Advanced Manufacturing of Atorvastatin Side Chain Intermediates via Optimized Nitro-Reduction Pathways
Advanced Manufacturing of Atorvastatin Side Chain Intermediates via Optimized Nitro-Reduction Pathways
The global demand for high-purity statin intermediates continues to surge as cardiovascular therapies remain a cornerstone of modern medicine. Patent CN102391243A introduces a transformative approach to synthesizing the critical Atorvastatin side chain intermediate, specifically (3R,5S)-7-amino-3,5-O-isopropylidene-3,5-dihydroxyheptanoic acid tert-butyl ester. This technical breakthrough addresses long-standing inefficiencies in chiral synthesis by replacing hazardous lithiation steps with a robust zinc-mediated Reformatsky-type coupling. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediates supplier, this methodology offers a compelling alternative to legacy processes that rely on cryogenic conditions and expensive noble metals. By leveraging (S)-epichlorohydrin as a chiral pool starting material, the process ensures stereochemical integrity while drastically simplifying the operational workflow. The following analysis dissects the mechanistic advantages and commercial viability of this seven-step sequence, highlighting its potential to redefine cost structures in lipid-lowering drug manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Atorvastatin side chains has been plagued by complex multi-step sequences that introduce significant supply chain vulnerabilities. Traditional routes, such as those described in prior art patents like CN 200910061164.4A, often necessitate an eight-step synthesis starting from epichlorohydrin but requiring a chiral resolution step using Salen(Co) catalysts. More critically, established methodologies frequently depend on the preparation of (R)-(-)-4-cyano-3-hydroxybutyrate ethyl ester, an intermediate known for its low synthetic efficiency and difficult purification profiles. Furthermore, conventional pathways often employ noble metal lithium compounds for carbon-carbon bond formation, which not only inflates raw material costs but also mandates stringent safety protocols due to the pyrophoric nature of organolithium reagents. The requirement for ultra-low temperatures, specifically around -80°C during lithiation steps, imposes a heavy energy burden on manufacturing facilities and limits the scalability of batch reactors. These factors collectively result in a fragmented production landscape where yield losses accumulate at every stage, ultimately driving up the cost of goods sold for the final API.
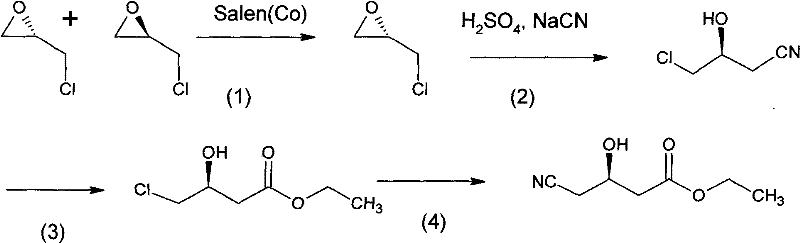
The Novel Approach
In stark contrast, the methodology outlined in patent CN102391243A streamlines the synthesis into a more direct and economically viable seven-step sequence that bypasses the problematic cyano-ester intermediate entirely. By initiating the synthesis with a direct ring-opening of (S)-epichlorohydrin using nitromethane, the process establishes the necessary carbon backbone while preserving the chiral center without the need for expensive enzymatic or metal-complex resolutions. The substitution of lithium reagents with activated zinc powder for the key coupling reaction represents a paradigm shift in process safety and cost management. Zinc is abundant, inexpensive, and far easier to handle on a multi-ton scale compared to pyrophoric organometallics. Additionally, the reaction conditions are significantly milder, with most transformations occurring between 0°C and 50°C, thereby eliminating the need for energy-intensive cryogenic cooling systems. This novel approach not only enhances the overall yield to a range of 80% to 86% but also achieves a final purity exceeding 99.0%, demonstrating that efficiency and quality can be simultaneously optimized through intelligent route design.
Mechanistic Insights into Zinc-Mediated Reformatsky Coupling and Nitro Reduction
The core innovation of this synthetic route lies in the strategic application of a Reformatsky-type reaction coupled with a nitro-group manipulation strategy. The process begins with the Lewis acid-catalyzed ring opening of (S)-epichlorohydrin by nitromethane, utilizing catalysts such as boron trifluoride or hydrogen chloride to generate 1-chloro-4-nitro-(S)-2-butanol with high regioselectivity. This intermediate is subsequently subjected to nucleophilic substitution with sodium cyanide in an ethanol-water mixture, converting the chloro-group into a nitrile while maintaining the stereochemical configuration at the hydroxyl-bearing carbon. The pivotal step involves the generation of a zinc enolate from tert-butyl bromoacetate, which then attacks the nitrile-containing aldehyde precursor. Unlike lithiation, which requires strict anhydrous conditions and inert atmospheres, the zinc-mediated coupling is more tolerant and proceeds efficiently at moderate temperatures (20-50°C). Following the coupling, the ketone functionality is selectively reduced using sodium borohydride to establish the second hydroxyl group required for the statin pharmacophore.
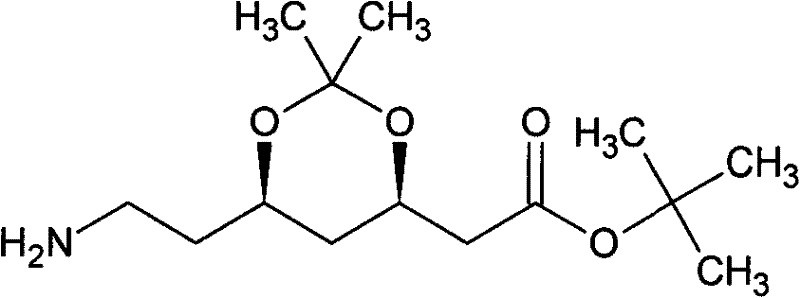
Impurity control is rigorously managed throughout the sequence, particularly during the protection and reduction phases. The diol intermediate is protected as an acetonide using 2,2-dimethoxypropane and methanesulfonic acid, a reaction that effectively masks the 3,5-dihydroxy motif against unwanted side reactions during the final reduction. The terminal nitro group is then converted to the primary amine via catalytic hydrogenation using Raney Nickel under 0.6 MPa of hydrogen pressure. This heterogeneous catalysis method is superior to homogeneous reductions as it allows for easy filtration and recovery of the catalyst, minimizing metal contamination in the final product. The use of Raney Nickel also avoids the generation of stoichiometric metal waste associated with iron or zinc dust reductions. By carefully controlling the pH during workup and employing vacuum distillation for purification, the process ensures that trace impurities such as unreacted nitro compounds or over-reduced amines are kept below detectable limits, satisfying the stringent specifications required for GMP-grade pharmaceutical intermediates.
How to Synthesize Atorvastatin Intermediate Efficiently
Implementing this synthesis requires precise control over reaction parameters, particularly during the exothermic cyanation and the moisture-sensitive zinc activation steps. The patent provides detailed embodiments illustrating that the ring-opening reaction should be maintained between 20°C and 50°C to balance reaction rate and selectivity. For the subsequent cyanation, temperature control is critical; the addition of sodium cyanide must be managed to prevent thermal runaway, with optimal results observed when the reaction mass is kept between 15°C and 50°C depending on the specific embodiment. The zinc reagent preparation demands activated zinc powder and anhydrous tetrahydrofuran to ensure successful enolate formation. Detailed standardized synthesis steps see the guide below.
- Ring-opening of (S)-epichlorohydrin with nitromethane using BF3 or HCl catalyst to form 1-chloro-4-nitro-(S)-2-butanol.
- Cyanation reaction with sodium cyanide in ethanol/water to yield (S)-3-hydroxy-5-nitro-valeronitrile.
- Reformatsky-type coupling with tert-butyl bromoacetate and zinc powder, followed by borohydride reduction and acetonide protection.
- Final catalytic hydrogenation using Raney Nickel to reduce the nitro group to the primary amine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this optimized synthetic route offers substantial strategic benefits beyond mere technical elegance. The elimination of noble metal lithium compounds directly translates to a significant reduction in raw material expenditure, as zinc powder is a commodity chemical with a stable and low market price compared to specialized organolithium reagents. Furthermore, the removal of ultra-low temperature requirements (-80°C) drastically lowers the energy consumption profile of the manufacturing process, allowing for the use of standard chilled water systems rather than specialized cryogenic brine loops. This simplification of utility requirements enhances the flexibility of production scheduling and reduces the capital expenditure needed for reactor infrastructure. The robustness of the process also implies fewer batch failures and less downtime, ensuring a more consistent flow of materials to downstream API synthesis units.
- Cost Reduction in Manufacturing: The replacement of expensive chiral resolution catalysts and pyrophoric lithium reagents with abundant zinc and simple acids creates a fundamentally lower cost base for production. By avoiding the synthesis of the inefficient (R)-(-)-4-cyano-3-hydroxybutyrate ethyl ester intermediate, the process reduces the number of unit operations, which in turn minimizes solvent usage, labor hours, and waste disposal costs. The high overall yield of 80% to 86% means that less starting material is required to produce the same amount of final product, effectively amplifying the purchasing power of raw material budgets. Additionally, the use of Raney Nickel, a reusable heterogeneous catalyst, further drives down the cost per kilogram compared to stoichiometric reducing agents that generate large volumes of solid waste.
- Enhanced Supply Chain Reliability: Relying on commodity chemicals like zinc powder, sodium cyanide, and tert-butyl bromoacetate mitigates the risk of supply disruptions often associated with specialty chiral catalysts or sensitive organometallics. These raw materials are widely available from multiple global suppliers, ensuring that production schedules are not held hostage by single-source bottlenecks. The simplified process flow, which avoids complex low-temperature logistics, also reduces the risk of shipping delays or storage issues for temperature-sensitive intermediates. Consequently, manufacturers can maintain higher safety stocks and respond more agilely to fluctuations in market demand for Atorvastatin APIs, providing a competitive edge in a volume-driven market.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing reaction conditions that are easily replicated in large-scale stainless steel reactors without the need for exotic materials of construction. The avoidance of halogenated solvents in certain steps and the ability to recover and recycle solvents like tetrahydrofuran and ethyl acetate align with modern green chemistry principles. Moreover, the heterogeneous nature of the final hydrogenation step simplifies wastewater treatment by reducing the load of dissolved heavy metals. This environmental compatibility facilitates smoother regulatory approvals and reduces the long-term liability associated with hazardous waste management, making the facility more sustainable and compliant with increasingly strict environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in patent CN102391243A, offering clarity on how this method outperforms legacy technologies in terms of safety, yield, and operational simplicity.
Q: How does this new process improve upon traditional lithiation methods?
A: The patented method eliminates the need for expensive and hazardous noble metal lithium compounds and avoids ultra-low temperature requirements (-80°C) typically associated with lithiation, significantly enhancing operational safety and reducing energy costs.
Q: What is the expected purity of the final Atorvastatin intermediate?
A: According to the patent data, the optimized synthesis route consistently achieves a final product purity of greater than or equal to 99.0%, with overall yields ranging between 80% and 86%.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process utilizes common reagents like zinc powder and Raney Nickel instead of sensitive organolithiums, and operates at moderate temperatures (0-50°C for most steps), making it highly scalable for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Atorvastatin Intermediate Supplier
As the pharmaceutical industry continues to demand higher purity and lower costs for life-saving medications, NINGBO INNO PHARMCHEM stands ready to leverage this advanced synthetic technology for your supply chain. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale optimization to full-scale manufacturing is seamless and efficient. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of (3R,5S)-7-amino-3,5-O-isopropylidene-3,5-dihydroxyheptanoic acid tert-butyl ester meets the highest global standards. Our commitment to technical excellence means we can adapt this zinc-mediated route to fit your specific capacity requirements while maintaining the cost and safety advantages inherent to the process.
We invite you to engage with our technical procurement team to discuss how this optimized route can impact your bottom line. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the potential economic benefits specific to your operation. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions about securing a stable, high-quality supply of this critical cardiovascular intermediate.
