Scalable Synthesis of Isopropyl-Beta-D-Thiogalactoside via Novel Thiourea Activation
Introduction to Advanced IPTG Manufacturing Technologies
The biotechnology and pharmaceutical sectors rely heavily on high-purity induction agents for recombinant protein expression, with Isopropyl-Beta-D-Thiogalactoside (IPTG) standing as a cornerstone reagent for lac operon systems. However, the historical reliance on hazardous raw materials has created significant bottlenecks in the reliable supply of this critical fine chemical intermediate. Patent CN103087121A introduces a transformative synthetic methodology that fundamentally restructures the production landscape by replacing volatile mercaptans with a stable thiourea-based activation strategy. This innovation not only addresses severe environmental, health, and safety (EHS) concerns but also resolves persistent issues regarding isomeric impurities that have long plagued conventional manufacturing processes. By leveraging penta-acetyl galactose as a robust starting material, this novel pathway ensures a consistent supply of high-purity IPTG suitable for sensitive molecular biology applications. The strategic shift towards this cleaner synthesis route represents a pivotal advancement for any organization seeking a reliable agrochemical intermediate supplier or pharma partner committed to sustainable chemistry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional industrial synthesis of IPTG typically involves the direct reaction of penta-acetyl galactose with isopropyl mercaptan under Lewis acid catalysis, a process fraught with substantial operational and safety liabilities. The primary drawback lies in the intrinsic properties of isopropyl mercaptan, which possesses an intensely unpleasant odor and significant toxicity, necessitating expensive containment infrastructure and complex waste treatment protocols to protect personnel and the environment. Furthermore, the direct glycosylation conditions often lack sufficient stereocontrol, leading to the concurrent formation of alpha-anomeric impurities alongside the desired beta-product. These isomeric byproducts are structurally similar to the target molecule, making their removal via crystallization or chromatography exceptionally difficult and costly, thereby driving down the overall process yield. Consequently, these legacy methods are often restricted to small-scale laboratory preparations and fail to meet the rigorous demands of modern commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
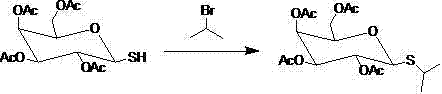
In stark contrast, the methodology disclosed in CN103087121A circumvents these challenges by employing a stepwise construction of the thio-glycosidic bond that completely bypasses the need for free isopropyl mercaptan. The process initiates with the displacement of the anomeric acetate by thiourea, generating a stable isothiourea salt intermediate that can be easily isolated and purified, effectively locking in the stereochemistry early in the sequence. Subsequent cleavage of this salt with pyrosulfite releases the sugar-bound thiol functionality in a controlled manner, which is then alkylated using isopropyl bromide, a reagent that is significantly easier to handle and less odorous than its mercaptan counterpart. This strategic decoupling of sulfur introduction and alkylation steps ensures superior regioselectivity and minimizes the generation of hard-to-remove isomers. The result is a streamlined workflow that delivers high-purity products with simplified downstream processing, offering a compelling solution for cost reduction in electronic chemical manufacturing and broader fine chemical sectors.
Mechanistic Insights into Thiourea-Mediated Glycosyl Activation
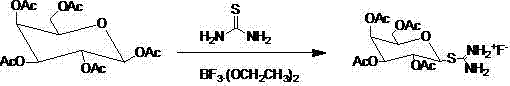
The core chemical innovation of this patent lies in the utilization of thiourea as a soft nucleophile to activate the anomeric center of the galactose derivative under Lewis acidic conditions. In the initial step, boron trifluoride etherate coordinates with the anomeric oxygen of penta-acetyl galactose, facilitating the departure of the acetate group and generating a reactive oxocarbenium ion species. Thiourea then attacks this electrophilic center to form the S-tetraacetylgalactose isothiourea fluoride salt, a crystalline solid that serves as a masked thiol equivalent. This transformation is critical because it avoids the equilibrium issues and reversibility often seen in direct thiol additions, effectively driving the reaction to completion with high stereochemical fidelity. The stability of this isothiourea salt allows for intermediate storage or transport, adding flexibility to the manufacturing schedule that is rarely available in continuous one-pot mercaptan reactions.
Following the formation of the isothiourea salt, the mechanism proceeds through a nucleophilic cleavage mediated by metapyrosulfite ions in a biphasic solvent system. The pyrosulfite acts as a specific reagent to hydrolyze the C-N bonds of the isothiourea moiety, liberating the free thiol group on the sugar scaffold without affecting the protecting acetyl groups on the other hydroxyls. This generated S-tetraacetylgalactose thiol is then poised for the final coupling step, where it acts as a nucleophile against isopropyl bromide in the presence of a mild base like potassium carbonate. This SN2-type alkylation is highly efficient and occurs under relatively mild thermal conditions, preserving the integrity of the carbohydrate ring. The final deacetylation under controlled pH conditions (pH 9-11) removes the protecting groups to yield the final water-soluble IPTG, ensuring that the impurity profile remains exceptionally clean throughout the entire synthetic sequence.
How to Synthesize Isopropyl-Beta-D-Thiogalactoside Efficiently
The implementation of this synthesis route requires precise control over reaction stoichiometry and solvent selection to maximize the yield of the crystalline intermediates. The process begins with the dissolution of penta-acetyl galactose in dichloromethane, followed by the addition of thiourea and boron trifluoride etherate at ambient temperatures to precipitate the isothiourea salt.
- React penta-acetyl galactose with thiourea and boron trifluoride to form S-tetraacetylgalactose isothiourea fluoride salt.
- Cleave the isothiourea salt using sodium metabisulfite to generate the S-tetraacetylgalactose thiol intermediate.
- Alkylate the thiol intermediate with isopropyl bromide followed by alkaline deacetylation to yield pure IPTG.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this thiourea-based synthesis route offers profound advantages in terms of operational expenditure and risk mitigation. By eliminating the requirement for isopropyl mercaptan, facilities can drastically reduce the costs associated with specialized odor-control scrubbing systems and hazardous material handling protocols, leading to significant overhead savings. The avoidance of toxic raw materials also simplifies regulatory compliance and reduces the liability exposure for the manufacturing site, ensuring uninterrupted production schedules even in regions with stringent environmental regulations. Furthermore, the high purity of the crude product obtained through this method minimizes the need for extensive recrystallization or chromatographic purification, directly translating to reduced solvent consumption and shorter cycle times. These factors collectively contribute to a more resilient supply chain capable of meeting the fluctuating demands of the global biotechnology market without compromising on quality or delivery timelines.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous mercaptan handling infrastructure results in substantial capital and operational expenditure savings for production facilities. The high yield of the crystallization steps reduces material loss, while the simplified purification train lowers utility and solvent costs significantly. Additionally, the use of commodity chemicals like thiourea and isopropyl bromide ensures stable raw material pricing and availability, shielding the supply chain from volatile specialty chemical markets.
- Enhanced Supply Chain Reliability: Utilizing stable solid intermediates like the isothiourea salt allows for decoupled production stages, enabling manufacturers to stockpile key precursors and respond rapidly to surges in demand. The robustness of the reaction conditions, which tolerate standard industrial solvents and ambient pressures, reduces the risk of batch failures due to equipment sensitivity. This reliability ensures a consistent flow of high-purity IPTG to downstream customers, preventing bottlenecks in their own protein expression workflows.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to work efficiently with standard reactor setups without requiring exotic high-pressure or cryogenic equipment. The reduction in toxic waste streams, particularly the absence of mercaptan residues, simplifies effluent treatment and aligns with green chemistry principles. This environmental compatibility facilitates easier permitting for capacity expansion and supports corporate sustainability goals, making it an attractive option for long-term partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation to provide clarity on process feasibility.
Q: How does this synthesis method improve safety compared to traditional routes?
A: This method eliminates the direct use of isopropyl mercaptan, a highly toxic and foul-smelling reagent, by generating the sulfur functionality via a thiourea displacement pathway.
Q: What is the primary advantage regarding product purity?
A: The stepwise approach prevents the formation of difficult-to-remove isomeric impurities often associated with direct Lewis acid-catalyzed glycosylation using mercaptans.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the patent explicitly highlights high yields and simple operation conditions, such as ambient temperature reactions and standard solvent systems, making it ideal for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Isopropyl-Beta-D-Thiogalactoside Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality induction agents play in the success of modern biopharmaceutical research and development. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We adhere to stringent purity specifications and utilize rigorous QC labs to verify that every batch of IPTG meets the exacting standards required for sensitive genetic engineering applications. Our commitment to process excellence means we can deliver the benefits of this advanced thiourea-based synthesis to our global clientele, guaranteeing a product free from the odorous and toxic contaminants associated with older methods.
We invite potential partners to engage with our technical procurement team to discuss how this optimized manufacturing route can benefit your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic advantages of switching to our superior grade intermediates. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your volume needs, ensuring a secure and high-performance supply of this essential biochemical reagent.
