Safe and Scalable Production of 4,5-Dimethyl-1,3-Dioxole-2-Ketone via Dimethyl Carbonate Transesterification
The pharmaceutical industry is constantly seeking safer, more sustainable pathways for synthesizing critical heterocyclic intermediates, particularly those used in cardiovascular and antibiotic therapies. Patent CN103483307A introduces a transformative methodology for the preparation of 4,5-dimethyl-1,3-dioxole-2-ketone, a pivotal building block for drugs such as Olmesartan and Prulifloxacin. This innovation fundamentally shifts the synthetic paradigm away from hazardous phosgene-based chemistry toward a benign transesterification-cyclization sequence using dimethyl carbonate. By leveraging alkali metal alcoholates as catalysts within an aliphatic ether solvent system, this process achieves high purity levels exceeding 99% while drastically mitigating the occupational health risks associated with traditional carbonylation agents. For R&D directors and procurement strategists, this patent represents a viable route to secure supply chains against regulatory tightening on toxic reagents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of cyclic carbonates like 4,5-dimethyl-1,3-dioxole-2-ketone relied heavily on the reaction of diols or hydroxy-ketones with phosgene, diphosgene, or triphosgene. These reagents are classified as extreme safety hazards; phosgene is a potent pulmonary irritant and chemical warfare agent, while triphosgene decomposes violently upon contact with moisture or at elevated temperatures to release toxic gas. The handling of such materials necessitates expensive, specialized containment infrastructure, rigorous safety protocols, and complex waste treatment systems to neutralize residual toxicity. Furthermore, the thermal instability of triphosgene above 130°C creates inherent risks during the exothermic reaction phases, often leading to batch inconsistencies and potential reactor runaway scenarios. These factors collectively inflate the cost of goods sold and introduce significant liability into the supply chain, making conventional routes increasingly untenable for modern green chemistry mandates.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes dimethyl carbonate (DMC) as a non-toxic, biodegradable carbonyl source that reacts efficiently with 3-hydroxy-2-butanone. This method operates under relatively mild conditions, initiating with a transesterification step at 60-100°C followed by a cyclization phase at 110-160°C. The elimination of halogenated reagents not only removes the risk of acute toxicity but also simplifies the downstream purification process, as there are no chloride salts or phosgene residues to manage. The use of recoverable aliphatic ether solvents and the ability to distill off methanol byproduct drive the equilibrium toward completion, resulting in a robust process with excellent atom economy. This shift allows manufacturers to operate with reduced insurance premiums and lower environmental compliance burdens, directly translating to a more resilient and cost-effective production model for high-value pharmaceutical intermediates.
Mechanistic Insights into Alkali Metal Alcoholate-Catalyzed Cyclization
The core of this synthesis lies in a sequential base-catalyzed mechanism that transforms the hydroxy-ketone substrate into the target cyclic carbonate through a stable intermediate. Initially, the alkali metal alcoholate catalyst deprotonates the hydroxyl group of 3-hydroxy-2-butanone, generating a nucleophilic alkoxide species. This alkoxide attacks the carbonyl carbon of dimethyl carbonate, displacing a methoxide ion to form a mono-methyl carbonate ester intermediate. This transesterification step is reversible, which is why the process requires the continuous removal of methanol at 60-100°C to prevent the reverse reaction and ensure high conversion rates. The choice of catalyst, such as sodium methylate or potassium ethylate, is critical as it must be strong enough to initiate the nucleophilic attack but compatible with the ether solvent system to maintain homogeneity throughout the reaction mixture.
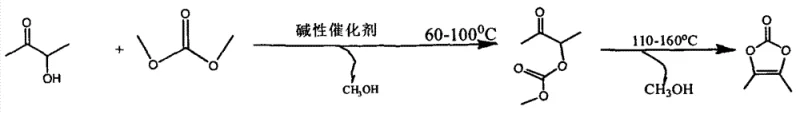
Following the formation of the linear carbonate intermediate, the reaction temperature is elevated to 110-160°C to induce intramolecular cyclization. At this higher thermal energy, the remaining carbonyl oxygen or the adjacent ketone functionality participates in a ring-closing event, expelling the second molecule of methanol. This step is thermodynamically driven by the entropy gain from releasing volatile methanol and the stability of the five-membered dioxolane ring. The precise control of this temperature window is essential; too low, and the cyclization is incomplete, leaving linear impurities; too high, and thermal degradation of the sensitive carbonate linkage may occur. The result is a clean conversion to 4,5-dimethyl-1,3-dioxole-2-ketone, which can be isolated via simple crystallization after neutralizing the basic catalyst, ensuring a final product with minimal impurity profiles suitable for sensitive API applications.
How to Synthesize 4,5-Dimethyl-1,3-Dioxole-2-Ketone Efficiently
Implementing this synthesis requires careful attention to the stoichiometric ratios and thermal profiling to maximize yield and purity. The process begins by charging the reactor with 3-hydroxy-2-butanone, dimethyl carbonate, and the chosen aliphatic ether solvent under an inert atmosphere. The detailed standardized synthesis steps, including specific molar ratios and distillation parameters, are outlined below to guide process engineers in replicating the high-efficiency results demonstrated in the patent examples.
- Mix 3-hydroxy-2-butanone, dimethyl carbonate, alkali metal alcoholate catalyst, and aliphatic ether solvent. Heat at 60-100°C for 3-6 hours to perform transesterification while distilling off methanol.
- Raise the reaction temperature to 110-160°C and maintain for 2-5 hours to induce cyclization, continuing to remove methanol byproduct.
- Cool the mixture, neutralize with hydrochloric acid, recover solvents via fractionation, and crystallize the residue at 0-5°C to obtain the pure product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this dimethyl carbonate-based route offers substantial strategic benefits beyond mere technical feasibility. The primary advantage lies in the drastic reduction of operational risk; by eliminating Class A toxic substances like phosgene from the supply chain, facilities can avoid the costly permitting processes and security measures required for hazardous material storage. This inherently safer design principle leads to significant cost reductions in manufacturing overhead, as the need for specialized scrubbing systems and emergency containment protocols is minimized. Furthermore, the raw materials involved, specifically dimethyl carbonate and acetoin, are commodity chemicals with stable global supply networks, ensuring consistent availability and shielding production schedules from the volatility often associated with regulated toxic reagents.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the simplification of the workflow and the elimination of expensive hazard mitigation infrastructure. Without the need for phosgene generators or complex neutralization trains for toxic off-gases, capital expenditure for new plants is lowered, and operating expenses for existing facilities are reduced. The ability to recover and recycle the aliphatic ether solvent and excess dimethyl carbonate further enhances the cost efficiency, creating a closed-loop system that minimizes raw material waste. Additionally, the high selectivity of the reaction reduces the burden on downstream purification, lowering the consumption of energy and solvents during the crystallization and drying phases.
- Enhanced Supply Chain Reliability: Reliability is bolstered by the use of non-restricted raw materials that do not face the stringent transportation regulations imposed on phosgene derivatives. This ease of logistics allows for more flexible sourcing strategies and reduces the lead time for high-purity pharmaceutical intermediates, as suppliers are not bottlenecked by hazardous material shipping windows. The robustness of the reaction conditions also means that batch failure rates due to reagent instability are significantly diminished, providing a more predictable output volume for long-term supply agreements. This stability is crucial for maintaining continuous API production lines where interruptions can have cascading effects on drug availability.
- Scalability and Environmental Compliance: From an environmental standpoint, this route aligns perfectly with green chemistry principles, generating methanol as the primary byproduct rather than chlorinated waste streams. This simplifies wastewater treatment and reduces the environmental footprint of the manufacturing site, facilitating easier compliance with increasingly strict international environmental regulations. The process has been proven scalable from laboratory glassware to multi-kilogram reactors without loss of efficiency, demonstrating its readiness for commercial scale-up of complex cyclic carbonates. This scalability ensures that as demand for the downstream APIs grows, the intermediate supply can be ramped up rapidly without requiring fundamental process re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on process capabilities and product quality standards.
Q: Why is the dimethyl carbonate route preferred over phosgene methods for this intermediate?
A: Traditional methods utilize highly toxic phosgene or triphosgene, posing severe safety risks and requiring specialized containment. The dimethyl carbonate route eliminates these hazards, offering a greener, safer profile suitable for modern regulatory environments.
Q: What represents the critical control points in the thermal cyclization step?
A: Precise temperature control between 110-160°C is vital. This range ensures complete cyclization of the mono-methyl carbonate intermediate while driving the equilibrium forward by continuously removing methanol, maximizing yield without degrading the sensitive cyclic carbonate structure.
Q: Can this process be scaled for industrial API production?
A: Yes, the patent explicitly demonstrates scalability from laboratory glassware to 50L reactors with consistent purity (>99%) and yield. The use of common solvents like triglyme and standard distillation equipment facilitates straightforward commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4,5-Dimethyl-1,3-Dioxole-2-Ketone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting safer, more efficient synthetic routes for key pharmaceutical intermediates like 4,5-dimethyl-1,3-dioxole-2-ketone. Our CDMO team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale patent examples to industrial reality is seamless. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the exacting standards required for API synthesis, delivering a product that supports the highest levels of drug safety and efficacy.
We invite you to collaborate with us to optimize your supply chain for this vital intermediate. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our implementation of this green synthesis technology can enhance your production reliability and reduce overall manufacturing costs.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
