Advanced Solid Phase Synthesis of Thymopentin: Scaling High-Purity Immunomodulatory Peptides
Advanced Solid Phase Synthesis of Thymopentin: Scaling High-Purity Immunomodulatory Peptides
The pharmaceutical industry continuously seeks robust methodologies for producing bioactive peptides that balance high purity with operational safety. Patent CN1260250C introduces a significant advancement in the preparation of Thymopentin (TP-5), a critical immunomodulatory pentapeptide consisting of the sequence Arg-Lys-Asp-Val-Tyr. This intellectual property outlines a refined solid-phase peptide synthesis (SPPS) strategy that replaces hazardous hydrogen fluoride (HF) cleavage protocols with a milder trifluoroacetic acid (TFA) system utilizing Wang resin. For R&D directors and procurement specialists evaluating peptide intermediates, this transition represents a pivotal shift towards safer, more scalable manufacturing processes. By leveraging the acid-labile properties of 4-hydroxymethyl phenoxymethyl polystyrene (Wang resin) combined with Fmoc-protected amino acids, the method achieves simultaneous side-chain deprotection and resin cleavage. This technical breakthrough not only mitigates the severe safety hazards associated with traditional HF gas generation units but also streamlines the downstream purification workflow, offering a compelling value proposition for commercial-scale API intermediate production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Thymopentin relied heavily on Merrifield resin coupled with Boc (tert-butyloxycarbonyl) protection strategies. While technically mature, this conventional approach suffers from profound logistical and safety deficiencies that hinder modern large-scale manufacturing. The primary bottleneck lies in the cleavage step, which necessitates the use of liquid hydrogen fluoride (HF). HF is an extremely corrosive and toxic gas that demands specialized, expensive containment infrastructure and rigorous safety protocols to prevent catastrophic leakage and personnel injury. Furthermore, the HF cleavage process often requires gradient conditions that limit the batch size of peptide resins that can be processed simultaneously, creating a bottleneck in throughput. Additionally, alternative methods described in prior art, such as those utilizing glycolamide ester linkages, introduce their own complexities, including the need for specific, costly resins and multi-step cleavage procedures involving saponification. These multi-step processes often introduce inorganic salt impurities, such as sodium chloride, which complicate the subsequent high-performance liquid chromatography (HPLC) purification and reduce overall yield.
The Novel Approach
The methodology disclosed in CN1260250C fundamentally reengineers the synthesis pathway by adopting Wang resin as the solid support and employing an Fmoc (fluorenylmethyloxycarbonyl) protection strategy. This novel approach capitalizes on the acid sensitivity of the benzyl ester linkage in Wang resin, allowing the peptide chain to be cleaved under relatively mild acidic conditions using TFA. Unlike the harsh HF treatment, TFA cleavage can be performed in standard glass-lined or stainless steel reactors without the need for exotic gas-handling systems. A critical innovation in this process is the utilization of DIC (N,N'-diisopropylcarbodiimide) as the coupling activator in conjunction with HOBT. This specific reagent combination prevents the formation of N,N'-dicyclohexylurea (DCU), an insoluble byproduct common with DCC coupling that is notoriously difficult to wash away from the resin matrix. By eliminating DCU precipitation, the new method ensures a cleaner resin bed, facilitates more efficient washing cycles, and significantly reduces the risk of particulate contamination in the final crude peptide, thereby enhancing the efficiency of the refining process.
Mechanistic Insights into Wang Resin-Mediated Acidolysis
The core of this technological advancement lies in the chemical stability profile of the Wang resin linker. The Wang resin functions through a benzyl ether linkage that connects the polystyrene matrix to the growing peptide chain via an ester bond at the C-terminus. This ester bond is stable to the basic conditions used for Fmoc deprotection (typically piperidine in DMF) but is highly labile to strong acids like trifluoroacetic acid. During the final acidolysis step, the protonation of the benzylic oxygen generates a stable carbocation intermediate, leading to the cleavage of the ester bond and the release of the free carboxylic acid of the C-terminal tyrosine residue. Crucially, the TFA cocktail, often supplemented with scavengers like phenol, simultaneously removes the acid-labile side-chain protecting groups (such as tBu on Tyrosine and Asp, and Boc on Lys and Arg). This concurrent cleavage and deprotection mechanism collapses what was previously a multi-stage operation into a single, efficient unit operation, drastically reducing processing time and solvent consumption.
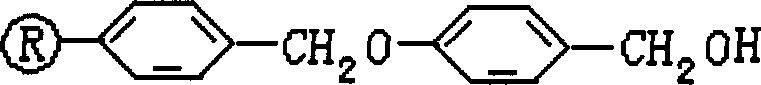
Furthermore, the choice of DIC as the coupling agent plays a vital role in maintaining the integrity of the peptide sequence and minimizing racemization. In traditional peptide synthesis, the formation of urea byproducts can physically trap reactants within the resin beads, leading to incomplete couplings and deletion sequences. The soluble nature of the diisopropylurea byproduct formed by DIC ensures that the resin pores remain open and accessible for subsequent amino acid additions. This mechanistic advantage directly translates to higher stepwise yields and a cleaner impurity profile. For quality control teams, this means the crude TP-5 obtained after ether precipitation contains fewer structurally related impurities, reducing the load on preparative HPLC columns and extending their operational lifespan. The precise control over the reaction environment, maintained between 10°C and 40°C during loading and coupling, further suppresses epimerization, ensuring the biological activity of the final immunomodulatory product remains consistent with pharmacopeial standards.
How to Synthesize Thymopentin Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for transitioning from laboratory benchtop to pilot plant operations. The process begins with the loading of the first amino acid, Fmoc-Tyr(tBu)-OH, onto the Wang resin using a defined molar ratio of activators to ensure high substitution levels without overcrowding the resin matrix. Following the initial loading, the peptide chain is elongated through iterative cycles of deprotection and coupling. Each cycle involves treating the resin with a piperidine solution to remove the Fmoc group, followed by the addition of the next Fmoc-protected amino acid activated by DIC and HOBT. The sequence proceeds logically from Tyrosine to Valine, Aspartic Acid, Lysine, and finally Arginine. The final step involves the global deprotection and cleavage using a TFA/phenol mixture, followed by precipitation with anhydrous diethyl ether to isolate the crude powder. For a detailed breakdown of the specific reagent quantities, reaction times, and washing protocols required to execute this synthesis, please refer to the standardized guide below.
- Load Fmoc-Tyr(tBu)-OH onto Wang resin using DIC/HOBt activation in DMF to form the initial resin-bound amino acid.
- Perform sequential coupling cycles: Deprotect Fmoc with piperidine, then couple Fmoc-Val-OH, Fmoc-Asp(OtBu)-OH, Fmoc-Lys(Boc)-OH, and Boc-Arg-OH using DIC/HOBt.
- Cleave the protected pentapeptide from the resin and remove side-chain protecting groups simultaneously using a TFA/phenol mixture, followed by ether precipitation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this Wang resin-based synthesis route offers substantial strategic benefits beyond mere technical feasibility. The elimination of hydrogen fluoride from the process map removes a significant regulatory and safety burden, allowing manufacturing to occur in facilities that may not be equipped for handling extreme hazardous gases. This flexibility expands the pool of potential contract manufacturing organizations (CMOs) capable of producing the intermediate, thereby strengthening supply chain resilience and reducing the risk of production stoppages due to safety audits or equipment failures. Moreover, the simplified workflow, characterized by the absence of DCU filtration and the consolidation of cleavage and deprotection steps, leads to a drastic reduction in cycle time. Faster cycle times mean increased asset turnover and the ability to respond more agilely to market demand fluctuations for immunomodulatory therapies.
- Cost Reduction in Manufacturing: The economic implications of switching to this methodology are profound, primarily driven by the reduction in operational complexity and waste management costs. By utilizing DIC instead of DCC, the process eliminates the labor-intensive and solvent-heavy steps required to filter out insoluble DCU byproducts, directly lowering solvent procurement and disposal expenses. Furthermore, the replacement of HF with TFA removes the need for capital-intensive HF scrubbing systems and specialized containment vessels, significantly lowering the barrier to entry for production and reducing depreciation costs per kilogram of output. The higher condensation yields reported in the patent imply less raw material waste, as fewer amino acids are required to achieve the same theoretical output, contributing to a leaner cost of goods sold (COGS) structure.
- Enhanced Supply Chain Reliability: Supply continuity is often jeopardized by the reliance on hazardous reagents that face strict transportation and storage regulations. TFA and Fmoc-amino acids are commodity chemicals with robust, global supply chains, unlike the specialized and tightly controlled hydrogen fluoride required for traditional methods. This shift ensures that raw material availability is less susceptible to geopolitical or regulatory disruptions. Additionally, the mild reaction conditions reduce the likelihood of equipment corrosion and maintenance downtime, ensuring that production lines remain operational for longer periods. The simplified purification process, resulting from the cleaner crude product profile, also shortens the lead time for releasing batches, enabling faster delivery to downstream formulation partners.
- Scalability and Environmental Compliance: As the demand for peptide therapeutics grows, the ability to scale processes without exponential increases in environmental footprint is critical. This method's reliance on liquid-phase reagents rather than hazardous gases simplifies the engineering controls needed for scale-up, making it easier to transition from 100 kg to multi-ton annual production capacities. The reduction in hazardous waste generation, particularly the avoidance of fluoride-containing waste streams, aligns with increasingly stringent environmental, social, and governance (ESG) mandates. This compliance advantage not only mitigates regulatory risk but also enhances the brand reputation of the manufacturer as a sustainable partner in the pharmaceutical value chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Thymopentin using this advanced solid-phase methodology. These insights are derived directly from the comparative data and experimental examples provided in the patent literature, aiming to clarify the operational benefits for potential partners. Understanding these nuances is essential for evaluating the feasibility of integrating this intermediate into your existing supply network.
Q: Why is Wang resin preferred over Merrifield resin for Thymopentin production?
A: Wang resin is acid-labile, allowing cleavage with trifluoroacetic acid (TFA) under mild conditions. In contrast, Merrifield resin requires hazardous hydrogen fluoride (HF) for cleavage, which poses severe safety risks and requires specialized industrial equipment.
Q: How does the use of DIC improve the purity of the final peptide?
A: Unlike DCC (dicyclohexylcarbodiimide), the coupling agent DIC (diisopropylcarbodiimide) forms a soluble urea byproduct. This eliminates the formation of insoluble DCU precipitates, simplifying resin washing and significantly reducing particulate impurities in the crude product.
Q: What are the scalability advantages of this TFA-based method?
A: The process eliminates the need for gaseous HF handling systems and complex multi-step cleavage procedures. The one-step TFA acidolysis allows for simpler reactor designs and faster turnaround times, facilitating easier scale-up from laboratory to commercial tonnage.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Thymopentin Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to safer, more efficient peptide synthesis routes is critical for the future of immunomodulatory drug development. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical advantages of the Wang resin/TFA method are fully realized in a GMP-compliant environment. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify the identity and potency of every batch of Thymopentin. Our commitment to quality ensures that the impurity profiles of our intermediates meet the demanding requirements of global regulatory agencies, providing a solid foundation for your final drug product filings.
We invite you to collaborate with us to optimize your peptide supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our advanced manufacturing capabilities can drive value and security for your organization.
