Advanced Convergent Synthesis of N-(2-methyl-5-aminophenyl)-4-(3-pyridyl)-2-pyrimidinamine for Industrial Scale-up
Introduction to Patent CN111440147B Technology
The pharmaceutical industry constantly seeks robust and scalable pathways for critical oncology intermediates, and Patent CN111440147B presents a significant breakthrough in the synthesis of N-(2-methyl-5-aminophenyl)-4-(3-pyridyl)-2-pyrimidinamine, a key precursor for the blockbuster drug Imatinib. This proprietary technology addresses long-standing challenges in the manufacturing of tyrosine kinase inhibitor intermediates by introducing a convergent synthetic strategy that bypasses the limitations of linear routes. By leveraging a novel cyclization approach using thiourea and a controlled oxidation sequence, the patent outlines a method that achieves exceptional purity levels while maintaining mild reaction conditions suitable for global supply chains. For R&D directors and procurement specialists, this represents a viable alternative to legacy processes that often suffer from low yields and hazardous reagent profiles. The technical depth of this disclosure provides a clear roadmap for producing high-quality pharmaceutical intermediates with enhanced environmental compliance and operational efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Imatinib intermediates has been plagued by inefficient linear routes that rely on hazardous starting materials and extreme reaction conditions. Prior art methods frequently utilize toxic aminonitriles and high-boiling, toxic solvents like pyridine, which pose significant safety risks and complicate waste management protocols in large-scale facilities. Furthermore, earlier strategies often required cryogenic temperatures, such as minus 30 degrees Celsius, necessitating expensive cooling infrastructure and increasing energy consumption drastically. Some existing pathways also involve tandem reactions with poor selectivity, leading to the formation of difficult-to-remove isomers that compromise the final product quality. These factors collectively result in low overall yields, often hovering around 43 percent, and create substantial bottlenecks for manufacturers aiming to secure a reliable supply of high-purity intermediates for clinical and commercial applications.
The Novel Approach
In stark contrast, the methodology described in Patent CN111440147B employs a convergent synthesis strategy that constructs the pyrimidine ring directly from readily available enaminone precursors and thiourea. This innovative route eliminates the need for dangerous aminonitriles and avoids the use of carcinogenic solvents like hexamethylphosphoric triamide (HMPA) entirely. A key advancement is the substitution of aggressive methylating agents with the less toxic methyl p-toluenesulfonate, which offers comparable reactivity with a much-improved safety profile. Additionally, the oxidation step utilizes a catalytic system of sodium tungstate and hydrogen peroxide, ensuring complete conversion of the sulfide to the sulfone without generating excessive impurities. This holistic redesign of the synthetic pathway results in significantly higher yields and purity, establishing a new standard for cost reduction in pharmaceutical intermediate manufacturing while adhering to strict environmental regulations.
Mechanistic Insights into Convergent Pyrimidine Construction and Oxidation
The core of this technological advancement lies in the efficient construction of the pyrimidine scaffold through a condensation reaction between (E)-3-(dimethylamino)-1-(pyridin-3-yl)prop-2-en-1-one and thiourea. This cyclization is facilitated by sodium methoxide in ethanol, creating a 2-methylthio-4-(3-pyridyl)pyrimidine intermediate with high regioselectivity. The subsequent methylation step is critical, where the thiol group is converted to a methylthio ether using methyl p-toluenesulfonate under basic conditions. This specific choice of methylating agent minimizes side reactions and ensures the stability of the pyrimidine ring during the transformation. The mechanistic precision here allows for the isolation of the intermediate in high purity, setting the stage for the subsequent oxidation without carrying over significant organic impurities that could affect downstream processing.
Following the methylation, the process employs a green chemistry approach for oxidation, utilizing hydrogen peroxide activated by sodium tungstate to convert the methylthio group into a methylsulfonyl group. This catalytic oxidation is highly selective, preventing over-oxidation or degradation of the sensitive pyridine and pyrimidine rings, which is a common issue with stoichiometric oxidants. The resulting sulfone is an excellent leaving group, facilitating the nucleophilic aromatic substitution with 2-methyl-5-nitroaniline to form the nitro-intermediate. Finally, the nitro group is reduced to the primary amine using a palladium on carbon catalyst under hydrogen pressure. This final hydrogenation step is clean and efficient, avoiding the heavy metal waste associated with iron or tin reduction methods, thereby simplifying the purification process and ensuring the final product meets stringent purity specifications required for API synthesis.
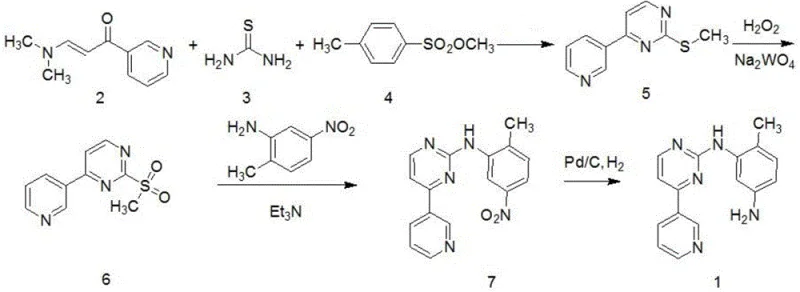
How to Synthesize N-(2-methyl-5-aminophenyl)-4-(3-pyridyl)-2-pyrimidinamine Efficiently
Implementing this synthesis requires precise control over reaction parameters to maximize the benefits of the convergent design. The process begins with the preparation of the pyrimidine core, followed by sequential functionalization to install the necessary groups for coupling. Operators must pay close attention to the oxidation step, ensuring the catalyst loading and temperature are maintained to achieve full conversion of the sulfide to the sulfone. The final reduction step demands careful monitoring of hydrogen uptake to prevent over-reduction or catalyst poisoning. Detailed standard operating procedures regarding solvent removal, crystallization temperatures, and filtration techniques are essential to reproduce the high yields reported in the patent data. For a comprehensive guide on the exact molar ratios, specific temperatures, and workup procedures, please refer to the standardized synthesis protocol below.
- Cyclization of (E)-3-(dimethylamino)-1-(pyridin-3-yl)prop-2-en-1-one with thiourea followed by methylation with methyl p-toluenesulfonate.
- Catalytic oxidation of the methylthio intermediate to the methylsulfonyl derivative using hydrogen peroxide and sodium tungstate.
- Nucleophilic substitution with 2-methyl-5-nitroaniline to form the nitro-intermediate.
- Catalytic hydrogenation using Pd/C under hydrogen pressure to reduce the nitro group to the final amine product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic route offers profound advantages for procurement managers and supply chain leaders focused on stability and cost efficiency. By shifting away from scarce or heavily regulated reagents like aminonitriles and n-butyllithium, manufacturers can secure a more resilient supply chain that is less susceptible to market volatility. The use of commodity chemicals such as thiourea, hydrogen peroxide, and methyl p-toluenesulfonate ensures that raw material sourcing remains straightforward and cost-effective across different geographic regions. Furthermore, the elimination of cryogenic steps reduces the capital expenditure required for specialized reactor equipment, allowing for production in standard multipurpose facilities. This flexibility translates directly into lower overhead costs and faster turnaround times for batch production, enabling suppliers to respond more agilely to fluctuating demand from API manufacturers.
- Cost Reduction in Manufacturing: The replacement of expensive and hazardous reagents with cheaper, safer alternatives drives down the direct material costs significantly. Avoiding the use of noble metal catalysts in the early stages and utilizing a reusable tungsten catalyst for oxidation further optimizes the cost structure. The high selectivity of the reactions minimizes the formation of by-products, which reduces the burden on purification units and lowers solvent consumption during workup. Consequently, the overall cost of goods sold is reduced, providing a competitive pricing advantage for the final intermediate without compromising on quality standards.
- Enhanced Supply Chain Reliability: The reliance on widely available industrial chemicals mitigates the risk of supply disruptions that often plague specialty reagent markets. Since the process does not depend on single-source suppliers for exotic catalysts or solvents, procurement teams can diversify their vendor base to ensure continuity of supply. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality, reducing the rate of batch failures. This reliability is crucial for maintaining consistent inventory levels and meeting the just-in-time delivery requirements of major pharmaceutical clients.
- Scalability and Environmental Compliance: The mild reaction temperatures and absence of highly toxic waste streams make this process inherently scalable from pilot plant to commercial tonnage. The use of aqueous hydrogen peroxide as an oxidant generates water as the primary by-product, aligning with green chemistry principles and simplifying wastewater treatment protocols. This environmental friendliness reduces the regulatory burden and disposal costs associated with hazardous waste, facilitating smoother approvals for capacity expansion. As a result, manufacturers can scale up production rapidly to meet growing market demand for Imatinib generics while maintaining a strong sustainability profile.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation. Understanding these nuances is vital for technical teams evaluating the feasibility of adopting this route for their own production lines. The answers highlight the specific improvements in safety, yield, and operational simplicity that distinguish this method from conventional approaches.
Q: How does this synthesis route improve safety compared to traditional methods?
A: This route eliminates the use of highly toxic aminonitriles and carcinogenic solvents like HMPA found in prior art, utilizing safer reagents like methyl p-toluenesulfonate and aqueous hydrogen peroxide instead.
Q: What is the overall purity achievable with this method?
A: The optimized process conditions, including specific recrystallization steps and selective catalytic oxidation, allow for the production of the final intermediate with purity reaching up to 99.8%.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the reaction conditions are mild (mostly room temperature to 78°C), avoid cryogenic temperatures like -30°C, and use readily available raw materials, making it highly scalable for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-(2-methyl-5-aminophenyl)-4-(3-pyridyl)-2-pyrimidinamine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global supply of life-saving oncology medications. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated chemistry described in Patent CN111440147B can be translated into reliable industrial output. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of N-(2-methyl-5-aminophenyl)-4-(3-pyridyl)-2-pyrimidinamine meets the exacting standards required for API synthesis. Our commitment to process excellence means we can deliver this complex intermediate with the consistency and documentation necessary for regulatory filings.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthesis route can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits specific to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project needs. Let us collaborate to bring efficient, high-purity pharmaceutical solutions to the market faster and more sustainably.
