Advanced Transesterification Strategy for Commercial Scale-up of Cloquintocet-Mexyl Production
Advanced Transesterification Strategy for Commercial Scale-up of Cloquintocet-Mexyl Production
The global demand for high-efficiency herbicide safeners continues to drive innovation in agrochemical intermediate synthesis, particularly for compounds like Cloquintocet-mexyl (CAS 99607-70-2). A pivotal advancement in this domain is detailed in patent CN101805288A, which introduces a novel transesterification-based methodology that fundamentally restructures the production workflow. This technical breakthrough addresses long-standing inefficiencies in traditional manufacturing by shifting the synthetic focus to a more thermodynamically favorable pathway. By leveraging the reaction between 5-chloro-8-hydroxyquinoline and methyl chloroacetate to form a stable intermediate, followed by a catalytic exchange with 2-heptanol, the process achieves superior control over impurity profiles. For R&D directors and procurement strategists, understanding this shift is critical, as it represents a move away from equilibrium-limited esterifications toward a driven, vacuum-assisted transesterification that promises both economic and operational superiority.
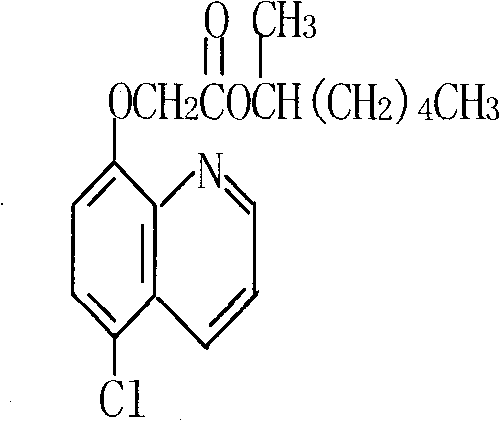
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Cloquintocet-mexyl relied heavily on a route involving the direct esterification of monochloroacetic acid with 2-heptanol (also known as 1-methylhexanol or 2-enanthol) to generate a chloroacetate intermediate. This conventional approach is fraught with significant kinetic and thermodynamic challenges that hinder large-scale efficiency. The esterification reaction between monochloroacetic acid and the secondary alcohol 2-heptanol is inherently difficult to drive to completion due to steric hindrance and equilibrium constraints, often resulting in prolonged reaction times and suboptimal conversion rates. Furthermore, the purification of this initial intermediate is notoriously difficult, requiring energy-intensive separation techniques to remove unreacted acids and alcohols. When this impure intermediate is subsequently reacted with 5-chloro-8-hydroxyquinoline, the presence of residual impurities catalyzes side reactions, leading to a complex mixture of by-products that drastically lower the overall yield and compromise the quality of the final agrochemical safener.
The Novel Approach
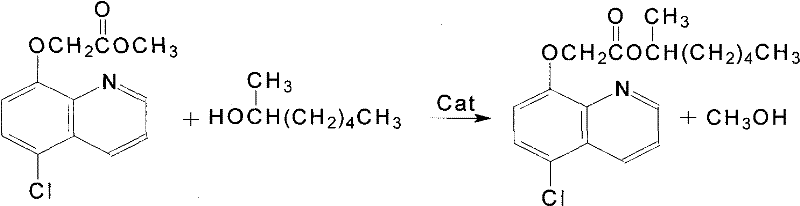
In stark contrast, the novel method disclosed in the patent data utilizes a strategic inversion of the synthetic sequence, prioritizing the formation of the quinoline-ester bond first. This approach begins with the alkylation of 5-chloro-8-hydroxyquinoline using methyl chloroacetate, a reaction that proceeds with high selectivity under basic conditions to form 5-chloro-8-quinolyloxy methyl acetate. The subsequent step involves a transesterification reaction with 2-heptanol, which is kinetically more favorable and easier to control than the direct acid-alcohol esterification of the old route. By employing a catalytic system under reduced pressure, the process effectively removes methanol as it forms, shifting the chemical equilibrium decisively toward the desired product. This not only simplifies the downstream processing but also allows for the recovery and recycling of methanol-containing waste streams, transforming a potential environmental liability into a recoverable resource while ensuring the final product meets stringent purity specifications required for modern agricultural applications.
Mechanistic Insights into Base-Catalyzed Transesterification
The core of this technological advancement lies in the precise manipulation of nucleophilic acyl substitution mechanisms during the transesterification phase. In the second step of the synthesis, the intermediate methyl ester acts as the electrophile, while 2-heptanol serves as the nucleophile. Under the influence of a catalyst—typically a mixture of organic bases or mineral alkalis—the alkoxide ion of the 2-heptanol attacks the carbonyl carbon of the acetate group. This forms a tetrahedral intermediate which subsequently collapses to expel the methoxide leaving group. The critical innovation here is the application of vacuum conditions (0.025-0.07 MPa) combined with elevated temperatures (60-120°C). This physical removal of the volatile methanol byproduct prevents the reverse reaction from occurring, effectively locking the equilibrium in the forward direction. This mechanistic control is vital for minimizing the formation of hydrolysis by-products or ether impurities that typically plague non-optimized esterification processes, thereby ensuring a cleaner reaction profile.
Impurity control is further enhanced by the specific thermal management of the first alkylation step. By controlling the addition of methyl chloroacetate at temperatures between 30-90°C after an initial azeotropic dehydration phase, the process minimizes the risk of poly-alkylation or degradation of the sensitive quinoline ring system. The use of a biphasic solvent system involving DMSO and toluene facilitates the removal of water generated during the initial salt formation, which is crucial because water can hydrolyze the chloroacetate reagent. This rigorous exclusion of moisture and precise temperature ramping ensures that the intermediate formed is of high chemical integrity before it even enters the transesterification reactor. Consequently, the final crude product requires less aggressive purification, preserving yield and reducing the load on solvent recovery systems, which is a key consideration for sustainable manufacturing.
How to Synthesize Cloquintocet-Mexyl Efficiently
The synthesis of Cloquintocet-mexyl via this novel transesterification route offers a robust framework for laboratory and pilot-scale operations aiming to transition to commercial production. The process is defined by two distinct stages: the initial construction of the quinoline-acetate scaffold and the subsequent exchange of the methyl group for the 2-heptyl chain. Operators must pay close attention to the azeotropic dehydration in the first step and the vacuum maintenance in the second step to ensure optimal kinetics. The following guide outlines the standardized operational parameters derived from the patent examples, providing a clear roadmap for replicating this high-efficiency synthesis while maintaining strict adherence to safety and quality protocols.
- Synthesize the intermediate 5-chloro-8-quinolyloxy methyl acetate by reacting 5-chloro-8-hydroxyquinoline with methyl chloroacetate in the presence of sodium hydroxide and sodium carbonate, utilizing azeotropic dehydration.
- Perform transesterification by reacting the intermediate with 2-heptanol (1-methylhexanol) using an organic or mineral base catalyst under vacuum conditions to remove methanol.
- Cool the reaction mixture to induce crystallization, followed by filtration and drying to obtain high-purity Cloquintocet-mexyl.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this transesterification technology translates directly into tangible operational improvements and risk mitigation. The shift away from the traditional monochloroacetic acid esterification route eliminates a major bottleneck characterized by slow reaction rates and difficult separations. By simplifying the synthetic pathway, manufacturers can achieve a more streamlined production schedule, reducing the overall cycle time per batch. This efficiency gain is compounded by the ability to recycle methanol-containing waste liquor, which not only lowers raw material consumption but also reduces the costs associated with waste treatment and disposal. The result is a manufacturing process that is inherently more cost-effective and environmentally compliant, aligning with the increasing regulatory pressures faced by the agrochemical industry.
- Cost Reduction in Manufacturing: The novel process significantly lowers production costs by eliminating the need for expensive and difficult purification steps associated with the traditional intermediate (1). In the conventional route, the incomplete esterification of monochloroacetic acid necessitates complex workups to remove unreacted starting materials, which consumes substantial amounts of solvents and energy. By contrast, the transesterification route drives the reaction to completion through the physical removal of methanol, resulting in a cleaner crude product that requires less refining. Furthermore, the recovery of methanol from the waste stream provides an additional economic offset, reducing the net cost of goods sold and improving the overall margin profile for the final agrochemical intermediate.
- Enhanced Supply Chain Reliability: Supply continuity is bolstered by the use of readily available and stable raw materials such as 5-chloro-8-hydroxyquinoline and methyl chloroacetate. Unlike the traditional route, which relies on the difficult coupling of a secondary alcohol with a reactive acid chloride or acid, this method utilizes robust reagents that are less prone to supply chain disruptions. The milder reaction conditions (60-120°C) also reduce the stress on reactor equipment, minimizing downtime for maintenance and repairs. This operational stability ensures that production targets can be met consistently, reducing the lead time for high-purity agrochemical intermediates and providing downstream herbicide manufacturers with a dependable source of critical safener materials.
- Scalability and Environmental Compliance: The process is explicitly designed for large-scale industrial production, featuring simple reaction controls that are easily automated in standard stainless steel reactors. The ability to operate under moderate vacuum and temperature conditions means that the process does not require specialized high-pressure or cryogenic equipment, facilitating easier scale-up from pilot to commercial tonnage. Additionally, the reduction in hazardous waste generation, specifically through the recycling of methanol streams and the avoidance of harsh acidic conditions found in older methods, enhances the environmental footprint of the facility. This alignment with green chemistry principles supports long-term sustainability goals and ensures compliance with increasingly stringent environmental regulations governing chemical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of Cloquintocet-mexyl, based on the detailed process parameters and advantages outlined in the patent literature. These insights are intended to clarify the operational benefits of the transesterification method for technical teams evaluating process feasibility. Understanding these nuances is essential for making informed decisions about technology transfer and vendor qualification in the competitive agrochemical market.
Q: What are the primary advantages of the transesterification method over traditional esterification for Cloquintocet-mexyl?
A: The transesterification method avoids the difficult and incomplete esterification of monochloroacetic acid with 2-heptanol found in traditional routes. It offers milder reaction conditions, easier process control, higher product purity with fewer impurities, and allows for the recycling of methanol-containing waste liquor, significantly reducing production costs.
Q: What are the critical reaction conditions for the transesterification step?
A: The transesterification step requires maintaining a temperature between 60-120°C and applying a vacuum of 0.025-0.07 MPa. This vacuum is crucial for continuously removing the generated methanol and partial solvent, driving the equilibrium towards the product. Catalysts such as organic bases, mineral alkalis, or their mixtures are employed to facilitate the reaction.
Q: How does this synthesis route impact supply chain reliability for agrochemical manufacturers?
A: By utilizing readily available raw materials like 5-chloro-8-hydroxyquinoline and methyl chloroacetate, and eliminating complex purification steps associated with intermediate (1) in older methods, this route ensures a more stable and continuous supply. The simplified process is highly suitable for large-scale industrial production, reducing lead times and enhancing overall supply chain resilience.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cloquintocet-Mexyl Supplier
As the agrochemical sector evolves, the need for partners who can bridge the gap between innovative patent chemistry and reliable commercial supply has never been greater. NINGBO INNO PHARMCHEM stands at the forefront of this transition, leveraging deep technical expertise to master complex synthetic pathways like the transesterification route for Cloquintocet-mexyl. Our facilities are equipped to handle the nuanced requirements of this chemistry, from precise vacuum control to efficient solvent recovery systems. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent supply of material that meets stringent purity specifications. Our rigorous QC labs employ advanced analytical techniques to verify the identity and purity of every batch, guaranteeing that the safener performance in the field matches the theoretical potential of the molecule.
We invite global agrochemical leaders to collaborate with us to optimize their supply chains and reduce manufacturing costs through superior process chemistry. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis that evaluates how our optimized synthesis routes can improve your bottom line. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments tailored to your volume requirements. Let us demonstrate how our commitment to technical excellence and operational efficiency can become a cornerstone of your strategic sourcing strategy for high-value agrochemical intermediates.
