Advanced Fmoc-Solid Phase Strategy for Commercial Eptifibatide Manufacturing and Process Optimization
Introduction to Next-Generation Eptifibatide Synthesis
The pharmaceutical landscape for cardiovascular therapeutics demands increasingly efficient and environmentally sustainable manufacturing processes, a standard exemplified by the innovative methodology disclosed in patent CN1222537C. This pivotal intellectual property details a robust Fmoc-strategy solid-phase synthesis for Eptifibatide, also known commercially as Integrilin, a potent glycoprotein IIb/IIIa inhibitor used in acute coronary syndromes. Unlike traditional liquid-phase syntheses that suffer from protracted timelines and laborious purification steps, or Boc-strategy solid-phase methods that rely on hazardous hydrogen fluoride, this novel approach leverages mild Fmoc chemistry to achieve superior operational safety and product quality. For global procurement leaders and R&D directors, understanding this shift is critical, as it represents a transition towards greener, more cost-effective peptide manufacturing that aligns with modern regulatory expectations for impurity control and worker safety.
The core innovation lies in the strategic combination of Fmoc-protected amino acids with a specialized resin system, specifically Fmoc-Rink Amide AM resin, which facilitates the construction of the complex cyclic heptapeptide structure with high fidelity. By systematically removing the Fmoc protecting group with piperidine and coupling subsequent residues using activated esters, the process ensures a linear assembly that is easily monitored via Kaiser tests. This level of process analytical technology (PAT) integration allows for immediate detection of incomplete reactions, thereby preventing the accumulation of difficult-to-remove deletion impurities that often plague large-scale peptide production. Consequently, this patent provides a blueprint for a reliable eptifibatide intermediate supplier to deliver materials with consistently high purity profiles, essential for meeting the stringent specifications of finished drug products.
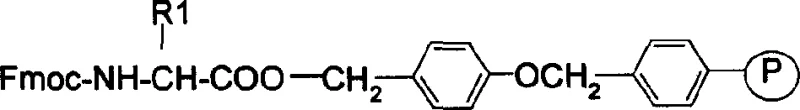
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of complex cyclic peptides like Eptifibatide has been hindered by the inherent inefficiencies of liquid-phase synthesis and the safety hazards associated with Boc-chemistry. In liquid-phase methodologies, every synthetic step necessitates extensive workup and purification procedures, such as crystallization or chromatography, which drastically extend the overall synthesis cycle to impractical durations often described metaphorically as spanning entire harvest seasons. Furthermore, the Boc-strategy solid-phase alternative, while faster, introduces severe safety and environmental liabilities due to its reliance on anhydrous hydrogen fluoride for final deprotection and resin cleavage. HF is not only acutely toxic and corrosive, requiring specialized equipment and rigorous safety protocols, but its disposal generates significant environmental burdens that increase operational costs and complicate regulatory compliance for any cost reduction in peptide manufacturing initiative.
The Novel Approach
The Fmoc-strategy outlined in the patent data offers a transformative solution by replacing harsh acidic conditions with mild base-labile deprotection, effectively mitigating the risks associated with HF handling. This approach utilizes piperidine in DMF for Fmoc removal, a reaction that is rapid, quantitative, and easily scalable without the need for exotic corrosion-resistant reactors. Moreover, the process incorporates a unique fragment condensation strategy where the challenging homoarginine residue is pre-synthesized from lysine under homogeneous conditions, ensuring complete conversion before attachment to the growing peptide chain. This hybrid solution-phase/solid-phase tactic overcomes the difficulty of monitoring heterogeneous reactions on-resin, thereby enhancing the overall yield and simplifying the downstream purification workflow. For supply chain heads, this translates to a more predictable production schedule and reduced lead time for high-purity cyclic peptides, securing a stable supply of this critical cardiovascular API.
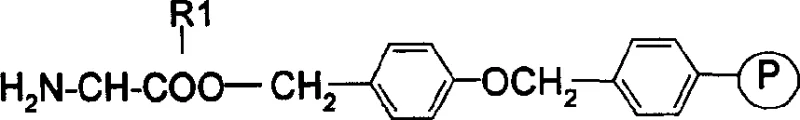
Mechanistic Insights into Fmoc-Solid Phase Peptide Synthesis
The mechanistic elegance of this process rests on the orthogonality of the Fmoc protecting group, which allows for selective deprotection in the presence of acid-labile side-chain protectors like Trt (trityl) and tBu (tert-butyl). The synthesis initiates with the loading of the C-terminal amino acid onto the Fmoc-Rink Amide AM resin, establishing a stable anchor that will eventually yield the C-terminal amide upon cleavage. As the chain elongates, reagents such as HBTU (O-Benzotriazole-N,N,N',N'-tetramethyl-uronium-hexafluoro-phosphate) and HOBt (Hydroxybenzotriazole) are employed to activate the carboxyl groups of incoming Fmoc-amino acids, forming reactive O-acylisourea intermediates that couple efficiently with the free amine of the resin-bound peptide. This activation method minimizes racemization, a critical concern in peptide synthesis, ensuring that the stereochemical integrity of the chiral centers is preserved throughout the assembly of the seven-residue sequence.
A distinct advantage of this mechanism is the ability to track reaction progress in real-time using the Kaiser test (ninhydrin test), which provides a colorimetric indication of free primary amines. If the test indicates incomplete coupling, the reaction can be immediately driven to completion by adding fresh reagents, a capability that is absent in many liquid-phase processes where intermediates are isolated blindly. Furthermore, the specific handling of the homoarginine moiety involves converting the epsilon-amino group of a lysine derivative into a guanidino group in solution, creating the Mpr-Har fragment. This fragment is then coupled to the N-terminus of the resin-bound pentapeptide, a strategy that circumvents the steric and electronic challenges of introducing bulky guanidino groups directly on the solid support. The final cleavage using Reagent K (a cocktail of TFA, phenol, water, thioanisole, and ethanedithiol) simultaneously removes all side-chain protecting groups and releases the linear peptide, which is subsequently oxidized and cyclized to form the disulfide bridge essential for biological activity.

How to Synthesize Eptifibatide Efficiently
The implementation of this Fmoc-solid phase protocol requires precise adherence to reaction conditions to maximize yield and minimize epimerization. The process begins with the swelling of the resin and sequential deprotection cycles, followed by the coupling of standard amino acids like Cysteine, Proline, Tryptophan, Aspartic Acid, and Glycine. Critical attention must be paid to the preparation of the Mpr-Har fragment, where lysine is first protected, esterified, and then reacted with S-triphenylmercaptopropionic acid before undergoing guanidinylation. Once the full linear sequence is assembled on the resin, the crude peptide is cleaved using the specific Reagent K formulation (TFA:phenol:water:thioanisole:ethanedithiol in an 82.5:5:5:5:2.5 ratio), which acts as a scavenger system to prevent side reactions during acidolysis.
- Deprotect Fmoc-Rink Amide AM resin using 20% piperidine in DMF to obtain H2N-Rink Amide AM resin.
- Sequentially couple protected amino acids (Fmoc-Cys(Trt)-OH, Fmoc-Pro-OH, etc.) using HBTU/HOBt activation.
- Prepare the Mpr-Har fragment separately from Lysine and couple it to the resin-bound peptide chain.
- Cleave the peptide from the resin using Reagent K, followed by oxidation and cyclization to form the final cyclic heptapeptide.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers evaluating the total cost of ownership for Eptifibatide intermediates, the shift to this Fmoc-based solid-phase process offers substantial economic benefits derived from process intensification and hazard elimination. By removing the requirement for hydrogen fluoride, manufacturers can avoid the capital expenditure associated with HF-compatible infrastructure and the ongoing costs of hazardous waste disposal, leading to significant cost reduction in peptide manufacturing operations. Additionally, the ability to monitor each coupling step via Kaiser testing reduces the risk of batch failures due to incomplete reactions, thereby improving the overall material throughput and reducing the consumption of expensive protected amino acids. This efficiency gain is compounded by the shorter synthesis cycle, which allows for higher asset utilization rates and the ability to respond more agilely to market demand fluctuations without maintaining excessive inventory buffers.
- Cost Reduction in Manufacturing: The elimination of toxic HF and the reduction in purification steps directly lower the variable costs associated with safety compliance and solvent consumption. The use of standard Fmoc-amino acids, which are widely available from multiple global vendors, ensures competitive pricing and reduces dependency on single-source suppliers for exotic reagents. Furthermore, the improved crude purity resulting from the homogeneous Har-fragment synthesis reduces the load on preparative HPLC columns during final purification, extending column life and decreasing solvent usage per kilogram of product.
- Enhanced Supply Chain Reliability: The modular nature of solid-phase synthesis allows for flexible production scheduling, where different peptide sequences can be manufactured on the same equipment with minimal changeover time. This flexibility enhances supply chain resilience, enabling the rapid scaling of production volumes from pilot batches to commercial quantities of 100 kgs to 100 MT/annual commercial production as demand dictates. The robustness of the Fmoc chemistry also ensures consistent batch-to-batch quality, reducing the risk of supply disruptions caused by out-of-specification results that often delay product release in more volatile liquid-phase processes.
- Scalability and Environmental Compliance: The process aligns with green chemistry principles by substituting hazardous reagents with safer alternatives and minimizing waste generation through higher yields. The scalability of the solid-phase method is well-documented in the industry, allowing for the seamless transition from gram-scale R&D to multi-kilogram GMP manufacturing without fundamental changes to the reaction chemistry. This ease of scale-up, combined with the reduced environmental footprint, positions the manufacturer favorably for audits by environmentally conscious pharmaceutical clients who prioritize sustainability in their supplier selection criteria.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the Fmoc-solid phase production of Eptifibatide, providing clarity on the process advantages and quality controls. These insights are derived directly from the technical specifications of patent CN1222537C and represent the standard operating parameters for high-quality peptide synthesis. Understanding these details helps stakeholders assess the feasibility and reliability of this manufacturing route for their specific supply chain requirements.
Q: Why is the Fmoc-strategy preferred over Boc-strategy for Eptifibatide production?
A: The Fmoc-strategy eliminates the need for highly toxic and corrosive hydrogen fluoride (HF) used in Boc-deprotection, significantly improving operator safety and reducing environmental waste treatment costs.
Q: How does this process ensure high purity of the cyclic peptide?
A: By converting Lysine to Homoarginine under homogeneous conditions prior to coupling and utilizing Kaiser tests for real-time monitoring, the process minimizes deletion sequences and simplifies final HPLC purification.
Q: Is this synthesis method scalable for commercial supply?
A: Yes, the solid-phase approach reduces the synthesis cycle to approximately 1-2 days per batch compared to lengthy liquid-phase methods, facilitating rapid scale-up from pilot to multi-ton commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Eptifibatide Supplier
At NINGBO INNO PHARMCHEM, we leverage advanced synthetic methodologies like the Fmoc-solid phase strategy to deliver high-purity Eptifibatide intermediates that meet the rigorous demands of the global pharmaceutical market. Our facility is equipped with state-of-the-art peptide synthesizers and purification systems, backed by extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We maintain stringent purity specifications and operate rigorous QC labs to ensure that every batch of cyclic peptide we produce adheres to the highest standards of quality and safety, providing our partners with the confidence they need to advance their drug development pipelines.
We invite you to collaborate with us to optimize your supply chain for cardiovascular therapeutics. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our optimized Fmoc-process can enhance your project's efficiency and profitability.
