Advanced Synthesis of trans-7,cis-9-dodecadieneacetic Ester for Scalable Agrochemical Production
The global demand for effective and environmentally friendly pest control solutions has never been higher, driving intense innovation in the field of insect sex pheromones. A pivotal development in this sector is detailed in patent CN113480428B, which discloses a novel and highly efficient method for preparing trans-7, cis-9-dodecadieneacetic ester, a critical sex pheromone for controlling the cabbage caterpillar, a notorious quarantine pest. This patent represents a significant technological leap forward, addressing long-standing challenges in stereoselectivity and yield that have plagued previous synthetic routes. By leveraging a strategic combination of palladium/copper co-catalysis and iron-mediated coupling, the invention offers a streamlined pathway that drastically reduces synthesis time while enhancing product purity. For R&D directors and procurement specialists alike, this technology signals a shift towards more sustainable and cost-efficient manufacturing paradigms for high-value agrochemical intermediates.
The significance of this patent extends beyond mere academic interest; it provides a robust framework for industrial application where consistency and cost are paramount. The cabbage caterpillar poses a severe threat to agricultural productivity, and reliable access to its specific pheromone is essential for integrated pest management (IPM) strategies. The methodology outlined in CN113480428B utilizes readily available starting materials such as 1-butyne and 1,2-dichloroethylene, effectively bypassing the need for complex, multi-step precursors often required in older protocols. This foundational shift in raw material selection not only lowers the barrier to entry for production but also stabilizes the supply chain against fluctuations in specialty chemical markets. As we delve deeper into the technical specifics, it becomes clear that this process is designed with scalability and operational safety at its core.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of (7E, 9Z)-7,9-dodecadienyl acetate has been fraught with inefficiencies that hinder commercial viability. Early approaches, such as those described by Roelofs et al., relied on the oxidation of 7-bromoheptanoic acid methyl ester followed by a Wittig reaction. While chemically sound in theory, these routes often resulted in crude yields as low as 30% and produced significant amounts of unwanted isomers, necessitating laborious and costly purification steps. Similarly, methods involving boronation and subsequent protonation, like those by Labovitz et al., introduced additional complexity through the use of sensitive organoborane intermediates and cryogenic conditions, leading to total yields often falling below 10%. These traditional pathways are characterized by their reliance on expensive reagents, harsh reaction conditions, and poor atom economy, which collectively drive up the cost of goods sold (COGS) and limit the ability to scale production to meet global agricultural demands.
The Novel Approach
In stark contrast, the methodology presented in patent CN113480428B introduces a streamlined, four-step sequence that elegantly circumvents these historical bottlenecks. The process initiates with a highly efficient coupling of 1-butyne and 1,2-dichloroethylene, catalyzed by a palladium compound and cuprous iodide, to generate 1-chloro-3-alkyne-1-hexene. This intermediate is then subjected to a groundbreaking coupling reaction with a di-Grignard reagent derived from 6-chloro-1-hexanol, uniquely catalyzed by ferric triacetylacetonate. This iron-catalyzed step is crucial as it facilitates the formation of the carbon-carbon bond with high stereoselectivity under mild conditions. The subsequent reduction using zinc powder and final acetylation complete the synthesis with remarkable efficiency. This novel approach not only shortens the synthesis period significantly but also eliminates the need for protecting groups and complex workup procedures, thereby offering a distinct competitive advantage in terms of both time and resource utilization.
Mechanistic Insights into Iron-Catalyzed Coupling and Zinc Reduction
The core of this synthetic breakthrough lies in the sophisticated interplay between the organometallic reagents and the transition metal catalysts employed. The formation of the di-Grignard reagent, ClMgO(CH2)6MgCl, via a one-pot method from 6-chloro-1-hexanol is a masterstroke of process chemistry. By generating this bimodal reagent in situ, the process avoids the isolation of unstable intermediates and the necessity for hydroxyl protection, which traditionally adds at least two steps to the synthesis. When this reagent reacts with 1-chloro-3-alkyne-1-hexene in the presence of ferric triacetylacetonate and hexamethylphosphoric triamide (HMPA), the iron catalyst likely operates through a single-electron transfer (SET) mechanism. This mechanism promotes the cross-coupling reaction while preserving the delicate trans-alkene geometry established in the first step. The addition of HMPA serves to activate the less reactive chloro-leaving group, ensuring high conversion rates without the need for excessive heat or pressure.
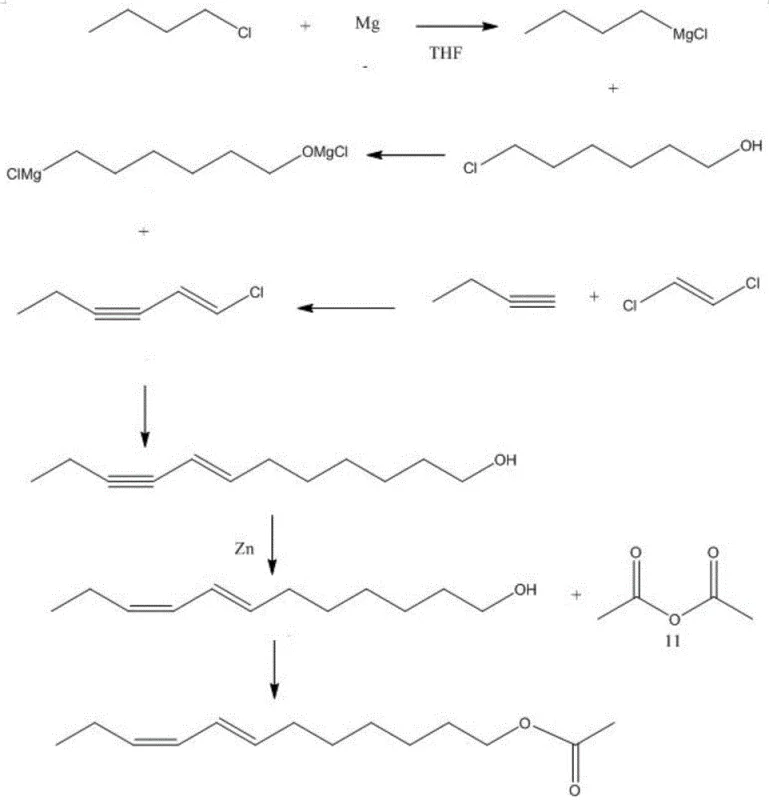
Following the coupling, the stereoselective reduction of the internal alkyne to a cis-alkene is achieved using zinc powder in 1-butanol. This step is mechanistically distinct from catalytic hydrogenation, which often struggles to differentiate between alkene and alkyne reduction or requires high-pressure equipment. The zinc-mediated reduction proceeds via a radical anion intermediate on the metal surface, favoring the thermodynamic cis-product due to steric constraints during protonation. This ensures the formation of the biologically active (7E, 9Z) isomer with high fidelity. Finally, the acetylation with acetic anhydride and pyridine is a standard yet optimized esterification that proceeds rapidly at room temperature. The entire sequence demonstrates a profound understanding of impurity control, as each step is designed to minimize side reactions, resulting in a final product with purity exceeding 90%, which is critical for the efficacy of pheromone-based pest control systems.
How to Synthesize trans-7,cis-9-dodecadieneacetic Ester Efficiently
Implementing this synthesis route requires precise control over reaction parameters, particularly temperature and stoichiometry, to maximize yield and stereoselectivity. The process begins with the careful preparation of the 1-butyne solution in tetrahydrofuran at low temperatures to prevent polymerization, followed by the controlled addition of the palladium and copper catalysts. The subsequent Grignard formation must be initiated carefully to manage the exotherm, ensuring the safety of the operation. Once the di-Grignard reagent is formed, the addition of the iron catalyst and the alkyne intermediate must be performed under strict inert atmosphere conditions to prevent oxidation. The reduction step with zinc powder requires reflux conditions to drive the reaction to completion, monitored closely to ensure >99% conversion before quenching. For a comprehensive, step-by-step guide including exact quantities, temperatures, and workup procedures as validated in the patent examples, please refer to the standardized protocol below.
- Couple 1-butyne with 1,2-dichloroethylene using Pd/Cu catalysts to form 1-chloro-3-alkyne-1-hexene.
- React the alkyne intermediate with a one-pot prepared di-Grignard reagent derived from 6-chloro-1-hexanol using Fe(acac)3 catalysis.
- Perform stereoselective reduction of the alkyne bond using zinc powder in 1-butanol to establish the cis-geometry.
- Finalize the synthesis by acetylating the resulting alcohol with acetic anhydride and pyridine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the technology described in CN113480428B offers transformative potential for cost structures and operational reliability. The most immediate impact is seen in the raw material costs; by utilizing 1-butyne and 1,2-dichloroethylene, which are commodity chemicals with stable pricing, the process decouples production costs from the volatility associated with specialized fine chemical precursors. Furthermore, the elimination of multiple protection and deprotection steps significantly reduces the consumption of solvents and reagents, leading to a substantial reduction in waste disposal costs and environmental compliance burdens. The simplified post-treatment procedures, which rely on standard extraction and distillation rather than complex chromatography, further enhance the throughput capacity of existing manufacturing facilities without requiring significant capital expenditure on new equipment.
- Cost Reduction in Manufacturing: The substitution of expensive noble metal catalysts in the key coupling step with abundant and inexpensive iron salts represents a major driver for cost optimization. Additionally, the high atom economy of the coupling reactions minimizes raw material waste, directly improving the margin profile of the final product. The use of zinc powder for reduction avoids the need for high-pressure hydrogenation reactors, lowering both energy consumption and safety infrastructure costs. These cumulative efficiencies result in a significantly lower cost of goods sold, allowing for more competitive pricing in the global agrochemical market.
- Enhanced Supply Chain Reliability: The reliance on widely available bulk chemicals ensures a resilient supply chain that is less susceptible to disruptions. The robustness of the reaction conditions, which tolerate a broader range of operating parameters compared to sensitive organolithium or boronate routes, reduces the risk of batch failures and production delays. This reliability is crucial for meeting the seasonal demands of the agricultural sector, where timely delivery of pest control agents can determine the success of a harvest. The simplified synthesis also shortens the overall lead time from raw material intake to finished goods, enabling a more agile response to market fluctuations.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated effectively from laboratory to pilot scales with consistent results. The avoidance of toxic heavy metals like chromium or osmium, and the use of safer reducing agents, aligns perfectly with increasingly stringent global environmental regulations. The reduced solvent usage and simpler waste streams facilitate easier treatment and disposal, lowering the environmental footprint of the manufacturing process. This sustainability profile not only mitigates regulatory risk but also enhances the brand value of the end-product in markets that prioritize eco-friendly agricultural solutions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of trans-7, cis-9-dodecadieneacetic ester using this patented methodology. These insights are derived directly from the experimental data and technical specifications provided in the patent documentation, offering clarity on yield expectations, purity profiles, and process safety. Understanding these details is essential for stakeholders evaluating the feasibility of integrating this technology into their existing production portfolios or sourcing strategies.
Q: What are the primary advantages of this synthesis route over traditional Wittig methods?
A: Unlike traditional routes that suffer from low yields (often below 30%) and complex purification due to isomer formation, this patented method utilizes cheap 1-butyne and achieves high stereoselectivity with simplified post-treatment.
Q: How does the process ensure high purity for agrochemical applications?
A: The use of iron-catalyzed coupling followed by specific zinc powder reduction allows for precise control over the trans-7, cis-9 geometry, minimizing unwanted (E,E) isomers and achieving purity levels suitable for sensitive pest control applications.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the process avoids expensive noble metal catalysts in the key coupling step (using Iron instead) and utilizes safe, scalable reagents like zinc powder, making it highly viable for commercial scale-up from kilograms to metric tons.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable trans-7,cis-9-dodecadieneacetic Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality pheromones play in modern sustainable agriculture. Our team of expert chemists has thoroughly analyzed the technical potential of patent CN113480428B and is fully equipped to translate this innovative laboratory process into commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with unwavering consistency. Our state-of-the-art facilities are supported by rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of trans-7, cis-9-dodecadieneacetic ester meets the highest standards required for effective pest control applications.
We invite you to collaborate with us to leverage this advanced synthesis route for your agrochemical projects. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our manufacturing expertise can drive efficiency and reliability in your supply chain. Together, we can advance the availability of superior pest management solutions for a healthier global food system.
