacturing facilities. This compatibility means that contract manufacturing organizations (CMOs) can produce these materials without requiring significant capital investment in specialized equipment, thereby increasing the number of potential suppliers and enhancing overall supply chain resilience.
- Scalability and Environmental Compliance: The process is inherently green due to the atom economy of the Suzuki coupling and the recyclable nature of the silica mediator. Scaling from gram to kilogram scale is straightforward as the reaction kinetics are not dependent on exotic mixing or temperature profiles. The absence of halogenated solvents in the deprotection step (using toluene instead of DCM for the reaction, though DCM is used in workup) aligns with increasingly stringent environmental regulations regarding volatile organic compounds (VOCs). This regulatory alignment future-proofs the manufacturing process against tightening environmental laws, ensuring long-term operational continuity for partners investing in this technology.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these advanced fluorescent compounds. Understanding these details is essential for stakeholders evaluating the integration of 9,10-dipyrrolyl anthracene derivatives into their product portfolios. The answers are derived directly from the experimental data and technical specifications provided in the underlying patent documentation, ensuring accuracy and reliability for decision-making processes.
Q: What are the primary advantages of 9,10-dipyrrolyl anthracene over traditional AIE materials?
A: Unlike traditional ACQ materials that quench in solid state, 9,10-dipyrrolyl anthracene derivatives exhibit strong Aggregation-Induced Emission (AIE). They offer higher fluorescence quantum yields in aggregated states, better chemical stability, and simpler synthesis routes compared to complex tetraphenylethylene derivatives.
Q: How does the silica-mediated deprotection method improve the manufacturing process?
A: The use of silica gel powder as a weak acid source for Boc deprotection eliminates the need for harsh mineral acids like TFA or HCl. This mild condition prevents structural degradation of the sensitive anthracene core, simplifies workup procedures, and reduces hazardous waste generation, significantly enhancing process safety and environmental compliance.
Q: Can these compounds be scaled for industrial OLED production?
A: Yes, the synthesis relies on robust Suzuki-Miyaura coupling reactions using commercially available starting materials like 9,10-dibromoanthracene. The high yields reported (up to 96% for deprotection steps) and mild reaction conditions make the process highly amenable to kilogram-scale manufacturing for display and lighting applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 9,10-Dipyrrolyl Anthracene Supplier
As the demand for high-efficiency AIE materials grows in the OLED and bio-imaging sectors, having a manufacturing partner with deep technical expertise is crucial. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our rigorous QC labs and stringent purity specifications guarantee that every batch of 9,10-dipyrrolyl anthracene meets the exacting standards required for electronic grade applications, minimizing defects in downstream device fabrication.
We invite you to discuss how our optimized synthesis routes can drive value for your organization. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and route feasibility assessments to help you accelerate your development timelines and secure a competitive edge in the rapidly evolving market for advanced optoelectronic materials.
