Advancing Chiral Synthesis: Direct Asymmetric Hydrogenation of Enenoic Acids for Commercial Scale-up
Advancing Chiral Synthesis: Direct Asymmetric Hydrogenation of Enenoic Acids for Commercial Scale-up
The landscape of chiral intermediate manufacturing is constantly evolving, driven by the need for more efficient, cost-effective, and environmentally sustainable synthetic routes. A significant breakthrough in this domain is documented in patent CN111410604B, which discloses a novel method for the asymmetric hydrogenation of enenoic acid compounds. This technology addresses a longstanding challenge in organic synthesis: the direct enantioselective reduction of carbon-carbon double bonds in the presence of free carboxylic acid functionalities. Traditionally, the presence of acidic protons has complicated transition metal catalysis, often necessitating protective group strategies that add steps, cost, and waste to the process. The disclosed invention utilizes specialized iridium-phosphine oxazoline complexes to overcome these barriers, enabling the direct conversion of prochiral enenoic acids into valuable chiral building blocks with high stereocontrol. For R&D directors and process chemists, this represents a pivotal shift towards more atom-economical pathways for generating high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
In traditional synthetic workflows targeting chiral saturated acids, chemists have historically been forced to employ indirect strategies due to the incompatibility of free carboxylic acids with many homogeneous hydrogenation catalysts. The standard approach involves a multi-step sequence: first, the enenoic acid must be converted into an ester derivative, such as a methyl or ethyl ester, to mask the acidic proton and prevent catalyst deactivation or salt formation. Following the asymmetric hydrogenation of the ester, a subsequent hydrolysis step is required to regenerate the free carboxylic acid functionality. This detour not only increases the overall material cost due to the consumption of additional reagents for esterification and hydrolysis but also significantly impacts the environmental footprint through increased solvent usage and waste generation. Furthermore, each additional unit operation introduces potential yield losses and impurities, complicating the purification profile and extending the total production timeline, which is a critical bottleneck for supply chain efficiency in the fast-paced pharmaceutical industry.
The Novel Approach
The methodology presented in patent CN111410604B circumvents these inefficiencies by enabling the direct asymmetric hydrogenation of the free acid form. By employing a tailored iridium-phosphine oxazoline catalyst system, the process leverages the unique coordination chemistry between the metal center and the carboxyl group itself. Rather than viewing the acid group as a liability, this approach utilizes it as a directing group to enhance stereo-induction. As illustrated in the general reaction scheme below, the transformation proceeds smoothly from the unsaturated precursor to the chiral product without the need for protection or deprotection steps.
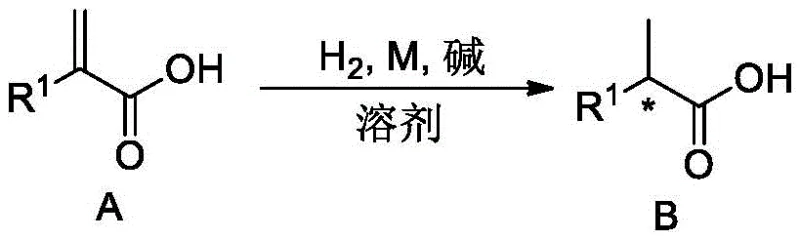
This streamlined approach drastically reduces the step count, effectively collapsing a three-step sequence into a single catalytic event. For procurement managers, this simplification translates directly into reduced raw material inventory requirements and lower utility costs associated with running fewer reactors. The ability to access chiral acids directly from readily available enenoic acid precursors opens new avenues for cost reduction in pharmaceutical intermediates manufacturing, allowing companies to optimize their supply chains by eliminating entire stages of chemical processing while maintaining rigorous quality standards.
Mechanistic Insights into Iridium-Phosphine Oxazoline Catalyzed Hydrogenation
The success of this transformation hinges on the precise structural design of the iridium-phosphine oxazoline complex. The catalyst features a chiral ligand framework that creates a well-defined steric environment around the iridium center, which is crucial for discriminating between the two faces of the prochiral double bond. Unlike simple phosphine ligands, the phosphine-oxazoline hybrid structure offers a unique combination of electronic and steric properties that stabilize the active catalytic species. The core innovation lies in the interaction between the cationic iridium complex and the anionic carboxylate formed in situ upon addition of a base. This ion-pairing interaction positions the substrate in a rigid conformation relative to the chiral pocket of the catalyst, ensuring that hydrogen delivery occurs with high facial selectivity.
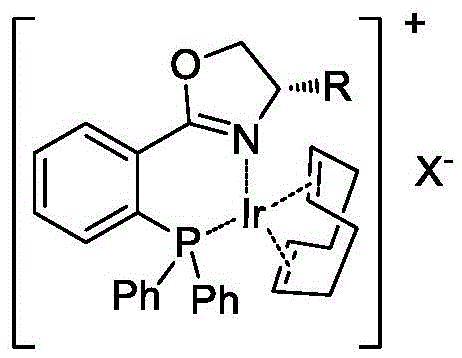
From an impurity control perspective, this mechanism is highly advantageous. The strong coordination prevents non-selective background reduction or isomerization side reactions that often plague less sophisticated catalytic systems. The patent data indicates that specific variants of the catalyst, such as those with bulky substituents on the oxazoline ring, can achieve enantiomeric excess (ee) values exceeding 90 percent. For R&D teams focused on regulatory compliance, this high level of stereochemical purity minimizes the burden on downstream crystallization or chromatographic purification steps. The robustness of the catalyst system across a range of substrates, including those with biphenyl and benzophenone motifs, suggests a versatile platform technology capable of supporting the synthesis of diverse drug candidates without requiring extensive re-optimization for each new molecule.
How to Synthesize Chiral Enenoic Acid Derivatives Efficiently
Implementing this technology requires careful attention to reaction parameters to maximize both conversion and enantioselectivity. The general protocol involves charging the enenoic acid substrate and the iridium catalyst into a reaction vessel under an inert atmosphere, typically nitrogen or argon, to prevent oxidation of the sensitive metal center. A stoichiometric amount of base, such as triethylamine, is added to generate the active carboxylate species, followed by the introduction of a protic solvent like methanol which aids in hydrogen activation. The reaction is then pressurized with hydrogen gas and heated to moderate temperatures. Detailed operational parameters, including specific catalyst loadings and workup procedures for maximizing yield, are critical for successful scale-up.
- Prepare the reaction vessel under inert atmosphere and charge with the enenoic acid substrate and the specific iridium-phosphine oxazoline catalyst at a substrate-to-catalyst ratio of approximately 400: 1.
- Add an organic base such as triethylamine (0.5 equivalents) and a protic solvent like methanol to facilitate the catalytic cycle and solubilize the carboxylic acid substrate.
- Pressurize the reactor with hydrogen gas to 10 atm and maintain the temperature between 20°C and 50°C for 18 hours to achieve high conversion and enantioselectivity.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders managing the commercial viability of chemical processes, the adoption of this direct hydrogenation technology offers compelling strategic benefits. The elimination of esterification and hydrolysis steps fundamentally alters the cost structure of producing chiral acids. By removing two distinct unit operations, manufacturers can significantly reduce capital expenditure requirements, as fewer reactors and separation units are needed to achieve the same output volume. This consolidation of the process flow also enhances operational safety by reducing the handling of hazardous reagents often associated with esterification, such as thionyl chloride or strong mineral acids used in hydrolysis. Consequently, the overall risk profile of the manufacturing campaign is lowered, aligning with modern ESG (Environmental, Social, and Governance) goals.
- Cost Reduction in Manufacturing: The most immediate impact is seen in the reduction of variable costs. Without the need for protecting group chemistry, the consumption of auxiliary reagents drops precipitously. The atom economy of the reaction is superior because every atom of the starting material ends up in the final product, minus only the hydrogen added. This efficiency means that for every kilogram of product produced, less waste is generated, leading to substantial cost savings in waste disposal and treatment. Furthermore, the high selectivity of the catalyst reduces the loss of valuable chiral material to unwanted stereoisomers, ensuring that the theoretical yield is closely approached in practice, which optimizes the return on investment for expensive starting materials.
- Enhanced Supply Chain Reliability: Simplifying the synthetic route inherently makes the supply chain more robust. Fewer steps mean fewer points of failure where a reaction could stall or produce off-spec material. The catalysts described are stable and effective at relatively low loadings, reducing dependency on large volumes of precious metal resources. This efficiency ensures that production schedules are more predictable and less susceptible to delays caused by complex multi-step purifications. For supply chain heads, this reliability translates to shorter lead times for high-purity pharmaceutical intermediates, allowing for more agile responses to market demand fluctuations and reducing the need for excessive safety stock inventory.
- Scalability and Environmental Compliance: The reaction conditions described, utilizing moderate hydrogen pressures and temperatures, are well-suited for transfer from laboratory to pilot and commercial scales. The use of common solvents like methanol and ethanol facilitates solvent recovery and recycling, further enhancing the green chemistry profile of the process. The reduction in solvent usage and waste generation simplifies regulatory compliance regarding environmental emissions. As regulatory bodies worldwide tighten restrictions on pharmaceutical manufacturing waste, adopting such streamlined, high-efficiency processes positions companies favorably for long-term operational continuity and minimizes the risk of regulatory bottlenecks.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this asymmetric hydrogenation technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for evaluating its applicability to specific project needs. Understanding these nuances is essential for technical teams assessing the feasibility of integrating this method into existing production pipelines.
Q: What is the primary advantage of using iridium-phosphine oxazoline complexes for enenoic acid hydrogenation?
A: The primary advantage is the ability to directly hydrogenate free carboxylic acids without prior esterification, leveraging strong coordination between the catalyst and the carboxyl group to achieve high enantioselectivity (up to 92% ee) and simplify the synthetic route.
Q: What are the typical reaction conditions for this asymmetric hydrogenation process?
A: The process typically operates under mild conditions, utilizing hydrogen pressures around 10 atm, temperatures ranging from room temperature to 50°C, and common organic solvents like methanol with a catalytic amount of organic base.
Q: Can this catalytic system accommodate diverse substrate structures?
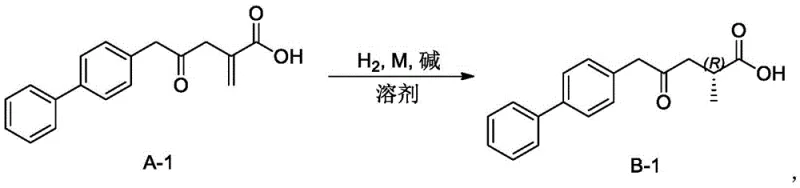
A: Yes, the catalytic system demonstrates broad substrate scope, effectively processing enenoic acids with various R1 groups including phenyl, biphenyl, substituted phenyl, and benzophenone moieties, making it versatile for different pharmaceutical intermediate syntheses.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Enenoic Acids Supplier
The technological advancements detailed in CN111410604B highlight the critical importance of advanced catalytic solutions in modern chemical synthesis. At NINGBO INNO PHARMCHEM, we recognize that translating such innovative laboratory protocols into reliable commercial supply requires deep expertise and infrastructure. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped with state-of-the-art high-pressure hydrogenation reactors and stringent purity specifications are maintained through our rigorous QC labs, ensuring that every batch of chiral intermediate meets the exacting standards required by global pharmaceutical clients.
We invite you to leverage our technical capabilities to accelerate your development timelines. Whether you require custom synthesis of complex enenoic acid derivatives or optimization of existing hydrogenation processes, our team is ready to assist. Please contact our technical procurement team to request a Customized Cost-Saving Analysis for your specific project. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing excellence can drive value for your organization.
