Scalable Synthesis of High-Purity 3-Methylamine Tetrahydrofuran for Next-Generation Agrochemicals
Introduction to Advanced Intermediate Synthesis
The global demand for third-generation neonicotinoid insecticides, particularly Dinotefuran, has driven an urgent need for high-quality precursors that ensure consistent efficacy and safety profiles. Patent CN107935970B introduces a groundbreaking preparation method for 3-methylamine tetrahydrofuran, a critical building block in this value chain, addressing long-standing industry challenges regarding purity and moisture control. This technology represents a significant leap forward in organic synthesis, moving away from cumbersome multi-step sequences toward a streamlined, efficient reductive amination pathway that aligns perfectly with modern green chemistry principles. By leveraging liquid ammonia and anhydrous methanol under strictly controlled低温 conditions, the process achieves a GC purity exceeding 99.0% and reduces water content to negligible levels below 0.2%, setting a new benchmark for reliable agrochemical intermediate supplier standards. For R&D directors and procurement specialists alike, understanding the nuances of this patented route is essential for securing a competitive advantage in the manufacturing of high-value crop protection agents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-methylamine tetrahydrofuran has relied on complex pathways starting from 1,4-butanediol, involving dehydration cyclization to 2,5-dihydrofuran followed by hydroformylation using cobalt acetate catalysts. These traditional methods are fraught with inefficiencies, including low conversion rates, difficult separation of cobalt residues, and the generation of significant by-products that compromise the purity of the final amine. Furthermore, the use of aqueous ammonia or hydroxylamine compounds in older amination strategies often introduces excessive moisture into the system, creating a barrier for downstream synthesis where anhydrous conditions are paramount. The presence of water not only complicates purification but can also lead to the hydrolysis of sensitive intermediates in subsequent steps, ultimately reducing the overall yield of the final pesticide active ingredient. Consequently, manufacturers have struggled with high production costs and inconsistent quality, necessitating a robust alternative that eliminates these bottlenecks.
The Novel Approach
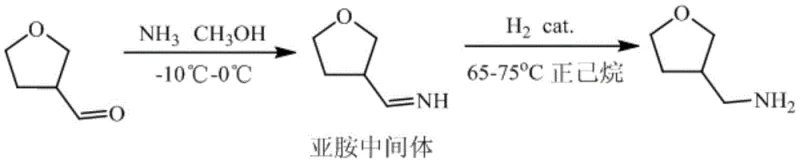
The innovative methodology disclosed in the patent circumvents these issues by employing a direct reductive amination strategy starting from 3-formaldehyde tetrahydrofuran. This approach simplifies the synthetic tree by condensing the reaction steps and utilizing a highly selective catalytic hydrogenation process. A key differentiator is the implementation of a specialized dehydration protocol using n-hexane for azeotropic distillation, which effectively strips residual water from the product matrix without degrading the amine functionality. By controlling the reaction temperature between -10°C and 0°C during the imine formation stage, the process minimizes side reactions and polymerization, ensuring a clean transformation to the imine intermediate. This precision engineering of reaction conditions results in a product that is not only chemically pure but also physically stable, offering substantial cost savings in manufacturing by reducing the need for extensive post-reaction polishing and drying operations.
Mechanistic Insights into Reductive Amination and Catalytic Hydrogenation
The core of this synthesis lies in the elegant mechanism of reductive amination, where the carbonyl group of 3-formaldehyde tetrahydrofuran reacts with liquid ammonia to form a Schiff base or imine intermediate. This initial condensation is equilibrium-driven and requires the removal of water or the use of excess ammonia to push the reaction forward; however, in this specific protocol, the low temperature (-10°C to 0°C) kinetically favors the formation of the imine while suppressing unwanted oligomerization. Once the imine is formed in situ, it undergoes catalytic hydrogenation in the presence of heterogeneous catalysts such as Palladium on Carbon (Pd/C) or Raney Nickel. The hydrogen gas, introduced at pressures between 1.0 MPa and 2.0 MPa, adsorbs onto the metal surface and adds across the carbon-nitrogen double bond of the imine, reducing it to the corresponding primary amine, 3-methylamine tetrahydrofuran. This hydrogenation step is highly exothermic and requires careful thermal management, typically maintained between 65°C and 75°C, to ensure complete conversion without over-reduction or ring-opening of the tetrahydrofuran moiety.
Following the reduction, the reaction mixture contains the target amine along with unreacted ammonia, methanol solvent, and trace water. The purification mechanism is equally sophisticated, utilizing the physicochemical properties of n-hexane to form a low-boiling azeotrope with water. During the distillation phase, the n-hexane co-distills with water, effectively dragging moisture out of the organic phase. This azeotropic dehydration is monitored closely, with sampling continuing until the water content stabilizes, ensuring the final product meets the stringent specification of less than 0.2% moisture. This level of control is critical for pharmaceutical and agrochemical applications where water can act as a poison for downstream catalysts or reactants. The ability to recycle the n-hexane, methanol, and ammonia further underscores the efficiency of the mechanism, creating a closed-loop system that minimizes waste discharge and maximizes atom economy.
How to Synthesize 3-Methylamine Tetrahydrofuran Efficiently
Implementing this synthesis route requires precise adherence to the operational parameters defined in the patent to guarantee reproducibility and safety. The process begins with the preparation of the imine intermediate under inert atmosphere, followed by a high-pressure hydrogenation step that demands rigorous safety protocols. The subsequent workup involves a dual-distillation strategy designed to first recover volatile solvents and then perform the critical dehydration. For technical teams looking to adopt this methodology, the following guide outlines the standardized operational flow derived from the patent examples, ensuring that the transition from laboratory scale to commercial production is seamless and compliant with quality standards.
- React 3-formaldehyde tetrahydrofuran with liquid ammonia in anhydrous methanol at -10°C to 0°C to form the imine intermediate.
- Perform catalytic hydrogenation using Pd/C or Raney Nickel at 65-75°C and 1.0-2.0 MPa pressure to reduce the imine.
- Remove water via n-hexane azeotropic distillation and recover solvents to obtain product with >99% purity and <0.2% water content.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented synthesis route offers profound advantages for supply chain stability and cost structure optimization. The elimination of cobalt-based catalysts and the shift towards more common hydrogenation catalysts like Pd/C or Raney Nickel reduces dependency on scarce or toxic metals, thereby mitigating supply risks associated with raw material volatility. Moreover, the high selectivity of the reaction means that fewer by-products are generated, which directly translates to simplified waste treatment processes and lower environmental compliance costs. For procurement managers, the ability to recycle key reagents such as ammonia and methanol within the plant infrastructure represents a significant opportunity for cost reduction in agrochemical intermediates manufacturing, as it drastically lowers the net consumption of raw materials per kilogram of finished product.
- Cost Reduction in Manufacturing: The process design inherently supports substantial cost savings through the integration of solvent and reagent recovery loops. By distilling and reusing ammonia and methanol, the facility minimizes the purchase volume of these bulk chemicals, leading to a leaner operational expenditure profile. Additionally, the high conversion rate and purity achieved reduce the burden on downstream purification units, saving energy and labor hours that would otherwise be spent on recrystallization or extensive chromatography. The avoidance of expensive transition metal catalysts like cobalt further contributes to a more favorable cost of goods sold (COGS), making the final intermediate more price-competitive in the global market.
- Enhanced Supply Chain Reliability: Utilizing readily available starting materials such as 3-formaldehyde tetrahydrofuran and liquid ammonia ensures a robust supply chain that is less susceptible to disruptions compared to routes relying on specialized or imported precursors. The scalability of the hydrogenation step, which operates at moderate temperatures and pressures compatible with standard industrial reactors, facilitates rapid scale-up from pilot batches to multi-ton production campaigns. This flexibility allows suppliers to respond quickly to fluctuating market demands for Dinotefuran and related derivatives, ensuring continuous availability for clients who rely on just-in-time delivery models for their own manufacturing schedules.
- Scalability and Environmental Compliance: The green chemistry attributes of this method, specifically the low waste generation and the absence of heavy metal contamination in the final product, align perfectly with increasingly strict global environmental regulations. The azeotropic dehydration using n-hexane, which is also recovered and reused, minimizes the volume of hazardous wastewater requiring treatment. This environmental stewardship not only reduces regulatory risk but also enhances the brand reputation of the manufacturer as a sustainable partner. The process is inherently scalable, having been demonstrated in stainless steel reactors up to 1000L capacity in the patent examples, providing a clear pathway for commercial scale-up of complex agrochemical intermediates without the need for exotic equipment.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating the feasibility of this synthesis route, we have compiled answers to common inquiries regarding the process specifics and quality outcomes. These insights are drawn directly from the experimental data and technical disclosures within the patent documentation, providing a transparent view of the technology's capabilities. Understanding these details is crucial for assessing how this intermediate fits into your broader production ecosystem and quality assurance frameworks.
Q: Why is low water content critical for 3-methylamine tetrahydrofuran in Dinotefuran synthesis?
A: High water content can interfere with subsequent coupling reactions, leading to hydrolysis of sensitive reagents and reduced yields in the final pesticide active ingredient. This patent ensures water content below 0.2%.
Q: How does this method improve upon traditional hydroformylation routes?
A: Unlike traditional routes starting from 1,4-butanediol which involve multiple steps and cobalt catalysts, this direct reductive amination offers higher conversion rates, simpler workup, and easier solvent recycling.
Q: Can the catalysts and solvents be recycled in this process?
A: Yes, the process allows for the filtration and reuse of Pd/C or Raney Nickel catalysts, and the distillation recovery of ammonia and methanol, significantly reducing raw material consumption.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Methylamine Tetrahydrofuran Supplier
At NINGBO INNO PHARMCHEM, we recognize that the quality of your final agrochemical product is only as good as the intermediates you start with. Our expertise in scaling diverse pathways from 100 kgs to 100 MT/annual commercial production ensures that we can meet your volume requirements without compromising on the stringent purity specifications demanded by the industry. Our rigorous QC labs are equipped to verify every batch against the high standards set by patents like CN107935970B, guaranteeing low water content and high GC purity that facilitate smooth downstream processing. We are committed to being a reliable 3-methylamine tetrahydrofuran supplier that adds value through technical excellence and supply chain consistency.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this high-efficiency method. We encourage potential partners to contact us for specific COA data and route feasibility assessments tailored to your specific production needs, ensuring a partnership built on transparency, quality, and mutual growth in the competitive agrochemical landscape.
