Advanced Synthesis of Isooctyl 2,4-D Ester for Scalable Agrochemical Manufacturing
The global agrochemical industry is constantly seeking more stable and environmentally compliant herbicide intermediates to replace volatile legacy compounds. Patent CN103772200A introduces a robust preparation method for isooctyl (2,4-dichlorophenoxy) acetate, a critical intermediate that addresses the severe drift issues associated with traditional 2,4-D butyl esters. This technical breakthrough offers a pathway to synthesize high-purity agrochemical intermediates with yields exceeding 95%, utilizing a streamlined esterification process that leverages azeotropic dehydration. For R&D directors and procurement managers, this patent represents a significant opportunity to optimize supply chains by adopting a method that balances high reaction efficiency with simplified downstream processing. The shift towards isooctyl derivatives is not merely a chemical substitution but a strategic move to enhance field efficacy and reduce environmental liability, making the reliable sourcing of this specific intermediate a priority for forward-thinking agricultural chemical manufacturers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the agricultural sector has relied heavily on 2,4-D butyl ester due to its ease of synthesis, but this compound suffers from critical physicochemical drawbacks that limit its modern application. The primary deficiency lies in its high volatility; at temperatures as low as 15°C, the butyl ester readily evaporates into a gaseous state, leading to significant spray drift during application. This drift causes unintended damage to broadleaf crops in adjacent fields, resulting in substantial economic losses and regulatory scrutiny for farmers and chemical formulators. Furthermore, conventional synthesis routes often struggle with controlling side reactions and achieving consistent purity without expensive purification steps, leading to variable product quality. The environmental persistence and off-target movement of these volatile esters have prompted a global push for alternatives that maintain herbicidal potency while offering superior physical stability and reduced vapor pressure.
The Novel Approach
The methodology outlined in the patent data presents a sophisticated solution by substituting the butyl group with an isooctyl chain, fundamentally altering the volatility profile of the final herbicide formulation. This novel approach utilizes a specific ratio of 2,4-dichlorophenoxyacetic acid to isooctyl alcohol, catalyzed by protonic acids in a toluene solvent system to drive the equilibrium forward efficiently. By employing toluene, the process facilitates continuous water removal through azeotropic distillation, which is crucial for pushing the esterification reaction to completion without requiring excessive temperatures that could degrade the product. This method not only achieves a remarkable reaction yield of over 95% but also ensures the final product purity reaches between 97% and 99% through a straightforward workup involving pH adjustment and phase separation. The result is a high-performance agrochemical intermediate that significantly mitigates drift risks while maintaining cost-effectiveness through simplified processing.
Mechanistic Insights into Acid-Catalyzed Esterification
The core of this synthesis lies in the acid-catalyzed Fischer esterification mechanism, where the carboxylic acid group of 2,4-D reacts with the hydroxyl group of isooctyl alcohol. The use of catalysts such as sulfuric acid or sodium pyrosulfate protonates the carbonyl oxygen, increasing the electrophilicity of the carbonyl carbon and facilitating nucleophilic attack by the alcohol. This step is critical for overcoming the kinetic barrier of the reaction, allowing it to proceed at moderate reflux temperatures rather than requiring extreme thermal conditions. The presence of toluene is mechanistically significant as it forms a low-boiling azeotrope with the water byproduct, continuously removing water from the reaction equilibrium and driving the conversion towards the ester product according to Le Chatelier's principle. This continuous dehydration prevents the reverse hydrolysis reaction, ensuring that the conversion of 2,4-D remains high throughout the 2 to 5-hour reaction window specified in the protocol.
Controlling impurities in this process is achieved through precise management of the reaction endpoint and subsequent neutralization steps. The patent specifies monitoring the residual 2,4-dichlorophenoxyacetic acid content, aiming for levels below 1% to 2% before terminating the reaction, which ensures minimal starting material remains in the crude mixture. Following the reaction, the mixture is cooled and treated with a 5% sodium hydroxide solution to adjust the pH to 10, a step designed to neutralize any remaining acid catalyst and unreacted 2,4-D by converting them into water-soluble salts. These salts partition into the aqueous layer during separation, leaving the organic layer enriched with the desired isooctyl ester. This chemical logic ensures that the final distillation step yields a product with high normalization purity, free from acidic contaminants that could destabilize downstream formulations.
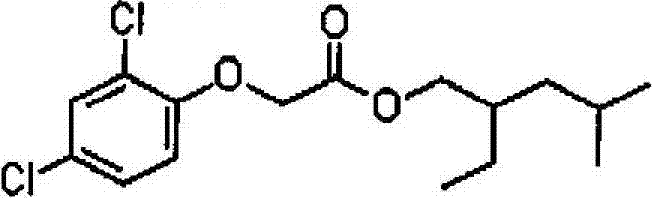
How to Synthesize Isooctyl (2,4-dichlorophenoxy) acetate Efficiently
Implementing this synthesis route requires careful attention to the stoichiometric ratios and thermal conditions to maximize the economic and technical benefits described in the patent. The process begins with the precise weighing of 2,4-dichlorophenoxyacetic acid and isooctyl alcohol, typically in a molar ratio ranging from 1:1 to 1:1.05, to ensure complete conversion of the acid without excessive alcohol waste. The reaction is conducted in a reactor equipped for reflux and water separation, where toluene is added as the entrainer to facilitate the removal of water generated during esterification. Detailed standardized synthesis steps, including specific temperature ramps, catalyst addition rates, and distillation parameters, are essential for reproducing the high yields and purity levels reported in the technical data. For a comprehensive guide on executing this protocol in a GMP-compliant environment, please refer to the structured synthesis guide below.
- Mix 2,4-dichlorophenoxyacetic acid with isooctyl alcohol and toluene solvent in a reactor equipped with a dehydration trap.
- Add a protonic acid catalyst such as sulfuric acid and reflux the mixture for 2 to 5 hours while removing water.
- Cool the reaction, neutralize with sodium hydroxide to pH 10, separate layers, and distill to isolate the final ester product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route offers tangible benefits in terms of cost structure and operational reliability. The high reaction yield, consistently reported above 95%, directly translates to improved raw material utilization, meaning less feedstock is required to produce the same volume of active intermediate. This efficiency reduces the overall cost of goods sold (COGS) by minimizing waste disposal costs and maximizing the throughput of existing reactor capacity. Furthermore, the simplicity of the workup procedure, which relies on standard liquid-liquid separation and distillation rather than complex chromatography or crystallization, lowers the operational expenditure associated with labor and energy consumption. These factors combine to create a more resilient supply chain capable of delivering high-purity agrochemical intermediates at competitive market prices.
- Cost Reduction in Manufacturing: The elimination of complex purification steps and the use of readily available catalysts like sulfuric acid significantly lower the variable costs associated with production. By achieving high conversion rates without the need for expensive transition metal catalysts or specialized reagents, the process ensures that the manufacturing budget is optimized for scale. The ability to recover and reuse the toluene solvent further enhances the economic viability of the route, reducing the recurring cost of raw materials. This qualitative improvement in process efficiency allows manufacturers to offer more competitive pricing structures to their downstream formulators without sacrificing margin.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as toluene, isooctyl alcohol, and sulfuric acid ensures that the supply chain is not vulnerable to the bottlenecks often associated with specialized or scarce reagents. The robustness of the reaction conditions, which tolerate slight variations in temperature and mixing without significant yield loss, adds a layer of operational security to the manufacturing process. This reliability is crucial for maintaining continuous production schedules and meeting the just-in-time delivery requirements of large-scale agrochemical companies. Consequently, partners can expect a steady flow of material with consistent quality, reducing the risk of production stoppages due to raw material shortages or process failures.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from laboratory benchtop experiments to multi-ton commercial production without requiring fundamental changes to the reaction engineering. The use of a closed reflux system with solvent recovery minimizes volatile organic compound (VOC) emissions, aligning with increasingly stringent environmental regulations. Additionally, the aqueous waste stream, primarily containing neutralized salts, is easier to treat compared to waste streams containing heavy metals or complex organic byproducts. This environmental compatibility simplifies the permitting process for new production lines and reduces the long-term liability associated with waste management, making it a sustainable choice for long-term manufacturing strategies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of isooctyl (2,4-dichlorophenoxy) acetate. These insights are derived directly from the patent specifications and are intended to clarify the operational parameters and quality expectations for potential partners. Understanding these details is essential for integrating this intermediate into broader herbicide formulation strategies and ensuring that the final product meets all regulatory and performance standards required by the global market.
Q: Why is isooctyl ester preferred over butyl ester for 2,4-D formulations?
A: Isooctyl esters exhibit significantly lower volatility compared to butyl esters, reducing vapor drift at temperatures as low as 15°C and minimizing crop damage.
Q: What purity levels can be achieved with this specific patent method?
A: The described process utilizing toluene dehydration and precise pH control consistently yields products with normalization purity between 97% and 99%.
Q: Is the catalyst recovery difficult in this synthesis route?
A: The process uses simple acid catalysts and aqueous neutralization steps, allowing for straightforward separation and minimizing complex catalyst recovery costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Isooctyl (2,4-dichlorophenoxy) acetate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of next-generation agrochemical solutions. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from patent data to industrial reality is seamless and efficient. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to verify that every batch of isooctyl (2,4-dichlorophenoxy) acetate meets the exacting standards required for herbicide formulation. Our infrastructure is designed to support the complex chemistry involved in esterification processes, providing a secure and reliable source for your supply chain needs.
We invite you to collaborate with us to optimize your herbicide portfolio with this advanced intermediate. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical constraints. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing you to make informed decisions based on verified performance metrics. By partnering with us, you gain access to a supply chain that prioritizes both technical excellence and commercial value, ensuring your products remain competitive in the global marketplace.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
