Advanced Manufacturing of 4-(3-Pyridyl)-1H-Imidazole via Novel Cyclization Technology
Advanced Manufacturing of 4-(3-Pyridyl)-1H-Imidazole via Novel Cyclization Technology
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for heterocyclic scaffolds that serve as critical building blocks for active pharmaceutical ingredients (APIs). A pivotal advancement in this domain is documented in Chinese Patent CN1152872C, which discloses a highly efficient preparation method for 4-(3-pyridyl)-1H-imidazole and its derivatives. This specific chemical architecture is renowned for its utility in medicinal chemistry, particularly in the development of kinase inhibitors and other targeted therapies where the pyridyl-imidazole motif provides essential binding interactions. The patent outlines a sophisticated multi-step sequence that transforms readily available oxime precursors into the desired imidazole core through a series of telescoped reactions, thereby enhancing overall process efficiency. By leveraging this proprietary methodology, manufacturers can achieve superior control over the reaction pathway, minimizing the formation of side products that often plague traditional synthesis routes. The structural integrity of the final compound, as depicted in the general formula below, is paramount for downstream biological activity, necessitating a manufacturing process that guarantees high purity and consistent quality. 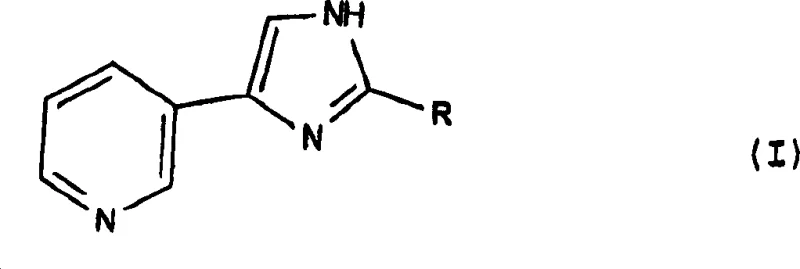
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of substituted imidazoles has been fraught with challenges related to harsh reaction conditions and the instability of key intermediates. Traditional approaches often require the isolation of reactive species such as aziridines or alpha-halo ketones, which are not only hazardous to handle on a large scale but also prone to decomposition during purification. Furthermore, conventional cyclization strategies frequently rely on high temperatures and strong acidic or basic media that can lead to the degradation of sensitive functional groups attached to the pyridine ring. These limitations result in lower overall yields and a complex impurity profile that demands extensive and costly downstream purification processes. For procurement managers, these inefficiencies translate into higher raw material consumption and increased waste disposal costs, while supply chain leaders face risks associated with batch-to-batch variability and potential safety incidents during production. The inability to telescope multiple steps into a single vessel further exacerbates these issues by increasing solvent usage and extending cycle times.
The Novel Approach
In stark contrast, the methodology described in CN1152872C introduces a streamlined pathway that circumvents the isolation of unstable intermediates, thereby significantly enhancing operational safety and chemical efficiency. The process initiates with the conversion of a specific oxime derivative, shown below, which serves as a stable and easily handled starting material. 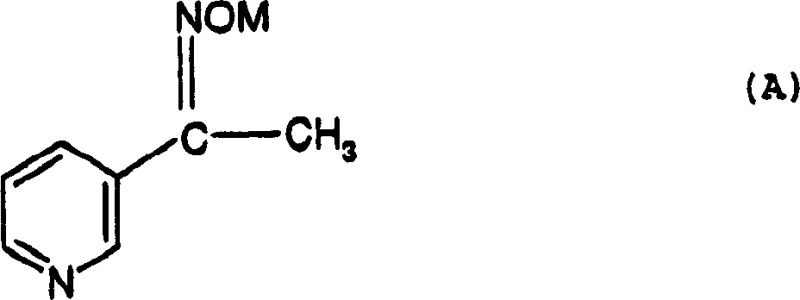 Instead of isolating the subsequent aziridine intermediate, the reaction mixture is directly treated with an acid, such as oxalic acid, to generate a stable amino-acetal species in situ. This telescoping strategy eliminates the need for intermediate work-ups, drastically reducing solvent consumption and processing time. The subsequent formylation and cyclization steps are conducted in a continuous manner within the same reaction medium, utilizing formamide both as a reagent and a solvent. This innovative approach not only simplifies the equipment requirements but also ensures that the reactive intermediates are immediately consumed in the next step, preventing their degradation and maximizing the yield of the final 4-(3-pyridyl)-1H-imidazole product.
Instead of isolating the subsequent aziridine intermediate, the reaction mixture is directly treated with an acid, such as oxalic acid, to generate a stable amino-acetal species in situ. This telescoping strategy eliminates the need for intermediate work-ups, drastically reducing solvent consumption and processing time. The subsequent formylation and cyclization steps are conducted in a continuous manner within the same reaction medium, utilizing formamide both as a reagent and a solvent. This innovative approach not only simplifies the equipment requirements but also ensures that the reactive intermediates are immediately consumed in the next step, preventing their degradation and maximizing the yield of the final 4-(3-pyridyl)-1H-imidazole product.
Mechanistic Insights into Formamide-Mediated Cyclization
The core of this synthetic innovation lies in the mechanistic pathway that converts the linear formamide intermediate into the cyclic imidazole structure. The process involves the generation of a key intermediate, N-[β,β-dimethoxy-2-(3-pyridyl)-ethyl]formamide, which possesses the necessary carbon and nitrogen atoms arranged in a conformation favorable for ring closure. 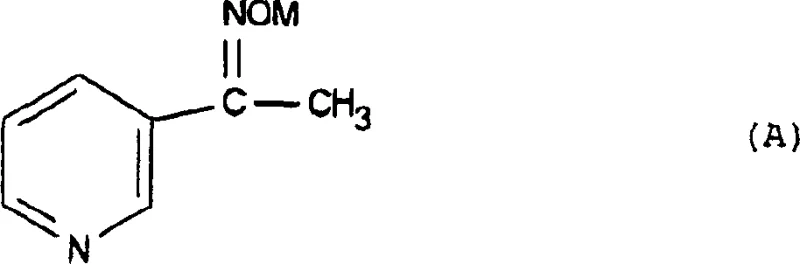 Upon heating, typically in the range of 80°C to 125°C, the formamide group undergoes a dehydration cyclization reaction. The mechanism likely proceeds through the nucleophilic attack of the imidazole nitrogen on the electrophilic carbon of the acetal moiety, facilitated by the thermal energy provided. This intramolecular condensation results in the expulsion of methanol and the formation of the aromatic imidazole ring. The use of formamide as the reaction medium is critical, as it stabilizes the transition state and solubilizes the polar intermediates effectively. From an R&D perspective, understanding this mechanism allows for precise optimization of the heating profile and reaction time to ensure complete conversion while minimizing the formation of polymeric by-products or over-alkylated species.
Upon heating, typically in the range of 80°C to 125°C, the formamide group undergoes a dehydration cyclization reaction. The mechanism likely proceeds through the nucleophilic attack of the imidazole nitrogen on the electrophilic carbon of the acetal moiety, facilitated by the thermal energy provided. This intramolecular condensation results in the expulsion of methanol and the formation of the aromatic imidazole ring. The use of formamide as the reaction medium is critical, as it stabilizes the transition state and solubilizes the polar intermediates effectively. From an R&D perspective, understanding this mechanism allows for precise optimization of the heating profile and reaction time to ensure complete conversion while minimizing the formation of polymeric by-products or over-alkylated species.
Impurity control is inherently built into this mechanism due to the avoidance of isolating the highly reactive aziridine intermediate. In traditional routes, the isolation of aziridines often leads to ring-opening polymerization or hydrolysis, generating difficult-to-remove impurities. By maintaining the reaction in a homogeneous or semi-homogeneous phase and immediately converting the aziridine to the more stable amino-acetal upon acidification, the process effectively 'locks' the carbon skeleton into a stable configuration. Furthermore, the final cyclization step is driven to completion by the distillation of methanol, which shifts the equilibrium towards the product according to Le Chatelier's principle. This thermodynamic driving force ensures high conversion rates and a cleaner crude product profile, reducing the burden on final crystallization or chromatography steps. For quality assurance teams, this translates to a more predictable impurity spectrum and easier validation of the cleaning procedures.
How to Synthesize 4-(3-Pyridyl)-1H-Imidazole Efficiently
The practical execution of this synthesis requires careful attention to temperature control and reagent addition rates to manage the exothermic nature of the initial oxime conversion and the subsequent acidification steps. The process begins by reacting the oxime precursor with sodium methoxide in anhydrous methanol under a nitrogen atmosphere to prevent moisture ingress, which could hydrolyze the sensitive intermediates. Following the formation of the aziridine, the mixture is cooled to near freezing temperatures before the controlled addition of oxalic acid, ensuring the safe generation of the amino-acetal intermediate.
- React 1-(3-pyridyl)-ethanone oxime derivative with sodium methoxide in methanol to form the aziridine intermediate in situ.
- Treat the suspension with oxalic acid at low temperature (0-5°C) to generate the beta,beta-dimethoxy amine intermediate without isolation.
- Add formamide and heat the mixture to induce cyclization, followed by acidification and basification to isolate the final imidazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers substantial strategic advantages beyond mere chemical elegance. The primary benefit lies in the significant reduction of manufacturing costs driven by the telescoped nature of the process. By eliminating multiple isolation and purification steps for intermediates, the facility saves on solvent volumes, filtration media, and drying energy, leading to a leaner and more cost-effective production model. Additionally, the use of commodity chemicals such as methanol, formamide, and oxalic acid ensures a stable and reliable supply of raw materials, mitigating the risk of supply chain disruptions associated with exotic or specialized reagents. The simplified workflow also reduces the total cycle time per batch, allowing for higher throughput and better responsiveness to market demand fluctuations without the need for capital-intensive expansion of reactor capacity.
- Cost Reduction in Manufacturing: The elimination of intermediate isolation steps drastically reduces the consumption of solvents and utilities associated with drying and handling solid intermediates. This streamlined approach minimizes waste generation and lowers the overall cost of goods sold (COGS), providing a competitive pricing advantage in the global market for pharmaceutical intermediates. Furthermore, the high atom economy of the cyclization step ensures that a greater proportion of the starting material mass is incorporated into the final product, reducing raw material costs per kilogram of output.
- Enhanced Supply Chain Reliability: The reliance on widely available and stable starting materials, such as the pyridyl ketone oxime, ensures a robust supply chain that is less susceptible to geopolitical or logistical bottlenecks. The process operates under relatively mild pressure and uses standard stainless steel equipment, meaning it can be easily replicated across multiple manufacturing sites to ensure business continuity. This flexibility allows for dual-sourcing strategies and reduces the lead time for delivering high-purity pharmaceutical intermediates to downstream API manufacturers.
- Scalability and Environmental Compliance: The process is inherently scalable from laboratory benchtop to multi-ton commercial production due to the absence of hazardous isolation steps and the use of manageable reaction temperatures. From an environmental perspective, the reduced solvent usage and the ability to recover and recycle methanol distilled during the cyclization step contribute to a lower environmental footprint. This aligns with increasingly stringent global regulations on industrial emissions and waste disposal, facilitating smoother regulatory approvals and enhancing the corporate sustainability profile.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of 4-(3-pyridyl)-1H-imidazole using this advanced methodology. These insights are derived directly from the technical specifications and experimental data provided in the patent literature, offering clarity on process capabilities and product quality. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this intermediate into their own drug development pipelines.
Q: What are the critical impurities in 4-(3-pyridyl)-1H-imidazole synthesis?
A: Critical impurities often arise from incomplete cyclization or hydrolysis of the acetal intermediate. The patented telescoped method minimizes these by avoiding the isolation of unstable intermediates like the aziridine derivative.
Q: Is the cyclization step exothermic or endothermic?
A: The cyclization of the formamide intermediate (Formula IV) to the imidazole requires heating, typically around 80°C to 125°C, indicating an endothermic activation energy barrier that must be overcome for ring closure.
Q: Can this process be scaled to metric ton production?
A: Yes, the process utilizes common solvents like methanol and formamide and avoids hazardous isolation steps, making it highly suitable for commercial scale-up in standard stainless steel reactors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-(3-Pyridyl)-1H-Imidazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality heterocyclic intermediates play in the successful development of next-generation therapeutics. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. We adhere to stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of 4-(3-pyridyl)-1H-imidazole meets the exacting standards required by global regulatory bodies. Our commitment to excellence extends beyond mere compliance; we actively collaborate with our partners to optimize processes for cost and sustainability.
We invite you to engage with our technical procurement team to discuss your specific requirements and explore how our advanced manufacturing capabilities can support your project goals. By requesting a Customized Cost-Saving Analysis, you can gain valuable insights into how our optimized synthesis route can reduce your overall project costs. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your unique application needs.
