Advanced Synthesis of 5-Hydroxy-4-Methyl-2(5H)-Furanone for High-Purity API Manufacturing
Advanced Synthesis of 5-Hydroxy-4-Methyl-2(5H)-Furanone for High-Purity API Manufacturing
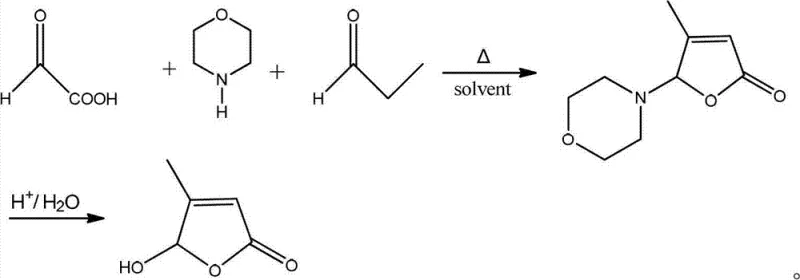
In the competitive landscape of fine chemical intermediates, the efficiency of synthetic routes directly dictates the viability of downstream pharmaceutical applications. Patent CN103570649A introduces a transformative methodology for the production of 5-hydroxy-4-methyl-2(5H)-furanone, a critical building block in the synthesis of various bioactive molecules. This innovation addresses long-standing inefficiencies in heterocyclic chemistry by redefining the role of morpholine from a mere catalyst to a stoichiometric reactant. By leveraging glyoxylic acid hydrate as a superior starting material compared to traditional acetaldehyde sources, this process achieves yields exceeding 90% with exceptional purity profiles. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, understanding the mechanistic advantages of this patent is essential for securing a stable supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 5-hydroxy-4-methyl-2(5H)-furanone has relied on processes described in earlier patents such as WO 2012007814 and JP 10298173. These conventional routes typically utilize acetaldehyde aqueous solutions and propionaldehyde in the presence of morpholine acting solely as a catalyst. While functional, these legacy methods suffer from inherent thermodynamic limitations, often capping yields at approximately 80%. The use of acetaldehyde introduces volatility and handling challenges, while the catalytic nature of morpholine fails to sufficiently drive the equilibrium towards the desired intermediate. Furthermore, the impurity profiles generated in these older processes often necessitate complex purification steps, increasing both the environmental footprint and the overall cost of goods sold for manufacturers attempting cost reduction in API manufacturing.
The Novel Approach
The methodology disclosed in CN103570649A represents a paradigm shift by fundamentally altering the reaction stoichiometry. Instead of relying on morpholine as a catalyst, the new process employs it as a substrate in a molar ratio of 1:(1-2) relative to glyoxylic acid hydrate. This strategic adjustment ensures that the formation of the key intermediate is thermodynamically favored, effectively pushing the reaction to completion. The substitution of acetaldehyde with glyoxylic acid hydrate provides a more stable and reactive carbonyl source, facilitating a cleaner condensation with propionaldehyde. This novel approach not only boosts yields to over 90% but also simplifies the downstream processing requirements, making it an ideal candidate for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Morpholine-Mediated Cyclization
The core of this synthetic breakthrough lies in the unique interaction between glyoxylic acid, morpholine, and propionaldehyde. In the initial stage, glyoxylic acid hydrate reacts with morpholine at low temperatures (0-5°C) to form a stable hemiaminal or aminal intermediate. This species acts as a masked aldehyde equivalent, preventing uncontrolled polymerization often seen with free aldehydes. Upon the addition of propionaldehyde and subsequent heating to 40-80°C, an aldol-type condensation occurs, constructing the carbon skeleton required for the furanone ring. The precise control of temperature during this phase is critical for minimizing side reactions and ensuring the regioselectivity of the bond formation, which is paramount for maintaining high optical and chemical purity in the final product.
Following the condensation, the reaction mixture undergoes vacuum concentration to remove solvents and excess volatile components, preparing the system for the crucial cyclization step. The addition of hydrochloric acid (6-10 mol/L) triggers the hydrolysis of the morpholine moiety and the subsequent intramolecular esterification to close the lactone ring. Heating the mixture to 70-90°C facilitates this dehydration and cyclization, releasing the morpholine and yielding the target 5-hydroxy-4-methyl-2(5H)-furanone. This acid-mediated step is highly efficient, converting the linear precursor into the cyclic furanone with minimal byproduct formation, thereby validating the robustness of this route for high-purity furanone derivatives production.
How to Synthesize 5-Hydroxy-4-Methyl-2(5H)-Furanone Efficiently
Implementing this synthesis requires strict adherence to the temperature profiles and stoichiometric ratios outlined in the patent to maximize efficiency. The process begins with the dissolution of glyoxylic acid hydrate in a polar protic solvent such as ethanol, followed by the controlled addition of morpholine under cooling to manage the exotherm. Once the intermediate is formed, propionaldehyde is introduced, and the system is heated to promote condensation. After the reaction reaches completion, the solvent is removed, and the residue is treated with hydrochloric acid to effect cyclization. The detailed standardized synthesis steps see the guide below for exact operational parameters.
- Dissolve glyoxylic acid hydrate in an organic solvent like ethanol and react with morpholine at 0-5°C to form the initial intermediate complex.
- Add propionaldehyde to the mixture and heat to 40-80°C for 4-6 hours to facilitate the condensation reaction.
- Concentrate the solution under vacuum, add hydrochloric acid solution, and heat to 70-90°C for cyclization and hydrolysis to obtain the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers substantial strategic benefits beyond simple yield improvements. The elimination of inefficient catalytic cycles and the use of readily available, stable raw materials like glyoxylic acid hydrate significantly de-risk the supply chain. By moving away from volatile acetaldehyde solutions, storage and handling costs are reduced, and safety protocols are simplified. This process optimization translates directly into a more resilient supply network, capable of meeting the rigorous demands of global pharmaceutical clients without the bottlenecks associated with older, less efficient technologies.
- Cost Reduction in Manufacturing: The most immediate financial impact stems from the dramatic increase in reaction yield, which moves from a historical average of 80% to over 90%. This improvement means that for every ton of raw material purchased, the output of saleable product increases significantly, effectively lowering the unit cost of production. Additionally, by utilizing morpholine as a recoverable substrate rather than a lost catalyst, and employing common solvents like ethanol, the overall material costs are optimized. The simplified purification process further reduces energy consumption and waste disposal fees, contributing to a leaner manufacturing cost structure.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as glyoxylic acid, morpholine, and propionaldehyde ensures that raw material sourcing is not dependent on niche suppliers. This abundance of feedstock guarantees continuity of supply even during market fluctuations. Furthermore, the robustness of the reaction conditions—operating at moderate temperatures and pressures—reduces the risk of batch failures due to equipment sensitivity. This reliability allows for consistent lead times, enabling downstream partners to plan their inventory levels with greater confidence and reducing the need for excessive safety stock.
- Scalability and Environmental Compliance: From an environmental perspective, this method aligns well with green chemistry principles by improving atom economy and reducing waste generation. The high selectivity of the reaction minimizes the formation of hazardous byproducts, simplifying wastewater treatment and effluent management. The process is inherently scalable, as demonstrated by the successful execution of the reaction in standard laboratory glassware which translates linearly to industrial reactors. This scalability ensures that as demand for high-purity pharmaceutical intermediates grows, production capacity can be expanded rapidly without requiring fundamental re-engineering of the process.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of 5-hydroxy-4-methyl-2(5H)-furanone. These answers are derived directly from the experimental data and claims presented in patent CN103570649A, providing a transparent view of the technology's capabilities. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this intermediate into their own synthesis pipelines.
Q: What is the primary advantage of using morpholine as a substrate in this synthesis?
A: Unlike conventional methods where morpholine acts merely as a catalyst, this patent utilizes morpholine as a stoichiometric substrate (molar ratio 1:1 to 1:2). This shift significantly drives the reaction equilibrium forward, resulting in higher yields (over 90%) compared to the traditional 80% yield.
Q: How does this method improve impurity control compared to prior art?
A: By utilizing glyoxylic acid hydrate instead of acetaldehyde aqueous solution, the reaction pathway becomes more controlled. The specific temperature staging (0-5°C initially, then 40-80°C) minimizes side reactions and polymerization, leading to crude product purities exceeding 98% before final distillation.
Q: Is this process scalable for industrial production?
A: Yes, the process relies on common solvents like ethanol and standard unit operations such as vacuum concentration and distillation. The elimination of exotic catalysts and the use of robust reaction conditions make it highly suitable for commercial scale-up from kilogram to multi-ton batches.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Hydroxy-4-Methyl-2(5H)-Furanone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the quality of your final API depends heavily on the purity and consistency of your starting materials. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patent CN103570649A are fully realized in our manufacturing facilities. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 5-hydroxy-4-methyl-2(5H)-furanone meets the highest international standards. Our commitment to technical excellence allows us to deliver intermediates that facilitate smoother downstream processing for our global partners.
We invite you to collaborate with us to optimize your supply chain and reduce your overall manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our advanced synthesis capabilities can support your long-term business goals.
