Advanced Synthetic Route for Stable Isotope Labeled Phenylethanolamine A Intermediates
Advanced Synthetic Route for Stable Isotope Labeled Phenylethanolamine A Intermediates
The global demand for precise analytical standards in food safety and veterinary drug monitoring has never been more critical, particularly concerning beta-agonists like Phenylethanolamine A. Patent CN111777518A introduces a groundbreaking synthetic preparation method for stable isotope-labeled Phenylethanolamine A, addressing the urgent need for domestic production of high-purity internal standards. Historically, the reliance on imported isotopically labeled compounds has created significant bottlenecks in regulatory testing laboratories, driving up costs and extending lead times for crucial residue analysis. This patent outlines a robust, four-step chemical synthesis that utilizes cost-effective starting materials to achieve isotopic abundance and chemical purity exceeding 98 percent. By shifting the production paradigm from complex, low-yield imports to a streamlined, scalable domestic process, this technology offers a strategic advantage for reliable analytical standard suppliers aiming to support the agrochemical and pharmaceutical testing sectors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for acquiring stable isotope-labeled reference materials have long been plagued by exorbitant costs and supply chain fragility, primarily because these compounds were almost exclusively sourced from specialized international vendors. The conventional synthesis pathways often involve multiple protection and deprotection steps, requiring expensive deuterated reagents and sophisticated catalytic systems that are difficult to scale industrially. Furthermore, the lack of transparent process data in existing literature meant that procurement managers faced unpredictable pricing and inconsistent batch-to-batch quality, which is unacceptable for quantitative mass spectrometry applications. The absence of a viable domestic synthesis route forced regulatory bodies and third-party testing labs to endure long lead times, creating a vulnerability in the food safety monitoring infrastructure that this new patent aims to resolve effectively.
The Novel Approach
The innovative strategy detailed in the patent leverages a concise four-step sequence that dramatically simplifies the molecular construction of the target labeled compound. By starting with inexpensive commodity chemicals such as p-nitrobenzaldehyde and p-hydroxyacetophenone, the process bypasses the need for exotic precursors while maintaining high stereochemical control. The core of this novelty lies in the strategic application of a Mitsunobu reaction using deuterated methanol, which efficiently installs the stable isotope label at a late stage, minimizing isotopic dilution. This approach not only streamlines the workflow but also ensures that the final product retains high isotopic purity, making it ideal for use as an internal standard in LC-MS/MS detection. 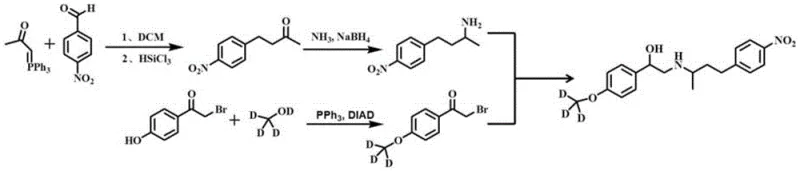
Mechanistic Insights into the Four-Step Synthesis Strategy
The synthesis begins with the reaction of p-nitrobenzaldehyde with acetyl triphenylphosphine, followed by a reduction using trichlorosilane to generate the key ketone intermediate known as Formula 1. This initial transformation is critical as it establishes the carbon backbone required for the subsequent amination step, proceeding under mild thermal conditions of 50-70°C to ensure high conversion rates without degrading the nitro group. Following this, the ketone undergoes a reductive amination reaction in the presence of ammonia and a borohydride reducing agent to yield the amine intermediate, Formula 2. This step is meticulously controlled at low temperatures, typically between 0-10°C during reduction, to prevent over-reduction or side reactions, thereby securing the integrity of the amine functionality needed for the final coupling.
The introduction of the stable isotope label occurs in the third step via a Mitsunobu reaction between bromo-p-hydroxyacetophenone and deuterated methanol, producing the deuterium-labeled intermediate shown in Formula 3. 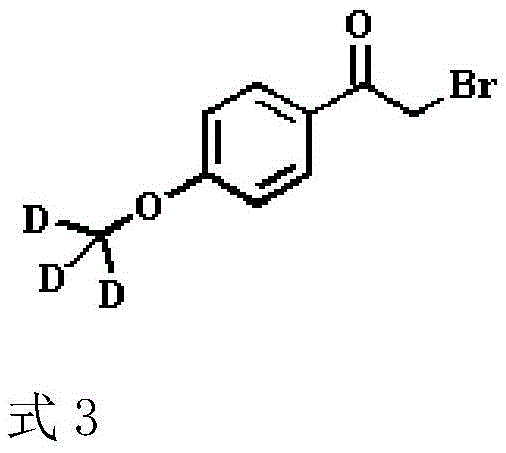 This specific transformation is pivotal for cost reduction in analytical standard manufacturing as it utilizes deuterated methanol, a relatively affordable labeling reagent, to achieve high isotopic incorporation efficiently. The final step involves a nucleophilic reaction between the amine (Formula 2) and the labeled ketone (Formula 3) in methanol, followed immediately by a hydroboration reduction. This telescoped operation eliminates the need for intermediate isolation, reducing solvent waste and processing time while delivering the final Phenylethanolamine A labeled product with exceptional purity profiles suitable for rigorous regulatory compliance.
This specific transformation is pivotal for cost reduction in analytical standard manufacturing as it utilizes deuterated methanol, a relatively affordable labeling reagent, to achieve high isotopic incorporation efficiently. The final step involves a nucleophilic reaction between the amine (Formula 2) and the labeled ketone (Formula 3) in methanol, followed immediately by a hydroboration reduction. This telescoped operation eliminates the need for intermediate isolation, reducing solvent waste and processing time while delivering the final Phenylethanolamine A labeled product with exceptional purity profiles suitable for rigorous regulatory compliance.
How to Synthesize Stable Isotope Phenylethanolamine A Efficiently
Executing this synthesis requires precise control over reaction stoichiometry and temperature profiles to maximize yield and isotopic retention. The process is designed to be operationally simple, avoiding the need for high-pressure equipment or cryogenic conditions that typically hinder scale-up. Detailed standardized operating procedures, including specific molar ratios for the Mitsunobu reagents and quenching protocols for the hydroboration step, are essential for reproducibility. For laboratory and pilot-scale teams looking to implement this pathway, the following guide outlines the critical operational parameters derived directly from the patent examples to ensure successful production.
- React p-nitrobenzaldehyde with acetyl triphenylphosphine and reduce with trichlorosilane to obtain the ketone intermediate.
- Perform reductive amination on the ketone intermediate using ammonia and a borohydride reducing agent to form the amine.
- Execute a Mitsunobu reaction between bromo-p-hydroxyacetophenone and deuterated methanol to install the stable isotope label.
- Conduct a nucleophilic reaction between the amine and labeled ketone intermediates, followed by hydroboration reduction to finalize the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route represents a significant opportunity to optimize spending and secure the supply of critical testing materials. The shift from importing finished labeled standards to manufacturing them domestically using commodity raw materials fundamentally alters the cost structure of the supply chain. By eliminating the markup associated with specialized international distributors and reducing the complexity of the synthesis, organizations can achieve substantial cost savings without compromising on the quality required for regulatory submissions. This localization of production also mitigates the risks associated with geopolitical trade disruptions, ensuring a steady flow of materials for continuous monitoring programs.
- Cost Reduction in Manufacturing: The utilization of p-nitrobenzaldehyde and p-hydroxyacetophenone as starting materials drastically lowers the raw material entry cost compared to proprietary labeled precursors. Furthermore, the elimination of transition metal catalysts and the use of common reducing agents like sodium borohydride simplify the purification process, removing the need for expensive heavy metal scavenging resins. This streamlined chemistry translates directly into lower operational expenditures and a more competitive pricing model for the final analytical standard.
- Enhanced Supply Chain Reliability: Dependence on single-source foreign suppliers for isotopically labeled compounds has historically created vulnerabilities in the testing supply chain. By establishing a domestic synthesis capability based on widely available industrial chemicals, companies can significantly reduce lead times and buffer against international logistics delays. This increased autonomy ensures that testing laboratories maintain uninterrupted access to the internal standards necessary for daily operations, safeguarding the continuity of food safety monitoring efforts.
- Scalability and Environmental Compliance: The four-step process is inherently scalable, moving seamlessly from gram-scale laboratory synthesis to multi-kilogram commercial production without requiring specialized reactor configurations. Additionally, the avoidance of hazardous solvents and the ability to recycle certain reagents align with modern green chemistry principles, reducing the environmental footprint of the manufacturing process. This compliance with environmental regulations facilitates smoother permitting and operation within strict industrial zones, further stabilizing the long-term supply outlook.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived from the specific experimental data and beneficial effects reported in the patent documentation, providing clarity for technical decision-makers. Understanding these nuances is crucial for evaluating the feasibility of adopting this route for in-house production or for qualifying new suppliers.
Q: Why is stable isotope labeled Phenylethanolamine A essential for regulatory testing?
A: It serves as a critical internal standard for Liquid Chromatography-Tandem Mass Spectrometry (LC-MS/MS), ensuring accurate quantification of veterinary drug residues in food products.
Q: What purity levels does the patented synthesis method achieve?
A: The described four-step process consistently yields the target compound with a chemical purity exceeding 98% and an isotopic abundance greater than 98%, meeting rigorous analytical requirements.
Q: How does this synthetic route impact supply chain stability?
A: By utilizing inexpensive and readily available raw materials like p-nitrobenzaldehyde, the method eliminates dependence on expensive imported standards, significantly enhancing supply security.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phenylethanolamine A Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global pharmaceutical and agrochemical testing landscape. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that complex synthetic routes like the one described in CN111777518A can be translated into robust manufacturing processes. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize advanced analytical techniques to verify isotopic abundance and chemical identity. Our capability to handle sensitive deuterated chemistry ensures that every batch meets the exacting standards required for reference material production.
We invite procurement leaders and R&D directors to collaborate with us to leverage this innovative technology for their supply chains. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that enhance both the quality and efficiency of your analytical operations.
