Scalable Synthesis of 3-Hydroxy-4-Methoxy-2-Nitrobenzoic Acid for PBD Antitumor Agents
Introduction to Advanced PBD Intermediate Synthesis
The development of potent antitumor agents often hinges on the availability of high-purity, structurally complex intermediates. Among these, Pyrrolobenzodiazepines (PBDs) represent a class of DNA-binding antibiotics with significant therapeutic potential, first identified in the mid-20th century. The compound 3-hydroxy-4-methoxy-2-nitrobenzoic acid serves as a critical building block for synthesizing these bioactive PBD analogs. Recent intellectual property, specifically patent CN111302945B, discloses a groundbreaking preparation method that addresses long-standing challenges in yield and purity associated with this key intermediate. This technical breakthrough offers a robust pathway for pharmaceutical manufacturers seeking to optimize their supply chains for oncology drug candidates.
The significance of this innovation lies in its ability to transform a previously inefficient synthetic sequence into a streamlined, industrially viable process. Traditional methods for accessing similar benzoic acid derivatives often suffer from poor regioselectivity during nitration and difficult purification profiles. The new methodology described in the patent leverages a strategic sequence of nitration, functional group manipulation, and selective deprotection to achieve superior outcomes. By starting from readily available 3-alkoxy-4-acetoxybenzaldehyde, the process ensures a stable and reproducible entry point for large-scale production.
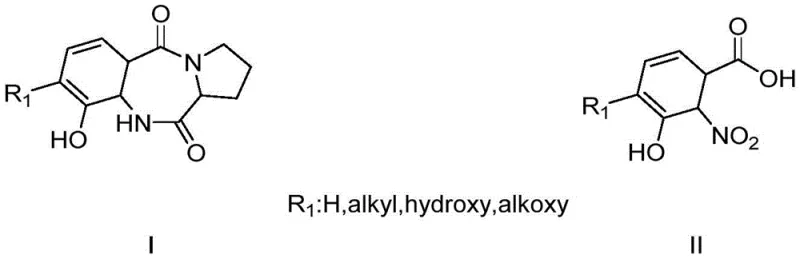
As illustrated in the structural context, the target molecule corresponds to the general Formula II found in PBD synthesis, where precise substitution patterns are mandatory for biological activity. The ability to reliably produce this intermediate with high fidelity directly impacts the quality and efficacy of the final antitumor antibiotic. For R&D directors and procurement specialists, understanding the nuances of this new synthetic route is essential for evaluating potential suppliers and securing long-term material availability for clinical and commercial programs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the innovations detailed in patent CN111302945B, the synthesis of 3-hydroxy-4-methoxy-2-nitrobenzoic acid was plagued by significant inefficiencies that hindered commercial scalability. Existing literature and traditional routes typically reported a total yield of merely around 24%, which is economically unsustainable for high-volume API manufacturing. These conventional pathways often involved multiple discrete isolation steps, each contributing to material loss and increased operational complexity. Furthermore, the control of regioisomers during the introduction of the nitro group was historically problematic, leading to difficult-to-separate impurities that compromised the purity profile required for pharmaceutical applications.
The reliance on harsh conditions or non-selective reagents in older methods frequently resulted in the degradation of sensitive functional groups, such as the aldehyde moiety present in early intermediates. This necessitated extensive downstream purification, often involving preparative chromatography, which is cost-prohibitive at the metric-ton scale. Additionally, the environmental footprint of these legacy processes was substantial, generating significant waste streams due to low atom economy and the use of stoichiometric oxidants without efficient recovery systems. For supply chain managers, these factors translated into volatile pricing, extended lead times, and a heightened risk of supply disruption for critical oncology intermediates.
The Novel Approach
In stark contrast to historical precedents, the novel approach disclosed in the patent achieves a total yield exceeding 50%, representing a more than twofold improvement in efficiency. This dramatic enhancement is achieved through a meticulously designed six-step sequence (conceptually grouped into three main stages) that maximizes atom economy and minimizes handling losses. The process initiates with the nitration of 3-alkoxy-4-acetoxybenzaldehyde to form a nitro-acetate intermediate, followed by a telescoped sequence of deacetylation, methylation, and oxidation. This consolidation of steps reduces the number of unit operations, thereby lowering labor costs and equipment occupancy time.
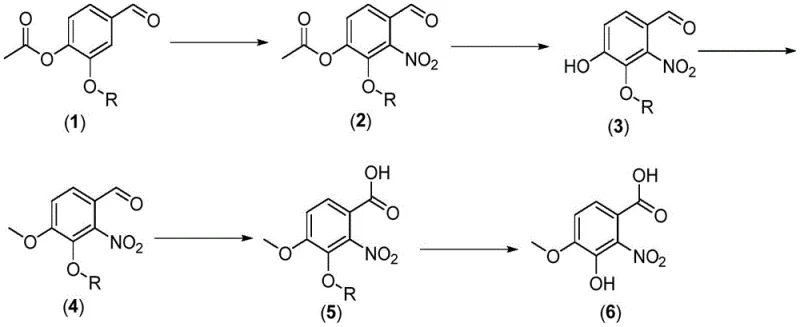
Crucially, the new method incorporates a highly effective purification strategy that relies on recrystallization rather than chromatography. By utilizing a specific mixed solvent system of isopropanol and water in the final step, the process successfully removes isomeric impurities that persist from the initial nitration stage. This capability to purge impurities through crystallization is a hallmark of a robust industrial process, ensuring that the final product meets stringent purity specifications (>99.0% by HPLC) without the need for expensive resin-based purification. The result is a cost-reduction in pharmaceutical intermediate manufacturing that is both significant and sustainable, aligning perfectly with the goals of modern green chemistry initiatives.
Mechanistic Insights into Regioselective Nitration and Oxidation
The cornerstone of this synthetic success lies in the precise control of the nitration reaction, which dictates the substitution pattern of the aromatic ring. When 3-alkoxy-4-acetoxybenzaldehyde is subjected to nitration using concentrated or fuming nitric acid, the reaction is highly exothermic and requires strict temperature control between -20°C and 5°C. Under these conditions, the electrophilic aromatic substitution occurs primarily at the position ortho to the acetoxy group, yielding the desired 2-nitro isomer. However, the mechanism inevitably produces a proportion of the undesired isomer (designated as 2A in the patent), typically ranging from 8% to 20%. Understanding the formation of this byproduct is vital for R&D teams aiming to replicate the process.
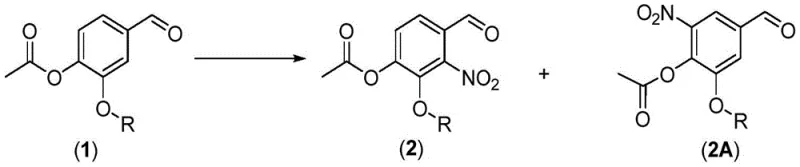
The presence of the isomer 2A poses a challenge, but the novelty of this patent is that it does not attempt to separate this impurity immediately. Instead, the process design allows the isomer to proceed through the subsequent hydrolysis and methylation steps. It is only in the final deprotection and recrystallization stage that the structural differences between the target molecule and the isomer are exploited for separation. This "carry-through" strategy avoids the yield penalties associated with early-stage purification of difficult isomers. Furthermore, the oxidation step, utilizing potassium permanganate or sodium permanganate under alkaline conditions, efficiently converts the aldehyde functionality to the carboxylic acid without affecting the nitro group or the methoxy substituents, demonstrating excellent chemoselectivity.
Impurity control is further enhanced by the choice of solvents and reagents in the deprotection step. The use of organic bases like triethylamine coupled with lithium salts (e.g., lithium bromide) facilitates the selective cleavage of the specific alkoxy group at the 3-position while leaving the 4-methoxy group intact. Alternatively, boron trichloride can be employed at low temperatures (0-15°C) to achieve similar selectivity. This mechanistic precision ensures that the final product is not contaminated with demethylated byproducts at the wrong position, which would be structurally analogous and extremely difficult to remove. The rigorous control over these reaction parameters underscores the sophistication of the process design.
How to Synthesize 3-Hydroxy-4-Methoxy-2-Nitrobenzoic Acid Efficiently
The synthesis of this high-value intermediate is executed through a logical progression of chemical transformations that balance reactivity with selectivity. The process begins with the careful addition of the starting aldehyde to a nitrating agent in a chlorinated solvent, followed by quenching into ice water to isolate the crude nitro-acetate. Subsequent steps involve treating this intermediate with an inorganic base to remove the acetyl protecting group, followed immediately by methylation using agents such as dimethyl sulfate or methyl iodide. The resulting aldehyde is then oxidized to the acid, and finally, the specific alkoxy group is cleaved to reveal the phenolic hydroxyl group. Detailed standardized synthesis steps see the guide below.
- Nitration of 3-alkoxy-4-acetoxybenzaldehyde using nitric acid at low temperature (-20 to 5°C) to form the nitro-intermediate.
- One-pot conversion involving deacetylation with base, methylation with dimethyl sulfate, and oxidation with permanganate.
- Selective deprotection of the alkoxy group using organic base/lithium salt or boron trichloride, followed by recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route offers transformative benefits that extend beyond mere technical metrics. The primary advantage is the substantial reduction in production costs driven by the doubling of overall yield compared to legacy methods. By minimizing material waste and maximizing the output per batch, manufacturers can offer significantly more competitive pricing for this critical intermediate. This cost efficiency is compounded by the use of commodity chemicals—such as nitric acid, potassium hydroxide, and potassium permanganate—which are readily available in the global market, reducing the risk of raw material shortages that often plague specialized syntheses.
- Cost Reduction in Manufacturing: The elimination of complex chromatographic purification steps in favor of simple filtration and recrystallization drastically lowers the operational expenditure (OPEX) associated with production. Chromatography resins are expensive consumables that require frequent replacement and generate significant solvent waste; replacing this with crystallization reduces both direct material costs and waste disposal fees. Furthermore, the ability to telescope multiple reaction steps (deacetylation, methylation, oxidation) into a single pot or sequential addition without isolation reduces labor hours and energy consumption, leading to a leaner and more cost-effective manufacturing profile.
- Enhanced Supply Chain Reliability: The robustness of this process ensures consistent batch-to-batch quality, which is paramount for maintaining regulatory compliance in the pharmaceutical sector. Because the purification relies on physical properties (solubility) rather than subtle chemical interactions, the process is less susceptible to minor variations in reagent quality or environmental conditions. This stability translates to reliable delivery schedules and reduced lead times for high-purity pharmaceutical intermediates, allowing downstream API manufacturers to plan their production cycles with greater confidence and lower safety stock requirements.
- Scalability and Environmental Compliance: The process is explicitly designed for industrial mass production, having been validated from kilogram to potential multi-ton scales. The use of standard reactors and the avoidance of exotic catalysts mean that the technology can be transferred to existing multipurpose facilities without major capital investment. Additionally, the improved yield inherently reduces the environmental footprint per kilogram of product, aligning with increasingly strict global regulations on chemical manufacturing emissions and waste generation, thus future-proofing the supply chain against regulatory tightening.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of 3-hydroxy-4-methoxy-2-nitrobenzoic acid. These answers are derived directly from the experimental data and process descriptions found in the underlying patent literature, providing a transparent view of the technology's capabilities and limitations for potential partners.
Q: How is the isomeric impurity controlled in this synthesis?
A: The process generates an isomer during the initial nitration step (approx. 8-20%). This isomer is carried through subsequent steps and finally removed via selective recrystallization using an isopropanol/water solvent system, achieving >99% purity.
Q: What are the key advantages of this route over conventional methods?
A: This novel route improves the total yield from approximately 24% in existing methods to over 50%. It also simplifies operations by allowing consecutive steps (deacetylation, methylation, oxidation) to be potentially combined under alkaline conditions.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the patent explicitly states the method is suitable for industrial mass production. It uses common solvents (DCM, Methanol) and reagents (KMnO4, KOH) and features robust purification via recrystallization rather than complex chromatography.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Hydroxy-4-Methoxy-2-Nitrobenzoic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful development of next-generation antitumor therapies depends on the secure supply of high-quality intermediates. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met whether you are in preclinical development or full-scale commercialization. Our commitment to quality is unwavering, supported by stringent purity specifications and rigorous QC labs that utilize advanced analytical techniques to verify the identity and purity of every batch we produce.
We invite you to collaborate with us to leverage this advanced synthetic technology for your PBD programs. Our technical team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this optimized route can improve your project economics. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how we can become your trusted partner in delivering complex pharmaceutical intermediates.
