Revolutionizing Fragrance Intermediates: Scalable One-Pot Synthesis of Cyclohexyloxy Allyl Acetate
The landscape of fine chemical manufacturing is constantly evolving, driven by the need for more efficient, sustainable, and cost-effective synthetic routes. A pivotal advancement in this domain is documented in patent CN107915627B, which discloses a novel one-pot method for synthesizing cyclohexyloxy allyl acetate, also known industrially as cyclic galbanum ester. This compound is highly valued in the fragrance industry for its intense, persistent green pineapple and apple notes, serving as an excellent fixative. The traditional synthesis pathways have long been plagued by multi-step complexities, but this new methodology streamlines the production into a continuous, single-vessel operation. By leveraging sequential nucleophilic substitutions in an anhydrous toluene medium, the process achieves yields exceeding 90 percent while maintaining exceptional selectivity. For global procurement leaders and R&D directors, understanding this technological shift is crucial for securing a reliable supply chain of high-purity fragrance intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of cyclohexyloxy allyl acetate has relied on a disjointed two-step sequence that poses significant logistical and economic challenges for manufacturers. The conventional route typically involves the initial condensation of cyclohexanol with chloroacetic acid to generate an epoxy acetic acid intermediate, which must then be isolated and purified before proceeding. Subsequently, this intermediate undergoes a dehydration and condensation reaction with allyl alcohol to yield the final ester. This fragmented approach not only necessitates multiple reactor setups and extensive downstream processing but also introduces opportunities for yield loss at each isolation stage. Furthermore, the handling of unstable intermediates and the requirement for distinct reaction conditions for each step complicate process control and increase the risk of impurity accumulation. These inefficiencies translate directly into higher operational expenditures and longer lead times, making the traditional method less competitive in a market demanding rapid turnaround and cost optimization.
The Novel Approach
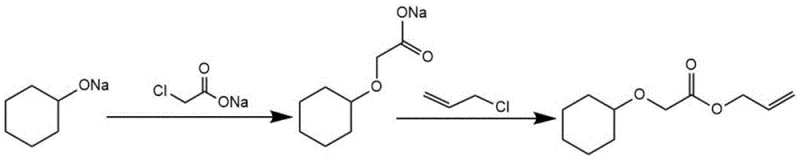
In stark contrast, the methodology outlined in patent CN107915627B introduces a sophisticated one-pot strategy that fundamentally redefines the synthesis workflow. By utilizing sodium salts of the starting materials—specifically sodium cyclohexanolate and sodium chloroacetate—the process enables a seamless tandem reaction within a single reactor vessel containing anhydrous toluene. The introduction of a phase transfer catalyst, such as tetrabutylammonium bromide (TBAB), facilitates the interaction between the ionic salts and the organic phase, driving the reaction forward with remarkable efficiency. Following the initial etherification, allyl chloride is introduced directly into the same mixture to effect esterification without the need for intermediate workup. This continuity not only simplifies the operational protocol but also drastically reduces solvent usage and waste generation. The result is a robust manufacturing process capable of delivering high-purity products with minimal byproduct formation, addressing the critical pain points of the legacy technology.
Mechanistic Insights into One-Pot Tandem Nucleophilic Substitution
The chemical elegance of this synthesis lies in its exploitation of sequential SN2 nucleophilic substitution mechanisms under carefully controlled thermal conditions. Initially, the sodium cyclohexanolate acts as a potent nucleophile, attacking the electrophilic carbon of the sodium chloroacetate to form the sodium cyclohexyloxy acetate intermediate. This step is critical and is optimized by refluxing in anhydrous toluene, which ensures the removal of any trace moisture that could hydrolyze the reactive species. The presence of the phase transfer catalyst is instrumental here, as it shuttles the anionic nucleophiles into the organic phase, enhancing reaction kinetics and ensuring homogeneity. Once this ether linkage is established, the carboxylate anion of the intermediate remains nucleophilic and ready for the second transformation.
Upon the addition of allyl chloride (chloropropene), the carboxylate oxygen attacks the allylic carbon, displacing the chloride ion to form the final ester bond. This second substitution occurs smoothly at temperatures ranging from 70 to 100 degrees Celsius, completing the molecular assembly in a single pot. The mechanistic pathway minimizes side reactions because the reaction environment is strictly anhydrous and the stoichiometry is tightly controlled, with molar ratios of sodium cyclohexanolate to chloropropene maintained between 1:1.2 and 1:2. This precise control over the reaction trajectory ensures that the impurity profile remains exceptionally clean, facilitating easier purification and resulting in a final product that can be refined to purities as high as 99.8 percent.
How to Synthesize Cyclohexyloxy Allyl Acetate Efficiently
Implementing this patented process requires adherence to specific preparatory and reaction protocols to maximize yield and safety. The synthesis begins with the preparation of the necessary sodium salts, either by reacting cyclohexanol with sodium metal or neutralizing chloroacetic acid with sodium hydroxide, followed by drying to a powder. These salts are then combined in anhydrous toluene with a catalytic amount of a quaternary ammonium salt. The mixture is heated under reflux until the first substitution is complete, monitored by the disappearance of starting materials. Subsequently, a solution of allyl chloride in toluene is added dropwise, and the reflux is continued to drive the esterification to completion. The detailed standardized synthetic steps, including specific workup procedures like suction filtration, aqueous extraction, and distillation, are outlined below.
- Prepare sodium cyclohexanolate and sodium chloroacetate salts separately or in situ using sodium metal and sodium hydroxide respectively.
- Reflux the sodium salts in anhydrous toluene with a phase transfer catalyst like TBAB to form the intermediate sodium cyclohexyloxy acetate.
- Add allyl chloride (chloropropene) solution to the mixture, maintain reflux to complete the esterification, then purify via extraction and distillation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this one-pot synthesis technology represents a strategic opportunity to optimize the sourcing of fragrance intermediates. The consolidation of two distinct chemical transformations into a single continuous process inherently reduces the manufacturing footprint and the associated utility costs. By eliminating the isolation and purification of the intermediate epoxy acetic acid, the process removes entire unit operations from the production schedule, thereby reducing labor intensity and equipment occupancy time. This streamlining translates into substantial cost savings in manufacturing, not through arbitrary percentage claims, but through the tangible reduction of solvent volumes, energy consumption for heating and cooling cycles, and waste disposal fees associated with intermediate handling.
- Cost Reduction in Manufacturing: The elimination of intermediate isolation steps significantly lowers the operational expenditure required for producing cyclohexyloxy allyl acetate. By avoiding the need for separate reactors, filtration units, and drying ovens for the intermediate, the capital intensity of the production line is reduced. Furthermore, the high atom economy of the tandem reaction ensures that raw materials are converted efficiently into the desired product, minimizing the cost of goods sold. The use of commodity chemicals like cyclohexanol, chloroacetic acid, and allyl chloride ensures that input costs remain stable and predictable, shielding the supply chain from volatility associated with exotic reagents.
- Enhanced Supply Chain Reliability: The simplicity and robustness of the one-pot method enhance the reliability of supply by reducing the number of potential failure points in the manufacturing process. Traditional multi-step syntheses are prone to delays if an intermediate fails quality control or if equipment bottlenecks occur between steps. In contrast, the continuous nature of this patented process allows for faster batch turnover and more consistent output. The ability to scale this reaction from laboratory benchtop to multi-kilogram pilots, as demonstrated in the patent examples, confirms its viability for meeting large-volume demands without compromising on delivery schedules or product consistency.
- Scalability and Environmental Compliance: From an environmental and regulatory standpoint, this process offers significant advantages that align with modern green chemistry principles. The reduction in solvent usage and the minimization of waste streams simplify the effluent treatment process, lowering the environmental compliance burden on the manufacturer. The high selectivity of the reaction means fewer byproducts are generated, which reduces the complexity of downstream purification and the volume of hazardous waste requiring disposal. This eco-friendly profile not only supports corporate sustainability goals but also future-proofs the supply chain against increasingly stringent environmental regulations in the fine chemical sector.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of cyclohexyloxy allyl acetate using this advanced one-pot methodology. These insights are derived directly from the technical disclosures in patent CN107915627B and are intended to clarify the feasibility and benefits of this synthetic route for industry stakeholders. Understanding these details is essential for making informed decisions about integrating this material into your fragrance formulations or supply portfolio.
Q: What are the primary advantages of the one-pot method over traditional synthesis?
A: The one-pot method eliminates the need for isolating the intermediate epoxy acetic acid, significantly reducing operational complexity, solvent consumption, and processing time compared to the conventional two-step condensation and dehydration route.
Q: What purity levels can be achieved with this patented process?
A: The process yields crude product with purity around 96%, which can be further refined through recrystallization or distillation to achieve ultra-high purity specifications exceeding 99.5%, suitable for high-end fragrance applications.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent demonstrates successful scaling from gram scale to 10kg batches with consistent yields above 80%, indicating strong potential for commercial scale-up due to its continuous reaction nature and simple workup procedures.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cyclohexyloxy Allyl Acetate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the creation of superior fragrance profiles. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent supply regardless of demand fluctuations. Our commitment to excellence is underpinned by stringent purity specifications and rigorous QC labs that verify every batch against the highest industry standards. We are fully equipped to implement the advanced one-pot synthesis techniques described in recent patents, offering our partners access to cutting-edge manufacturing capabilities that drive efficiency and quality.
We invite you to collaborate with us to leverage these technological advancements for your specific application needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how our optimized processes can enhance your bottom line. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate why we are the preferred partner for complex fine chemical synthesis.
