Advanced One-Pot Synthesis of 7-Hydroxy-1-Tetralone: A Breakthrough for Industrial Pharmaceutical Manufacturing
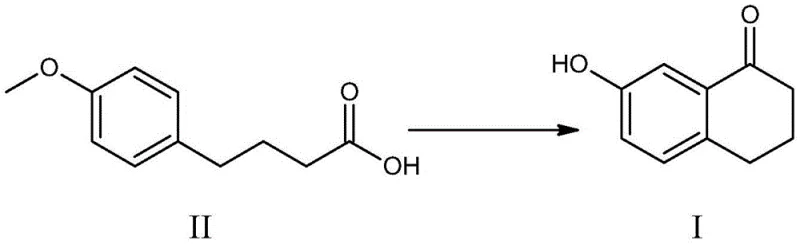
The pharmaceutical industry continuously seeks robust synthetic pathways for critical fused-ring intermediates, particularly those serving as scaffolds for complex active pharmaceutical ingredients (APIs). A pivotal advancement in this domain is detailed in Chinese Patent CN106977377B, which discloses a highly efficient preparation method for 7-hydroxy-1-tetralone (CAS: 22009-38-7). This molecule serves as an indispensable building block for synthesizing bioactive compounds such as prostaglandins and antiviral agents like Wipatasvir. The patented technology introduces a novel one-pot strategy that utilizes Lewis acid catalysis to simultaneously effect ring closure and demethylation, starting from 4-(4-methoxyphenyl)butyric acid. This approach represents a significant departure from legacy methodologies, addressing long-standing challenges related to environmental impact, operational complexity, and product quality. For global procurement and R&D teams, understanding the mechanistic elegance and commercial viability of this route is essential for securing a reliable pharmaceutical intermediate supplier capable of meeting stringent regulatory standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 7-hydroxy-1-tetralone has been plagued by inefficient multi-step sequences or hazardous reaction conditions that hinder scalable manufacturing. One prevalent conventional pathway involves the initial cyclization of 4-(4-methoxyphenyl)butyric acid using polyphosphoric acid (PPA) or phosphorus pentoxide to form 7-methoxy-1-tetralone, followed by a separate demethylation step. As illustrated in the reaction scheme below, this reliance on phosphorus-based reagents generates substantial quantities of phosphorus-containing wastewater, creating severe environmental burdens and escalating waste treatment costs for manufacturers.

Furthermore, alternative strategies attempting direct hydroxylation of 1-tetralone using superacids like fluorine antimonic acid have demonstrated poor selectivity. Literature indicates that such methods often result in conversion rates as low as 30%, accompanied by the formation of numerous isomers and dihydroxy byproducts. The corrosive nature of superacids also imposes extreme demands on reactor materials and operational safety protocols, rendering these processes economically unfeasible for large-scale cost reduction in pharma manufacturing. These legacy limitations necessitate a paradigm shift towards greener, more atom-economical solutions.
The Novel Approach
The methodology described in CN106977377B elegantly circumvents these obstacles by employing a tandem cyclization-demethylation sequence mediated by Lewis acids such as aluminum trichloride or aluminum tribromide. By utilizing 4-(4-methoxyphenyl)butyric acid as the starting material, the process achieves the target structure in a single reactor vessel. This consolidation of steps not only minimizes solvent usage and energy consumption but also inherently improves the overall mass balance of the production line. The elimination of phosphorus reagents ensures that the effluent profile is significantly cleaner, aligning with modern green chemistry principles. Moreover, the reaction conditions are milder compared to superacid protocols, allowing for the use of standard glass-lined or stainless steel equipment, thereby enhancing the commercial scale-up of complex fused ring intermediates.
Mechanistic Insights into Lewis Acid-Catalyzed Cyclization and Demethylation
The core innovation of this process lies in the dual functionality of the Lewis acid catalyst, which orchestrates two distinct chemical transformations concurrently. Initially, the Lewis acid activates the carboxylic acid moiety of the starting material, facilitating an intramolecular Friedel-Crafts acylation that constructs the tetralone skeleton. Subsequently, under the sustained thermal conditions and acidic environment, the methoxy ether linkage undergoes cleavage. This demethylation step is critical, as it directly installs the phenolic hydroxyl group at the 7-position without requiring isolation of the methoxy-intermediate. The synergy between the cyclization and ether cleavage events is driven by the strong electrophilic character of the aluminum species, which coordinates with oxygen atoms to lower activation energies for both bond formation and bond breaking events.
From an impurity control perspective, this mechanism offers superior selectivity compared to oxidative hydroxylation routes. The electrophilic aromatic substitution is directed by the existing alkyl chain, ensuring regioselective ring closure. Unlike radical-based oxidation methods that generate random hydroxylation patterns and over-oxidized byproducts, the Lewis acid pathway preserves the integrity of the aromatic system while precisely modifying the substitution pattern. This inherent selectivity translates to a crude product profile that is remarkably clean, simplifying downstream purification. The ability to achieve purity levels exceeding 99.9% directly after crystallization underscores the robustness of this mechanistic design, providing R&D directors with confidence in the consistency of the high-purity API intermediate supply.
How to Synthesize 7-Hydroxy-1-Tetralone Efficiently
The practical implementation of this synthesis involves dissolving the Lewis acid catalyst in a suitable aromatic solvent such as toluene or xylene, followed by the addition of the 4-(4-methoxyphenyl)butyric acid precursor. The mixture is heated to reflux temperatures ranging between 60°C and 120°C for a duration of 8 to 20 hours. Upon completion, the reaction is quenched into ice water, and the product is isolated through pH-controlled extraction and recrystallization. The detailed standardized operating procedures, including specific molar ratios and safety precautions for handling Lewis acids, are outlined below.
- Preparation of Precursor: Synthesize 4-(4-methoxyphenyl)butyric acid (Formula II) typically via reduction of the corresponding keto-acid using Palladium on Carbon in acetic acid.
- One-Pot Cyclization and Demethylation: React the precursor with a Lewis acid catalyst (such as AlCl3 or AlBr3) in a solvent like toluene or xylene at elevated temperatures (60-120°C) to simultaneously close the ring and remove the methyl group.
- Workup and Purification: Quench the reaction mixture into ice water, separate layers, adjust pH to isolate the product, and recrystallize from methanol/water to achieve high purity (>99.9%).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this Lewis acid-mediated route offers tangible strategic benefits that extend beyond mere chemical yield. The primary advantage stems from the drastic simplification of the process flow. By collapsing what was traditionally a two-step sequence (cyclization followed by demethylation) into a single pot, manufacturers can significantly reduce cycle times and labor costs. This operational efficiency directly contributes to reducing lead time for high-purity pharmaceutical intermediates, allowing for more responsive inventory management and faster time-to-market for downstream drug products. Additionally, the avoidance of hazardous superacids and phosphorus sludge mitigates regulatory risks associated with waste disposal.
- Cost Reduction in Manufacturing: The elimination of phosphorus-containing reagents removes the need for expensive and complex wastewater treatment infrastructure dedicated to phosphate removal. Furthermore, the one-pot nature of the reaction reduces solvent consumption and energy usage associated with intermediate isolation and drying. These factors collectively drive down the variable cost of goods sold (COGS), enabling more competitive pricing structures for bulk purchasers without compromising on quality margins.
- Enhanced Supply Chain Reliability: The starting material, 4-(4-methoxyphenyl)butyric acid, is readily accessible and can be synthesized via straightforward hydrogenation of commercially available keto-acids. This ensures a stable upstream supply base, minimizing the risk of raw material shortages. The robustness of the Lewis acid chemistry also means that the process is less sensitive to minor fluctuations in reaction parameters, ensuring consistent batch-to-batch output and reliable delivery schedules for long-term contracts.
- Scalability and Environmental Compliance: The process operates under conditions compatible with standard industrial reactors, avoiding the specialized corrosion-resistant alloys required for superacid chemistry. This facilitates seamless technology transfer from pilot plant to full commercial production scales. Moreover, the absence of persistent organic pollutants and heavy phosphorus loads in the effluent simplifies environmental permitting and ongoing compliance monitoring, safeguarding the manufacturer's social license to operate.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of 7-hydroxy-1-tetralone using this advanced methodology. These insights are derived directly from the experimental data and comparative examples provided in the patent literature, offering clarity on yield expectations, catalyst selection, and purity profiles.
Q: Why is the Lewis acid one-pot method superior to traditional Polyphosphoric Acid (PPA) routes?
A: Traditional PPA routes generate significant phosphorus-containing wastewater which is difficult and costly to treat. The novel Lewis acid method eliminates phosphorus reagents entirely, drastically reducing environmental compliance costs and simplifying waste management.
Q: What are the yield and purity advantages of this new synthesis route?
A: According to patent data, this method achieves yields exceeding 85% and purity levels greater than 99.9%. This contrasts sharply with direct hydroxylation methods which often suffer from low conversion (around 30%) and the formation of difficult-to-separate isomers.
Q: Which Lewis acid catalysts are most effective for this transformation?
A: The patent identifies Aluminum Trichloride (AlCl3) and Aluminum Tribromide (AlBr3) as the preferred catalysts. These reagents effectively promote both the intramolecular Friedel-Crafts acylation and the subsequent ether cleavage in a single operational step.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 7-Hydroxy-1-Tetralone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of your drug development programs. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with precision and speed. We adhere to stringent purity specifications and utilize rigorous QC labs to verify that every batch of 7-hydroxy-1-tetralone meets the exacting standards required for pharmaceutical applications, leveraging the efficient Lewis acid chemistry described in recent patents to optimize our manufacturing footprint.
We invite you to engage with our technical procurement team to discuss how our optimized synthesis routes can benefit your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper visibility into how our process efficiencies translate into value for your organization. Please contact us today to obtain specific COA data and route feasibility assessments tailored to your supply chain objectives.
