Scalable Manufacturing of N-Coumaroyldopamine: A Breakthrough in Pharmaceutical Intermediate Synthesis
Introduction to Advanced N-Coumaroyldopamine Synthesis
The pharmaceutical industry continuously seeks robust synthetic routes for bioactive molecules that balance high purity with economic viability. A critical analysis of patent CN103044279A reveals a transformative approach to synthesizing N-Coumaroyldopamine (CAS: 103188-46-1), a compound known for its inhibitory effects on prostaglandin and leukotriene biosynthesis. Historically, the production of this valuable pharmaceutical intermediate has been hindered by inefficient methodologies that struggle to meet the rigorous demands of modern Good Manufacturing Practice (GMP). The disclosed technology addresses these bottlenecks by introducing a novel protective group strategy that fundamentally alters the reaction landscape, enabling a transition from laboratory-scale curiosity to industrial reality.
This synthesis method specifically targets the structural vulnerabilities of the dopamine backbone, utilizing selective protection to prevent unwanted side reactions during the critical amide bond formation step. By shifting away from traditional acetyl protection towards more stable silyl or halohydrocarbon groups, the process achieves a dramatic improvement in yield and purity without relying on resource-intensive purification techniques. For R&D directors and procurement specialists alike, this represents a significant opportunity to secure a reliable pharmaceutical intermediates supplier capable of delivering consistent quality. The following sections will dissect the chemical ingenuity behind this process and its profound implications for supply chain stability.
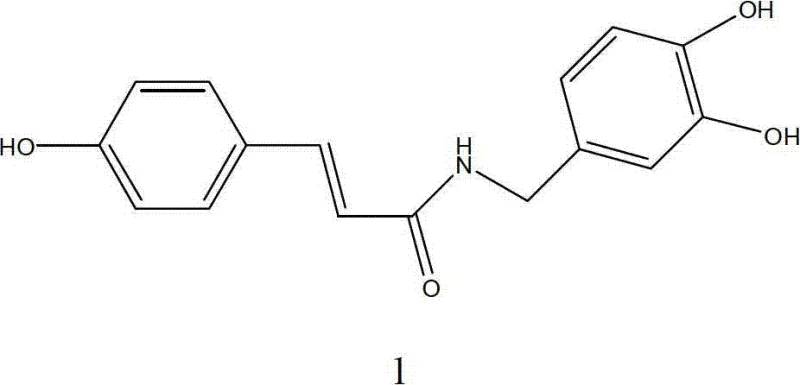
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies for synthesizing N-Coumaroyldopamine have long been plagued by inherent inefficiencies that render them unsuitable for commercial scale-up. The traditional route typically employs acetyl groups to protect the phenolic hydroxyls of dopamine, followed by coupling with 4-hydroxycinnamic acid via a mixed anhydride method. While chemically feasible on a small scale, this approach suffers from severe drawbacks when applied to larger batches. The primary issue lies in the generation of substantial by-products during the coupling phase, which complicates the isolation of the target molecule. Consequently, the crude product requires purification via column chromatography, a technique that is notoriously difficult to scale, expensive in terms of silica gel consumption, and detrimental to overall throughput.
Furthermore, the reported yield for these conventional methods hovers around a mere 37%, indicating a massive loss of valuable starting materials and a corresponding increase in the cost of goods sold (COGS). The reliance on chromatographic purification not only extends the production cycle time but also introduces variability in batch-to-batch consistency, a critical risk factor for regulatory compliance. For a procurement manager evaluating potential sources, a process dependent on such low-efficiency purification steps signals high supply risk and inflated pricing. The inability to simply recrystallize the intermediate means that the process lacks the robustness required for a reliable API intermediate supplier to guarantee continuous availability.
The Novel Approach
In stark contrast, the innovative method described in the patent data introduces a strategic shift by employing silyl or halohydrocarbon protecting groups, such as tert-butyldimethylchlorosilane (TBDMS-Cl), to mask the phenolic hydroxyls of dopamine. This modification creates a steric and electronic environment that significantly suppresses side reactions during the subsequent coupling with 4-hydroxycinnamic acid. The result is a much cleaner reaction profile where the desired N-Coumaroyldopamine derivative is formed with high selectivity. Crucially, this high selectivity allows for the purification of intermediates through simple recrystallization rather than column chromatography, drastically simplifying the downstream processing workflow.
The overall synthetic route is streamlined into three distinct phases: protection, coupling, and deprotection. The coupling step utilizes N,N'-Diisopropylcarbodiimide (DIC) to activate the carboxylic acid, facilitating a smooth amide bond formation under mild conditions. Following the coupling, the protecting groups are efficiently removed using a fluoride source, yielding the final product with exceptional purity levels exceeding 96%. This methodological overhaul directly addresses the cost reduction in pharmaceutical intermediates manufacturing by eliminating expensive purification media and reducing solvent usage. The ability to achieve high purity through crystallization is a hallmark of a mature, scalable process, making it highly attractive for commercial scale-up of complex pharmaceutical intermediates.
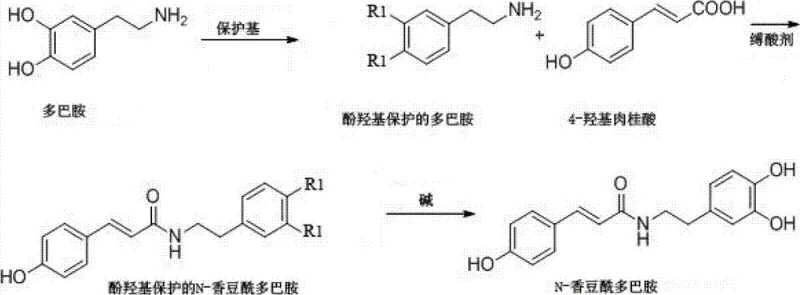
Mechanistic Insights into Silyl Protection and Carbodiimide Activation
To fully appreciate the technical superiority of this synthesis, one must examine the mechanistic details of the activation and protection steps. The core of the innovation lies in the activation of 4-hydroxycinnamic acid using DIC. Unlike traditional acid chloride formation which can be harsh and prone to racemization or degradation, the carbodiimide-mediated activation proceeds through the formation of an O-acylisourea intermediate. This species is highly reactive towards nucleophilic attack by the amine group of the protected dopamine. The patent data illustrates how the self-condensation of the acid under the influence of the dehydrating agent generates a reactive anhydride-like species in situ, which then rapidly couples with the amine. This mechanism ensures that the reaction proceeds efficiently at lower temperatures, minimizing thermal degradation of the sensitive catechol structure.
Simultaneously, the choice of the silyl protecting group plays a pivotal role in impurity control. The bulky tert-butyl dimethyl silyl (TBDMS) group provides excellent stability against the basic and nucleophilic conditions present during the coupling reaction. This stability prevents the phenolic oxygens from participating in unwanted acylation, a common side reaction in unprotected or acetyl-protected systems. The orthogonality of the silyl group allows it to remain intact during the amide formation and only be cleaved in the final step using a specific fluoride source like tetrabutylammonium fluoride (TBAF). This precise control over reactivity ensures that the final deprotection step yields the target molecule with minimal structural damage, thereby maintaining the integrity of the pharmacophore essential for biological activity.
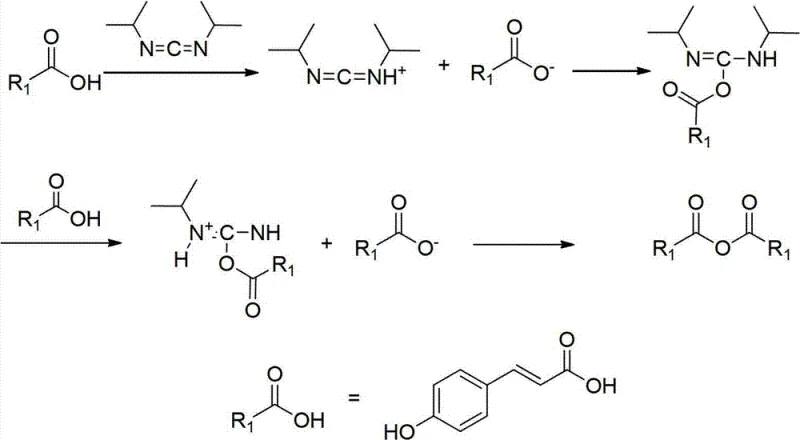
How to Synthesize N-Coumaroyldopamine Efficiently
Implementing this synthesis requires careful attention to stoichiometry and temperature control to maximize the benefits of the new protocol. The process begins with the dissolution of dopamine hydrochloride in a chlorinated solvent such as dichloromethane, followed by the addition of a base like imidazole to scavenge the generated acid. The protecting group reagent is then introduced at controlled temperatures to ensure complete conversion to the protected intermediate. Once the protected amine is isolated, it is reacted with the activated cinnamic acid derivative in a polar aprotic solvent mixture to facilitate the coupling. The detailed standardized synthesis steps below outline the precise operational parameters required to replicate this high-yield process in a pilot or production setting.
- Protect the phenolic hydroxyl groups of Dopamine HCl using a silyl protecting group reagent like Tert-Butyldimethylchlorosilane (TBDMS-Cl) in the presence of a base such as imidazole.
- Activate 4-hydroxycinnamic acid using a dehydrating agent like N,N'-Diisopropylcarbodiimide (DIC) to form a reactive intermediate, then couple with the protected dopamine.
- Remove the silyl protecting groups using a fluoride source such as tetrabutylammonium fluoride (TBAF) to obtain the final high-purity N-Coumaroyldopamine.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders responsible for the bottom line and supply continuity, the shift to this novel synthetic route offers compelling economic arguments. The most significant advantage is the complete elimination of column chromatography from the purification train. In industrial chemistry, chromatography is often the most expensive and slowest unit operation, consuming vast quantities of silica gel and solvents while limiting batch size. By replacing this with recrystallization, the process achieves a drastic simplification of the manufacturing workflow. This simplification translates directly into reduced operational expenditures (OPEX) and a smaller physical footprint for production, allowing for faster turnover of manufacturing suites and improved asset utilization.
Furthermore, the enhanced yield and purity profile of this method significantly mitigates supply chain risks. Higher yields mean that less raw material is required to produce the same amount of finished goods, providing a buffer against fluctuations in the pricing of key starting materials like dopamine and 4-hydroxycinnamic acid. The robustness of the recrystallization process also ensures that the high-purity pharmaceutical intermediates produced meet stringent quality specifications consistently, reducing the likelihood of batch failures or recalls. This reliability is crucial for reducing lead time for high-purity pharmaceutical intermediates, as it minimizes the need for re-processing or extensive quality troubleshooting. Ultimately, the process design prioritizes scalability, ensuring that supply can be ramped up seamlessly to meet market demand without compromising on quality or cost efficiency.
- Cost Reduction in Manufacturing: The removal of column chromatography eliminates the need for expensive silica gel and large volumes of elution solvents, leading to substantial savings in material costs. Additionally, the higher reaction yield reduces the consumption of starting materials per kilogram of product, further driving down the variable cost of production. The simplified workup procedures also reduce labor hours and energy consumption associated with solvent recovery and waste treatment.
- Enhanced Supply Chain Reliability: The use of readily available and stable reagents such as TBDMS-Cl and DIC ensures that the supply chain is not dependent on exotic or hard-to-source catalysts. The robustness of the recrystallization purification step means that the process is less sensitive to minor variations in reaction conditions, resulting in more predictable production schedules. This stability allows for better inventory planning and reduces the risk of stockouts for downstream drug manufacturers.
- Scalability and Environmental Compliance: The process is inherently designed for scale, utilizing standard unit operations like filtration and crystallization that are easily transferred from pilot plants to multi-ton reactors. The reduction in solvent usage and the avoidance of silica waste contribute to a greener manufacturing profile, aligning with increasingly strict environmental regulations. This eco-friendly approach not only reduces disposal costs but also enhances the corporate sustainability profile of the manufacturing entity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of N-Coumaroyldopamine using this advanced methodology. These answers are derived directly from the technical specifications and comparative data provided in the patent literature, offering clarity on why this route is superior to legacy methods. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this intermediate into their own supply chains.
Q: Why is the traditional mixed anhydride method unsuitable for large-scale production?
A: Traditional methods often rely on acetyl protection and mixed anhydride coupling, which generate significant by-products and necessitate column chromatography for purification. This results in low yields (around 37%) and high operational costs, making it economically unviable for industrial manufacturing.
Q: How does the silyl protection strategy improve product purity?
A: By utilizing robust silyl protecting groups (e.g., TBDMS) instead of acetyl groups, the process minimizes side reactions during the coupling phase. Furthermore, the resulting intermediates can be purified via simple recrystallization rather than chromatography, ensuring higher final purity suitable for pharmaceutical applications.
Q: What are the key advantages of using DIC as a dehydrating agent in this synthesis?
A: N,N'-Diisopropylcarbodiimide (DIC) facilitates efficient self-condensation of 4-hydroxycinnamic acid to form reactive intermediates under mild conditions. Its use avoids the harsh conditions associated with traditional acid chloride formations, leading to cleaner reaction profiles and easier downstream processing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Coumaroyldopamine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent to production requires more than just chemical knowledge; it demands engineering excellence and a commitment to quality. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project moves smoothly from development to market. Our facilities are equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of N-Coumaroyldopamine meets the highest international standards. We understand the critical nature of pharmaceutical intermediates and are dedicated to providing a seamless, compliant, and efficient manufacturing service.
We invite you to collaborate with us to leverage this advanced synthetic technology for your specific applications. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how this optimized route can benefit your bottom line. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you secure a stable, high-quality supply of this critical intermediate, empowering your R&D and production teams to focus on innovation and growth.
