Scalable Synthetic Route for High-Purity Crocetin: A Technical Breakthrough for Global Supply Chains
Scalable Synthetic Route for High-Purity Crocetin: A Technical Breakthrough for Global Supply Chains
The global demand for high-value carotenoids, particularly Crocetin, has surged due to its potent antioxidant, anticancer, and lipid-lowering properties, yet traditional supply chains remain bottlenecked by agricultural limitations. Patent CN101157645B introduces a robust, fully synthetic methodology that bypasses the extraction of saffron or gardenia, offering a transformative solution for the reliable crocetin supplier market. This technical insight report analyzes the proprietary three-step Wittig coupling strategy detailed in the patent, which utilizes 3,7-dimethyl octatriene dialdehyde and methyl 2-bromopropionate as key building blocks. By shifting from biological extraction to controlled chemical synthesis, manufacturers can achieve unprecedented consistency in impurity profiles and batch-to-batch reproducibility. The following analysis dissects the mechanistic advantages, process scalability, and strategic commercial implications for R&D directors and procurement leaders seeking to secure long-term access to this critical high-purity pharmaceutical intermediate.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the procurement of Crocetin has been plagued by the inherent volatility of natural product extraction, where yield is dictated by crop seasons, geographical origin, and climatic conditions. Conventional extraction from saffron stigmas or gardenia fruits involves complex downstream processing to remove co-extracted pigments, sugars, and proteins, often resulting in variable purity and significant batch-to-batch inconsistency. Furthermore, the low natural abundance of Crocetin in plant matrices necessitates the processing of massive quantities of raw biomass, leading to exorbitant costs and an unsustainable environmental footprint. For cost reduction in food additive manufacturing, reliance on these natural sources creates a fragile supply chain vulnerable to harvest failures and price speculation. Additionally, the presence of trace allergens or pesticide residues in botanical extracts poses significant regulatory hurdles for pharmaceutical applications, requiring extensive and costly purification protocols that traditional methods struggle to optimize efficiently.
The Novel Approach
The synthetic route disclosed in the patent represents a paradigm shift by constructing the carotenoid backbone through precise organic transformations rather than isolation. The process initiates with the generation of a phosphonium ylide from methyl 2-bromopropionate, which is subsequently coupled with chloroacetaldehyde to form the critical methyl chlorotigrate intermediate. This modular approach allows for the independent optimization of each fragment before the final assembly, ensuring that impurities are managed at the source rather than removed at the end. 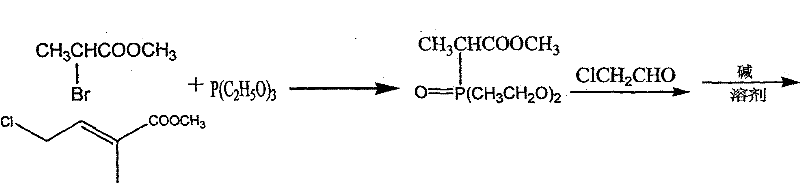 By utilizing readily available petrochemical-derived starting materials, the method decouples production from agricultural cycles, enabling continuous manufacturing operations. The use of standard organic solvents such as dimethylformamide and isopropyl ether, combined with manageable reaction temperatures between 45°C and 100°C, facilitates straightforward commercial scale-up of complex carotenoid intermediates. This synthetic flexibility not only guarantees a steady supply but also allows for the fine-tuning of stereochemistry to match the bioactivity of the natural all-trans isomer.
By utilizing readily available petrochemical-derived starting materials, the method decouples production from agricultural cycles, enabling continuous manufacturing operations. The use of standard organic solvents such as dimethylformamide and isopropyl ether, combined with manageable reaction temperatures between 45°C and 100°C, facilitates straightforward commercial scale-up of complex carotenoid intermediates. This synthetic flexibility not only guarantees a steady supply but also allows for the fine-tuning of stereochemistry to match the bioactivity of the natural all-trans isomer.
Mechanistic Insights into Wittig Olefination and Chain Elongation
The core of this synthetic strategy relies on the iterative application of the Wittig reaction, a powerful tool for constructing carbon-carbon double bonds with high stereoselectivity. In the initial phase, methyl 2-bromopropionate reacts with a phosphine reagent, such as triphenylphosphine, to generate a phosphonium salt, which is then deprotonated by a strong base like potassium hydroxide or sodium ethoxide to form the reactive ylide species. This nucleophilic ylide attacks the carbonyl carbon of chloroacetaldehyde, proceeding through a four-membered oxaphosphetane transition state that collapses to release triphenylphosphine oxide and form the desired alkene linkage. The careful control of reaction conditions, specifically maintaining a nitrogen atmosphere and utilizing antioxidants, is paramount to preventing the oxidative degradation of the conjugated polyene system, which is highly susceptible to radical attack. The subsequent extension of the carbon chain involves converting the chloro-intermediate into a second ylide, which then undergoes a double condensation with 3,7-dimethyl octatriene dialdehyde. 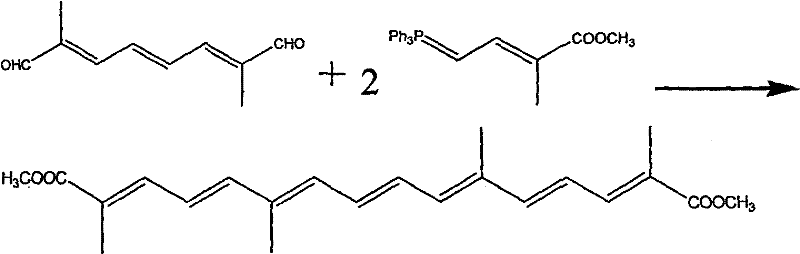 This final coupling step is the most critical determinant of the overall molecular architecture, establishing the extended conjugation responsible for Crocetin's characteristic gold color and UV absorption properties. The patent specifies a molar excess of the chloro-intermediate relative to the dialdehyde (ratios of 3:1 to 8:1) to drive the equilibrium towards the di-substituted product, minimizing the formation of mono-substituted byproducts that would complicate downstream purification.
This final coupling step is the most critical determinant of the overall molecular architecture, establishing the extended conjugation responsible for Crocetin's characteristic gold color and UV absorption properties. The patent specifies a molar excess of the chloro-intermediate relative to the dialdehyde (ratios of 3:1 to 8:1) to drive the equilibrium towards the di-substituted product, minimizing the formation of mono-substituted byproducts that would complicate downstream purification.
Impurity control is meticulously addressed through the selection of specific solvent systems and workup procedures tailored to the polarity of the intermediates. For instance, the precipitation of the ylide using aqueous base followed by recrystallization from ethyl acetate effectively removes unreacted phosphine salts and inorganic halides before they can contaminate the final coupling step. The hydrolysis of the dimethyl ester precursor is conducted under mild alkaline conditions (60°C to 100°C) to cleave the ester groups without inducing isomerization of the double bonds, a common side reaction in carotenoid chemistry. Final purification via recrystallization from mixed solvents, such as ethanol-chloroform or carbon tetrachloride, leverages the differential solubility of the target acid versus geometric isomers and oligomeric byproducts. This rigorous attention to mechanistic detail ensures that the final product meets stringent specifications, with HPLC analysis confirming purity levels exceeding 96%, thereby satisfying the rigorous demands of reducing lead time for high-purity pharmaceutical intermediates without compromising on quality.
How to Synthesize Crocetin Efficiently
The implementation of this synthetic route requires precise adherence to the stoichiometric ratios and atmospheric controls outlined in the patent to maximize yield and minimize waste. Operators must ensure that all glassware is thoroughly dried and purged with nitrogen prior to the addition of moisture-sensitive reagents like phosphines and alkoxides. The detailed standardized synthesis steps, including specific addition rates, temperature ramps, and quenching protocols, are essential for replicating the high purity demonstrated in the experimental examples.
- Preparation of methyl bromopropionate ylide using triphenylphosphine and base under nitrogen protection.
- Reaction of the ylide with chloroacetaldehyde to form methyl chlorotigrate intermediate.
- Final coupling with 3,7-dimethyloctatriene dialdehyde followed by hydrolysis and recrystallization to obtain pure Crocetin.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this synthetic Crocetin pathway offers profound strategic advantages that extend far beyond simple unit cost metrics. By eliminating the dependency on volatile agricultural commodities, organizations can stabilize their raw material costs and secure long-term supply contracts that are immune to the vagaries of weather patterns or geopolitical disruptions in farming regions. The ability to produce Crocetin on demand using established chemical infrastructure significantly reduces the risk of stockouts, ensuring business continuity for downstream formulations in the nutraceutical and pharmaceutical sectors. Furthermore, the simplified purification train, which avoids the complex chromatographic separations often required for natural extracts, streamlines the manufacturing workflow and reduces the consumption of expensive stationary phases and solvents.
- Cost Reduction in Manufacturing: The synthetic route utilizes commodity chemicals such as methyl 2-bromopropionate and chloroacetaldehyde, which are produced at massive scales globally, ensuring a stable and low-cost feedstock supply compared to the labor-intensive harvesting of saffron. By removing the need for extensive biomass processing and the associated waste disposal costs, the overall operational expenditure is drastically lowered. The elimination of transition metal catalysts in favor of phosphorus-based reagents further simplifies the removal of heavy metal impurities, reducing the cost and complexity of the final polishing steps required for regulatory compliance.
- Enhanced Supply Chain Reliability: Unlike natural extraction, which is constrained by harvest seasons and storage limitations of perishable plant materials, chemical synthesis can operate continuously throughout the year. This capability allows manufacturers to build strategic inventory buffers and respond rapidly to spikes in market demand without the long lead times associated with crop cultivation. The robustness of the reaction conditions, which tolerate standard industrial equipment and do not require cryogenic temperatures or ultra-high vacuum, ensures that production can be easily transferred between different manufacturing sites if necessary, enhancing overall supply chain resilience.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing solvents and reagents that are compatible with large-scale reactor systems and standard wastewater treatment protocols. The absence of toxic heavy metals and the use of recyclable organic solvents align with increasingly strict environmental regulations, reducing the liability and permitting hurdles associated with new manufacturing facilities. The high atom economy of the Wittig coupling steps, combined with efficient recovery of phosphine oxide byproducts, minimizes the generation of hazardous waste, supporting corporate sustainability goals and green chemistry initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic technology, providing clarity on its feasibility and advantages over traditional methods. These insights are derived directly from the experimental data and process descriptions within the patent documentation to ensure accuracy and relevance for technical decision-makers.
Q: What are the primary advantages of synthetic Crocetin over natural extraction?
A: Synthetic production eliminates dependence on seasonal agricultural crops like saffron or gardenia, ensuring consistent supply continuity, standardized purity levels above 96%, and independence from climate-related yield fluctuations.
Q: How is oxidation controlled during the Wittig reaction steps?
A: The process strictly utilizes nitrogen protection throughout all reaction stages and incorporates antioxidant systems to prevent the degradation of the sensitive polyene chain structure inherent to carotenoids.
Q: Is the synthetic route scalable for industrial manufacturing?
A: Yes, the methodology employs standard organic solvents and manageable temperature ranges (40°C to 100°C), making it highly adaptable for commercial scale-up from pilot plants to multi-ton annual production capacities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Crocetin Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of securing a stable supply of high-value carotenoids for your pharmaceutical and food applications. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistent quality. We employ stringent purity specifications and operate rigorous QC labs equipped with advanced HPLC and spectroscopic instrumentation to verify that every batch of synthetic Crocetin meets the highest industry standards for identity and potency. Our commitment to technical excellence allows us to deliver a product that not only matches but often exceeds the performance characteristics of natural extracts, providing you with a competitive edge in the marketplace.
We invite you to engage with our technical procurement team to discuss how this innovative synthetic route can be integrated into your supply chain to drive efficiency and reduce costs. Contact us today to request a Customized Cost-Saving Analysis tailored to your specific volume needs and to obtain specific COA data and route feasibility assessments. Let us partner with you to unlock the full potential of synthetic Crocetin, ensuring your projects proceed without interruption and with the highest level of quality assurance.
