Advanced Synthesis of 2-(3,4-Dimethoxyphenyl)ethylamine: A Safer, Scalable Route for Dopamine Production
The pharmaceutical industry continuously seeks robust synthetic pathways for critical cardiovascular agents, and the preparation of dopamine hydrochloride stands as a prime example of this ongoing optimization challenge. Patent CN114014766A introduces a significant technological advancement in the synthesis of the key precursor, 2-(3,4-dimethoxyphenyl)ethylamine, by leveraging a modified Houben-Hoesch reaction strategy. This innovative approach addresses long-standing safety and cost inefficiencies inherent in legacy manufacturing processes, offering a streamlined alternative that relies on readily available commodity chemicals rather than specialized, hazardous reagents. By shifting the synthetic paradigm away from explosive nitro-compounds and toxic cyanides, this methodology not only enhances operational safety but also simplifies the downstream purification protocols required for high-purity active pharmaceutical ingredients. The strategic implementation of Lewis acid catalysis combined with mild borohydride reduction represents a substantial leap forward in green chemistry principles applied to fine chemical manufacturing. For global procurement teams and R&D directors, understanding the nuances of this patent is essential for securing a reliable supply chain for dopamine intermediates that is both economically viable and environmentally compliant.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
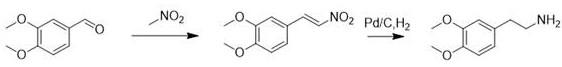
Historically, the industrial synthesis of 2-(3,4-dimethoxyphenyl)ethylamine has been plagued by significant safety hazards and complex waste management issues associated with traditional reaction pathways. One prevalent method involves the condensation of veratraldehyde with nitromethane via a Henry reaction, followed by catalytic hydrogenation using palladium on carbon. As illustrated in the reaction scheme below, this route necessitates the handling of nitromethane, a volatile and potentially explosive chemical that poses severe risks during large-scale storage and processing. Furthermore, the reliance on noble metal catalysts like palladium introduces substantial cost variability and requires rigorous removal steps to ensure residual metal levels meet stringent pharmacopeial standards. The waste streams generated from nitromethane usage are notoriously difficult to treat, creating an environmental burden that complicates regulatory compliance for modern manufacturing facilities.
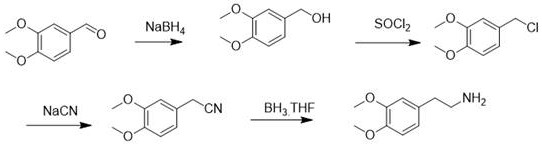
Another established pathway utilizes a multi-step sequence involving reduction, chlorination, cyanation, and subsequent reduction, which introduces sodium cyanide into the process flow. The use of sodium cyanide is heavily regulated due to its extreme toxicity, requiring specialized containment infrastructure and emergency response protocols that drive up capital expenditure. This route also suffers from lower atom economy due to the multiple transformation steps required to install the ethylamine side chain, leading to increased solvent consumption and reduced overall throughput. The cumulative effect of these factors results in a manufacturing process that is not only costly but also fragile in the face of tightening global safety regulations. Consequently, there is a pressing demand for alternative synthetic strategies that can deliver the same high-quality intermediate without the associated liabilities of explosive or highly toxic reagents.
The Novel Approach
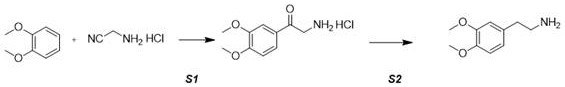
The methodology disclosed in CN114014766A offers a transformative solution by employing a direct acylation strategy known as the Houben-Hoesch reaction, followed by a selective reduction step. This novel approach utilizes o-dimethoxybenzene (veratrole) as a starting material, which is reacted directly with aminoacetonitrile hydrochloride in the presence of a Lewis acid catalyst such as aluminum chloride. This single-step acylation efficiently constructs the carbon-nitrogen backbone required for the final amine, bypassing the need for dangerous nitro-alkenes or toxic cyanide salts entirely. The subsequent reduction of the resulting ketone intermediate is achieved using mild hydride donors like sodium borohydride or potassium borohydride, which operate under ambient or mildly heated conditions. This eliminates the requirement for high-pressure hydrogenation equipment and expensive noble metal catalysts, drastically simplifying the reactor setup and operational requirements. The result is a cleaner, safer, and more cost-effective process that aligns perfectly with the goals of sustainable chemical manufacturing.
Mechanistic Insights into Lewis Acid Catalyzed Houben-Hoesch Acylation
The core of this innovative synthesis lies in the precise activation of the nitrile group through Lewis acid coordination, facilitating an electrophilic aromatic substitution on the electron-rich veratrole ring. In the first step, the Lewis acid, typically aluminum chloride or ferric chloride, coordinates with the nitrogen atom of the aminoacetonitrile hydrochloride, significantly increasing the electrophilicity of the nitrile carbon. Simultaneously, the introduction of dry hydrogen chloride gas ensures the protonation of the amino group, preventing unwanted side reactions and stabilizing the intermediate imine salt. The electron-donating methoxy groups on the veratrole ring activate the aromatic system, directing the attack to the ortho or para positions relative to the oxygen substituents, ultimately yielding the 2-amino-3',4'-dimethoxyacetophenone hydrochloride salt. This mechanism is highly efficient because it forms the carbon-carbon bond and the ketone functionality in a single operation, maximizing atom economy and minimizing the formation of byproducts that would complicate purification.
Following the acylation, the reduction mechanism proceeds via a nucleophilic attack of the hydride ion from the borohydride species onto the carbonyl carbon of the ketone intermediate. The choice of solvent, such as tetrahydrofuran (THF) or ethylene glycol dimethyl ether (DME), plays a critical role in solubilizing the Lewis acid complexes and ensuring smooth hydride transfer. Unlike catalytic hydrogenation which can sometimes lead to over-reduction or dehalogenation if impurities are present, borohydride reduction is chemoselective for the ketone moiety in this context, leaving the aromatic ring and ether linkages intact. The reaction is quenched carefully with dilute acid to decompose excess borohydride and hydrolyze any boron complexes, followed by basification to liberate the free amine. This controlled reduction environment ensures high purity of the final 2-(3,4-dimethoxyphenyl)ethylamine, minimizing the presence of alcohol impurities or reduced aromatic species that could affect downstream drug synthesis.
How to Synthesize 2-(3,4-Dimethoxyphenyl)ethylamine Efficiently
The execution of this synthesis requires careful attention to moisture control during the Lewis acid catalyzed step and precise temperature management during the reduction phase to ensure optimal yields and safety. The process begins with the mixing of o-dimethoxybenzene and aminoacetonitrile hydrochloride with a stoichiometric amount of anhydrous Lewis acid, followed by the slow introduction of dry HCl gas to initiate the Houben-Hoesch cyclization. Once the acylation is complete, the reaction mixture is quenched and the intermediate ketone salt is isolated through extraction and pulping, providing a stable solid that can be stored or immediately processed. The second stage involves dissolving this intermediate in a suitable ether solvent and adding the reducing agent in batches to manage the exotherm, ensuring the reaction temperature remains within the specified range of 60-70°C for complete conversion. Detailed standardized operating procedures regarding reagent addition rates, quenching protocols, and work-up sequences are critical for reproducibility and are outlined in the structured guide below.
- Perform Houben-Hoesch acylation by reacting o-dimethoxybenzene with aminoacetonitrile hydrochloride using a Lewis acid catalyst like aluminum chloride under dry HCl gas flow.
- Quench the reaction mixture with water, extract with dichloromethane, and purify the resulting 2-amino-3',4'-dimethoxyacetophenone hydrochloride via pulping in n-heptane.
- Reduce the ketone intermediate using sodium borohydride in a solvent like THF or DME, followed by pH adjustment and extraction to isolate the final amine product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patent-protected synthesis route offers profound advantages for procurement managers and supply chain directors tasked with optimizing the cost structure of dopamine production. The primary driver of cost reduction is the complete elimination of noble metal catalysts, which removes the volatility associated with palladium pricing and the logistical complexity of catalyst recovery and recycling programs. Furthermore, by avoiding the use of nitromethane and sodium cyanide, manufacturers can significantly reduce the costs associated with hazardous waste disposal, specialized storage infrastructure, and regulatory compliance reporting. The use of commodity chemicals like veratrole and aluminum chloride ensures a stable and resilient supply chain, insulating production schedules from the shortages that often plague specialized reagents. This stability translates directly into more predictable lead times and a lower total cost of ownership for the intermediate, allowing pharmaceutical companies to maintain competitive pricing for their final cardiovascular formulations.
- Cost Reduction in Manufacturing: The economic benefits of this route are derived principally from the substitution of expensive catalytic systems with inexpensive stoichiometric reagents that are globally available in bulk quantities. By removing the need for palladium on carbon, the process eliminates the capital expense of high-pressure hydrogenation reactors and the ongoing operational cost of catalyst filtration and metal scavenging resins. Additionally, the simplified work-up procedure, which relies on standard aqueous extractions and pulping rather than complex chromatographic purifications, reduces solvent consumption and labor hours per batch. These cumulative efficiencies result in a substantially lower cost of goods sold (COGS) for the intermediate, providing a clear margin advantage for manufacturers who adopt this technology.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly improved by relying on raw materials that are produced on a massive industrial scale for other sectors, such as fragrances and agrochemicals. Veratrole and aminoacetonitrile hydrochloride are not subject to the same geopolitical supply constraints or production bottlenecks as specialty catalysts or highly regulated toxic substances. This abundance ensures that production volumes can be scaled up rapidly to meet surges in demand for dopamine without the risk of raw material stockouts. Moreover, the avoidance of strictly controlled substances like sodium cyanide simplifies the logistics of transportation and warehousing, reducing the administrative burden and insurance costs associated with moving hazardous materials across borders.
- Scalability and Environmental Compliance: The environmental profile of this synthesis is markedly superior to conventional methods, facilitating easier permitting and long-term operational sustainability. The absence of nitromethane eliminates the generation of nitro-containing wastewater, which is notoriously difficult and expensive to treat in biological sewage systems. Similarly, the lack of cyanide removes the risk of catastrophic environmental contamination, aligning the process with increasingly stringent global environmental, social, and governance (ESG) criteria. The mild reaction conditions and the use of recyclable solvents like THF further enhance the green credentials of the process, making it an attractive option for companies aiming to reduce their carbon footprint and achieve zero-discharge goals in their manufacturing facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Houben-Hoesch based synthesis for dopamine intermediates. These answers are derived directly from the experimental data and beneficial effects described in the patent literature, providing a factual basis for evaluating the technology's fit within your existing manufacturing portfolio. Understanding these details is crucial for R&D teams assessing process feasibility and procurement officers negotiating supply contracts.
Q: Why is the Houben-Hoesch route preferred over nitromethane-based synthesis for dopamine intermediates?
A: The Houben-Hoesch route eliminates the use of nitromethane, an explosive reagent that creates difficult-to-treat waste streams, and avoids expensive palladium-on-carbon hydrogenation, significantly improving industrial safety and cost efficiency.
Q: What are the safety advantages of avoiding sodium cyanide in this synthesis?
A: By bypassing the cyanation step required in traditional routes, this method removes the need for handling highly toxic sodium cyanide, thereby reducing regulatory compliance burdens and minimizing risks associated with hazardous material storage and disposal.
Q: Can this process be scaled for commercial production without noble metal catalysts?
A: Yes, the process utilizes common Lewis acids like aluminum chloride and standard reducing agents like sodium borohydride, making it highly scalable and independent of the supply chain volatility often associated with precious metal catalysts.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-(3,4-Dimethoxyphenyl)ethylamine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the consistent production of life-saving cardiovascular medications. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering 2-(3,4-dimethoxyphenyl)ethylamine with stringent purity specifications, utilizing our rigorous QC labs to verify that every batch meets the exacting standards required for API synthesis. Our facility is equipped to handle the specific Lewis acid and reduction chemistries described in CN114014766A, guaranteeing a supply that is both chemically robust and commercially competitive.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can optimize your supply chain and reduce your overall manufacturing costs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the specific economic benefits applicable to your production volume. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions based on hard data and proven technical capability.
