Advanced Manufacturing of CDK Inhibitor Intermediates via Novel Palladium Catalysis
Advanced Manufacturing of CDK Inhibitor Intermediates via Novel Palladium Catalysis
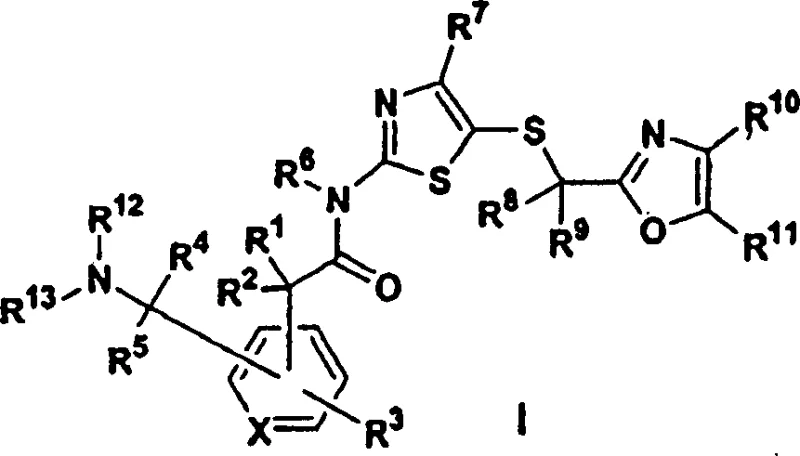
The pharmaceutical industry continuously seeks robust synthetic pathways for complex heterocyclic scaffolds that serve as critical building blocks for next-generation therapeutics. Patent CN1247573C discloses a groundbreaking methodology for the preparation of 5-(2-oxazolylalkylthio)-2-arylacetylaminothiazole compounds, which function as potent inhibitors of cyclin-dependent kinases (cdks). These kinase inhibitors represent a vital class of oncology agents capable of arresting cell cycle progression in malignant cells. The core innovation lies not merely in the final biological activity but in the revolutionary chemical strategy employed to construct the arylacetyl backbone. By reimagining the synthesis of formylarylacetic acids, this technology addresses long-standing bottlenecks in producing high-purity pharmaceutical intermediates. As a reliable pharmaceutical intermediate supplier, understanding these mechanistic shifts is crucial for ensuring the consistent quality required for clinical and commercial drug substance manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
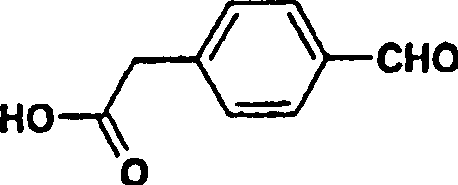
Historically, the synthesis of key precursors like 4-formylphenylacetic acid has been plagued by inefficiency and poor atom economy. Prior art literature, such as the work by J.W. Baker et al., describes a cumbersome four-step sequence starting from ethyl phenylacetate to achieve the desired formyl functionality. This legacy approach suffers from a dismal overall yield of less than 15%, rendering it economically unviable for large-scale commercial production. Furthermore, multi-step syntheses inherently accumulate impurities at each stage, necessitating rigorous and costly purification protocols to meet the stringent purity specifications demanded by regulatory bodies. The reliance on such low-yielding pathways creates significant supply chain vulnerabilities, where minor deviations in early steps can lead to catastrophic losses in final output. For procurement managers, this translates to volatile pricing and unreliable availability of critical raw materials needed for API manufacturing.
The Novel Approach
In stark contrast, the methodology outlined in the present invention streamlines the production of these vital intermediates into a highly efficient two-step process. By leveraging modern transition metal catalysis, specifically palladium-mediated cross-coupling, the new route directly constructs the carbon-carbon bond necessary for the side chain extension. This is immediately followed by a selective oxidative cleavage to unveil the aldehyde functionality. The reduction from four steps to two represents a paradigm shift in process chemistry, eliminating unnecessary unit operations and solvent exchanges. This consolidation not only boosts the overall yield significantly but also simplifies the operational complexity of the manufacturing plant. For supply chain heads, this simplified workflow means reduced lead times and enhanced capacity to scale up production from kilogram to metric ton quantities without compromising on the integrity of the molecular architecture.
Mechanistic Insights into Palladium-Catalyzed Heck Coupling and Oxidative Cleavage
The heart of this technological advancement is the sophisticated application of the Heck reaction coupled with Lemieux-Johnson oxidation. The process initiates with the reaction of a haloaryl acetic acid or ester with an alkene, such as ethyl acrylate, in the presence of a palladium catalyst like palladium diacetate. This catalytic cycle involves the oxidative addition of the aryl halide to the palladium center, followed by migratory insertion of the alkene and beta-hydride elimination to forge the vinyl-substituted aryl acetate. The choice of ligands, such as triphenylphosphine, and solvents like dimethylformamide (DMF), is critical for stabilizing the active catalytic species and ensuring high turnover numbers. Following this C-C bond formation, the vinyl group undergoes oxidative cleavage using osmium tetroxide and sodium periodate. This transformation selectively breaks the carbon-carbon double bond to generate the formyl group while preserving the sensitive acetic acid moiety, a feat difficult to achieve with harsher oxidants.
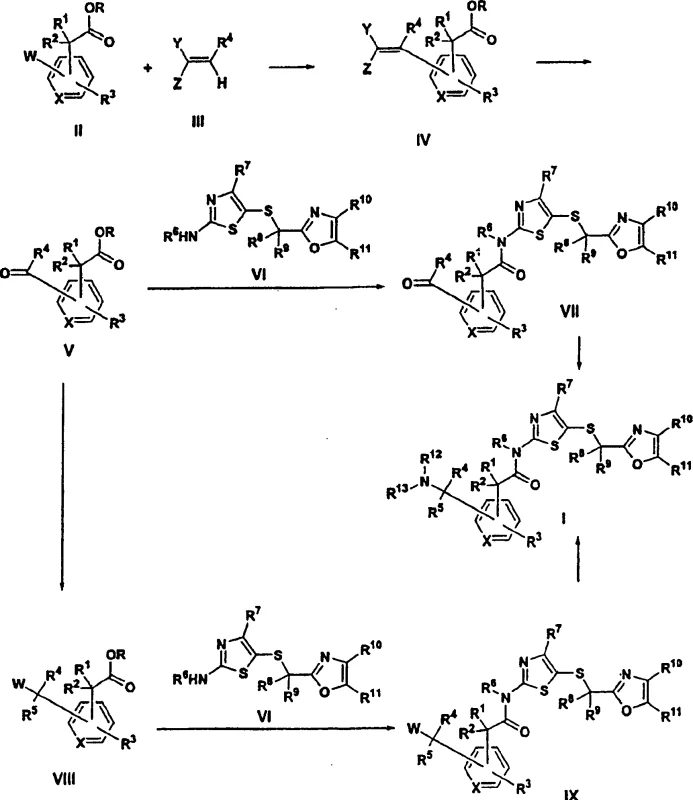
Controlling the impurity profile during these catalytic steps is paramount for downstream processing. The use of specific oxidants like sodium periodate in conjunction with catalytic osmium tetroxide allows for mild reaction conditions that minimize over-oxidation to carboxylic acids or degradation of the thiazole ring in later stages. The patent details the careful optimization of stoichiometry and temperature to suppress side reactions such as polymerization of the acrylate or homocoupling of the aryl halide. By maintaining strict control over the reaction parameters, manufacturers can ensure that the resulting formylarylacetic acid possesses the high chemical purity required for subsequent amide coupling. This level of mechanistic control is what distinguishes a laboratory curiosity from a commercially viable industrial process, providing R&D directors with the confidence to transfer this technology to pilot and production scales.
How to Synthesize 5-(2-oxazolylalkylthio)-2-arylacetylaminothiazoles Efficiently
Executing this synthesis requires precise adherence to the optimized conditions described in the patent examples to maximize yield and purity. The process begins with the preparation of the vinyl intermediate, typically achieved by heating the haloaryl acid with acrylate and palladium catalyst at elevated temperatures around 100°C. Once the vinyl species is isolated, it is subjected to oxidative cleavage in a biphasic solvent system, often involving dioxane and water, to generate the aldehyde. This aldehyde is then activated, usually via conversion to an acid chloride or using carbodiimide coupling reagents, before reacting with the aminothiazole component. The final step involves reductive amination or aminolysis to install the desired amine side chain, completing the assembly of the CDK inhibitor scaffold.
- Perform a palladium-catalyzed Heck coupling reaction between a haloaryl acetic acid derivative and an alkene such as ethyl acrylate to generate a vinyl-substituted intermediate.
- Execute oxidative cleavage of the vinyl double bond using osmium tetroxide and sodium periodate to efficiently yield the critical formylarylacetic acid intermediate.
- Couple the resulting aldehyde-acid with a 5-(2-oxazolylalkylthio)-2-aminothiazole derivative using standard peptide coupling reagents, followed by reductive amination.
Commercial Advantages for Procurement and Supply Chain Teams
The adoption of this novel synthetic route offers profound economic and logistical benefits for organizations involved in the manufacture of oncology therapeutics. By fundamentally altering the way key intermediates are constructed, the process eliminates the inefficiencies associated with traditional multi-step sequences. This leads to a drastic reduction in raw material consumption and waste generation, aligning with green chemistry principles while simultaneously lowering the cost of goods sold. For procurement teams, the ability to source these complex intermediates from a supplier utilizing this high-efficiency route ensures a more stable pricing structure and mitigates the risk of supply disruptions caused by low-yielding batch failures.
- Cost Reduction in Manufacturing: The transition from a four-step sequence with less than 15% yield to a two-step process with significantly higher yields directly impacts the bottom line. Eliminating two entire synthetic steps removes the associated costs of reagents, solvents, energy, and labor for those specific transformations. Furthermore, the improved yield means that less starting material is required to produce the same amount of final product, effectively amplifying the purchasing power of raw material budgets. This structural cost advantage allows for substantial savings in API manufacturing without compromising on quality standards.
- Enhanced Supply Chain Reliability: Simplified synthetic routes are inherently more robust and easier to scale. Reducing the number of isolation and purification steps decreases the probability of batch rejection due to out-of-specification impurities. This reliability is critical for maintaining continuous supply lines for clinical trials and commercial launches. Suppliers employing this technology can offer shorter lead times because the production cycle is compressed, allowing for faster response to market demand fluctuations and emergency orders from pharmaceutical partners.
- Scalability and Environmental Compliance: The use of established catalytic technologies like palladium coupling and osmium oxidation facilitates seamless scale-up from laboratory to commercial production. These reactions are well-understood in industrial settings, reducing the technical risk associated with process validation. Additionally, the reduction in step count inherently lowers the volume of chemical waste generated per kilogram of product. This supports environmental compliance goals and reduces the burden on waste treatment facilities, making the manufacturing process more sustainable and socially responsible.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of these arylacetyl aminothiazole derivatives. The answers are derived directly from the experimental data and claims presented in the patent documentation, ensuring accuracy and relevance for technical decision-makers. Understanding these details helps stakeholders evaluate the feasibility of integrating these intermediates into their own drug development pipelines.
Q: How does the new process improve yield compared to conventional methods?
A: Conventional methods for preparing 4-formylphenylacetic acid involved four steps with an overall yield of less than 15%. The novel process described in patent CN1247573C reduces this to just two steps with significantly higher yields, drastically improving material efficiency.
Q: What catalysts are essential for this synthesis?
A: The synthesis relies on a palladium catalyst system, preferably palladium diacetate with triphenylphosphine ligands, for the initial coupling. The subsequent oxidation step utilizes catalytic osmium tetroxide with sodium periodate as the stoichiometric oxidant.
Q: What are the primary therapeutic applications of these compounds?
A: Compounds of Formula I are potent inhibitors of cyclin-dependent kinases (cdks). They are indicated for the treatment of proliferative diseases including various cancers such as bladder, breast, colon, kidney, liver, and lung cancers, as well as inflammatory and autoimmune conditions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Arylacetyl Aminothiazole Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis and contract manufacturing, possessing the technical expertise to replicate and optimize the advanced pathways described in patent CN1247573C. Our facility is equipped with state-of-the-art reactors and analytical instrumentation capable of handling sensitive palladium-catalyzed reactions and oxidative transformations safely and efficiently. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can grow seamlessly from preclinical research to global market supply. Our commitment to quality is underscored by our stringent purity specifications and rigorous QC labs, which verify every batch against the highest industry standards.
We invite pharmaceutical companies and research institutions to collaborate with us to leverage this cutting-edge technology for their CDK inhibitor programs. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our manufacturing capabilities can accelerate your drug development timeline while optimizing your overall budget.
