Advanced Catalytic Synthesis of 2-Substituted Methylene Dihydrobenzothiazole Derivatives for Commercial Scale
Introduction to Patent CN111116510B Technology
The pharmaceutical and fine chemical industries are constantly seeking efficient routes to access complex heterocyclic scaffolds that serve as critical building blocks for bioactive molecules. Patent CN111116510B introduces a groundbreaking methodology for the synthesis of a specific class of 2-substituted methylene dihydrobenzo[d]thiazole derivatives, which are structurally distinct from their more common aromatic benzothiazole counterparts. This innovation addresses a significant gap in synthetic chemistry, where non-aromatic dihydrobenzothiazoles are historically underrepresented due to the difficulty in controlling their formation without over-oxidation to the aromatic system. The disclosed technology leverages a sophisticated tandem reaction sequence involving intramolecular enethiol tautomerization followed by C-S coupling, enabling the construction of these valuable bicyclic systems with exceptional precision.
![General chemical structure of 2-substituted methylene dihydrobenzo[d]thiazole derivatives (Formula 2)](/insights/img/dihydrobenzothiazole-synthesis-pharma-supplier-20260306104826-01.png)
From a commercial perspective, the ability to produce these derivatives under mild conditions represents a substantial advancement for supply chain stability and cost management. The patent details a protocol that utilizes readily available N-alkyl or N-aryl thioamide derivatives as starting materials, transforming them into high-value intermediates through a catalytic oxidative process. For procurement managers and R&D directors alike, this translates to a reliable source of complex intermediates that can be integrated into diverse drug discovery pipelines or material science applications. The versatility of the substituent groups tolerated by this reaction further enhances its utility, allowing for the rapid generation of structural analogs necessary for structure-activity relationship (SAR) studies without the burden of developing new synthetic routes for each variant.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the construction of benzothiazole-containing compounds has heavily favored the formation of fully aromatic systems, often necessitating harsh reaction conditions such as high temperatures, strong acids, or stoichiometric amounts of toxic heavy metal oxidants. These conventional pathways frequently suffer from poor chemoselectivity, leading to complex mixtures of by-products that require extensive and costly purification steps to isolate the desired material. Furthermore, accessing the dihydrobenzothiazole motif specifically is particularly challenging because the thermodynamic drive towards aromatization often overrides the kinetic control needed to stop at the dihydro stage. This limitation forces process chemists to design multi-step sequences with protecting group strategies, significantly increasing the overall step count, reducing atom economy, and inflating the cost of goods sold (COGS) for the final active pharmaceutical ingredient.
The Novel Approach
In stark contrast, the methodology described in CN111116510B offers a streamlined, single-pot solution that bypasses these historical hurdles through a clever mechanistic design. By employing a cobalt-catalyzed oxidative dehydrogenative coupling strategy, the process facilitates the direct cyclization of thioamide precursors into the dihydrobenzothiazole core with remarkable efficiency. 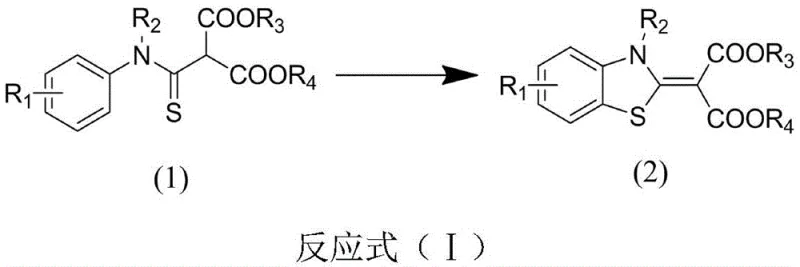
This novel approach operates at room temperature, eliminating the energy costs associated with heating and the safety risks of high-pressure reactors. The reaction demonstrates high regioselectivity and chemical selectivity, ensuring that the sulfur and nitrogen atoms couple precisely to form the desired bicyclic ring without affecting other sensitive functional groups present on the substrate. For a reliable pharmaceutical intermediate supplier, adopting this route means offering clients a cleaner process with higher throughput and reduced waste generation, directly addressing the industry's growing demand for greener and more sustainable manufacturing practices.
Mechanistic Insights into Cobalt-Catalyzed Oxidative Cyclization
The core of this technological breakthrough lies in the intricate interplay between the cobalt catalyst and the oxidant, which orchestrates a cascade of electronic rearrangements within the substrate molecule. The mechanism initiates with the activation of the thioamide derivative, likely through coordination with the cobalt species, which lowers the energy barrier for the subsequent enethiol tautomerization. This tautomerization is a critical prerequisite, as it generates the nucleophilic sulfur species necessary for the intramolecular attack on the adjacent carbon center. The presence of the oxidant, preferably DDQ (2,3-dichloro-5,6-dicyano-1,4-benzoquinone), serves to regenerate the active catalytic species and drive the equilibrium forward by removing hydrogen equivalents, effectively locking the molecule into the cyclized dihydrobenzothiazole form.
Furthermore, the choice of anhydrous cobalt bromide as the preferred catalyst is not arbitrary; it provides the optimal Lewis acidity and redox potential to facilitate this specific transformation without promoting over-oxidation to the aromatic benzothiazole. The reaction conditions are finely tuned to maintain a delicate balance where the C-S bond formation occurs rapidly, but the subsequent dehydrogenation required for aromatization is kinetically inhibited. This level of control is paramount for R&D teams focusing on impurity profiling, as it minimizes the formation of structurally related aromatic impurities that are notoriously difficult to separate. The result is a process that delivers high-purity dihydrobenzothiazole derivatives, meeting the stringent quality specifications required for clinical trial materials and commercial drug substances.
How to Synthesize 2-Substituted Methylene Dihydrobenzothiazole Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires strict adherence to the anhydrous conditions and reagent ratios specified in the patent to ensure reproducibility and maximum yield. The process begins with the dissolution of the N-alkyl/N-aryl thioamide derivative in anhydrous acetonitrile, followed by the sequential addition of the cobalt catalyst and the oxidant. Maintaining the reaction at room temperature is crucial, as deviations could alter the reaction kinetics and potentially compromise the selectivity for the dihydro product. Monitoring the reaction progress via thin-layer chromatography (TLC) allows operators to determine the precise endpoint, typically achieved within 4 to 20 hours depending on the electronic nature of the substituents on the aromatic ring.
- Prepare the reaction mixture by combining the N-alkyl/N-aryl thioamide derivative substrate with anhydrous cobalt bromide catalyst and DDQ oxidant in anhydrous acetonitrile solvent at room temperature.
- Stir the reaction mixture for 4 to 20 hours while monitoring progress via TLC until the starting material is completely consumed and the intramolecular C-S coupling is complete.
- Quench the reaction with saturated sodium thiosulfate solution, extract with dichloromethane, wash with bicarbonate, dry over sodium sulfate, and purify the residue via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible benefits that extend beyond mere chemical elegance, directly impacting the bottom line and operational resilience. The primary advantage lies in the significant reduction of manufacturing complexity, as the one-pot nature of the reaction eliminates the need for intermediate isolation and purification steps that are common in traditional multi-step syntheses. This simplification translates to shorter production cycles and reduced labor costs, allowing suppliers to respond more agilely to fluctuating market demands. Additionally, the use of commodity chemicals like cobalt bromide and acetonitrile ensures that raw material sourcing remains stable and cost-effective, mitigating the risks associated with supply chain disruptions for exotic or proprietary reagents.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts like palladium or rhodium, which are often required for C-S bond formation in other methodologies, results in substantial cost savings. Moreover, the high yields reported, reaching up to 99% in optimized examples, mean that less raw material is wasted, and the burden on downstream waste treatment facilities is significantly lowered. This efficiency allows for a more competitive pricing structure for the final intermediate, providing a clear economic advantage in cost reduction in fine chemical manufacturing without compromising on quality or purity standards.
- Enhanced Supply Chain Reliability: By utilizing robust and widely available reagents, the dependency on single-source suppliers for critical catalysts is minimized, thereby enhancing the overall reliability of the supply chain. The mild reaction conditions also reduce the wear and tear on manufacturing equipment, leading to lower maintenance costs and fewer unplanned downtime events. This operational stability ensures consistent delivery schedules for clients, which is a critical factor for pharmaceutical companies managing tight development timelines and regulatory submission deadlines.
- Scalability and Environmental Compliance: The scalability of this process is inherently high due to the absence of hazardous high-temperature or high-pressure steps, making it easier to transfer from gram-scale laboratory synthesis to ton-scale commercial production. From an environmental perspective, the atom-economical nature of the reaction and the reduced solvent usage align with modern green chemistry principles, helping manufacturers meet increasingly stringent environmental regulations. This compliance reduces the risk of regulatory fines and enhances the corporate sustainability profile, which is becoming a key differentiator in the global chemical market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis technology. These insights are derived directly from the experimental data and claims presented in CN111116510B, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this route into their existing manufacturing portfolios or research programs.
Q: What are the optimal reaction conditions for synthesizing dihydrobenzothiazole derivatives according to CN111116510B?
A: The patent specifies using anhydrous cobalt bromide as the preferred catalyst and DDQ as the oxidant in anhydrous acetonitrile at room temperature, achieving yields up to 99%.
Q: Why is this synthetic method superior to conventional benzothiazole construction techniques?
A: Unlike traditional methods that often require harsh conditions to form aromatic benzothiazoles, this protocol operates under mild room temperature conditions to access the rarer non-aromatic dihydrobenzothiazole scaffold with high atom economy.
Q: Is this process scalable for industrial production of pharmaceutical intermediates?
A: Yes, the use of inexpensive cobalt salts, common organic solvents, and ambient temperature conditions eliminates the need for specialized high-pressure or cryogenic equipment, facilitating straightforward commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Substituted Methylene Dihydrobenzothiazole Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of high-quality heterocyclic intermediates in the development of next-generation therapeutics and advanced materials. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. We are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 2-substituted methylene dihydrobenzothiazole derivatives meets the exacting standards required by global regulatory bodies. Our commitment to excellence extends beyond simple supply; we act as a strategic partner, offering technical support to optimize the application of these intermediates in your specific synthesis targets.
We invite you to contact our technical procurement team to discuss how this innovative synthetic route can benefit your project. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic potential of switching to this cobalt-catalyzed method. We are ready to provide specific COA data and route feasibility assessments tailored to your needs, ensuring that you have all the necessary information to make informed sourcing decisions. Partner with us to leverage cutting-edge chemistry for your supply chain, securing a competitive edge in the fast-evolving pharmaceutical and chemical landscapes.
