Advanced LDA-Mediated Synthesis of 2-Chloro-1,3-Thiazole-5-Formaldehyde for Commercial Scale-Up
The pharmaceutical industry continuously demands more efficient and scalable routes for critical heterocyclic building blocks, and patent CN109678814B presents a significant breakthrough in the synthesis of 2-chloro-1,3-thiazole-5-formaldehyde. This specific intermediate is pivotal for constructing various bioactive molecules, yet traditional manufacturing methods have long been plagued by stringent reaction conditions and poor selectivity issues that hinder mass production. The disclosed innovation utilizes lithium diisopropylamide (LDA) as a superior base to facilitate the lithiation of 2-chlorothiazole, enabling the reaction to proceed efficiently across a much broader temperature window than previously possible. By shifting away from highly reactive organolithium reagents that compromise product integrity, this method ensures a cleaner reaction profile and significantly higher yields, addressing the critical needs of a reliable pharma intermediates supplier. For R&D directors and process chemists, this represents a viable pathway to enhance purity specifications while minimizing the formation of difficult-to-remove impurities that often arise from non-selective base attacks. The strategic implementation of this technology allows for a more robust supply chain, ensuring that high-purity 2-chloro-1,3-thiazole-5-formaldehyde can be delivered consistently to meet the rigorous demands of downstream drug synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of 2-chloro-1,3-thiazole-5-formaldehyde relied heavily on the use of n-butyllithium as the lithiating agent, a method documented in earlier literature such as the work by Wilson et al. While capable of achieving conversion, n-butyllithium is notoriously aggressive and lacks the necessary chemoselectivity when confronted with electrophilic carbonyl sources required for the formylation step. This lack of selectivity leads to substantial side reactions where the base attacks the formylating agent itself rather than just deprotonating the thiazole ring, resulting in a complex mixture of byproducts that drastically reduce the overall isolated yield. Furthermore, the conventional process typically requires extremely low temperatures, often strictly limited to -78°C, to mitigate these side reactions, which imposes a heavy energy burden and operational complexity on the manufacturing infrastructure. Such cryogenic requirements are not only costly to maintain on a multi-ton scale but also introduce significant safety risks and engineering challenges related to heat transfer and mixing efficiency in large reactors. Consequently, the traditional n-butyllithium route is often deemed unsuitable for large-scale industrial popularization, creating a bottleneck for procurement managers seeking cost reduction in pharmaceutical intermediates manufacturing.
The Novel Approach
The innovative method described in CN109678814B overcomes these historical barriers by employing lithium diisopropylamide (LDA), a bulky, non-nucleophilic base that exhibits exceptional dehydrogenation selectivity. Unlike its n-butyllithium counterpart, LDA does not react with the carbonyl group of the formylating agent, thereby preserving the integrity of the electrophile and ensuring that the reaction proceeds primarily through the desired lithiation-formylation pathway. This fundamental shift in reagent chemistry allows the reaction to be conducted effectively over a wider temperature range, spanning from -78°C up to -30°C, without sacrificing yield or product quality. This flexibility is a game-changer for process engineers, as it reduces the dependency on extreme cryogenic cooling, thereby lowering operational expenditures and simplifying the thermal management of the reactor system. The ability to operate at slightly higher temperatures while maintaining high conversion rates translates directly into improved throughput and energy efficiency, making the commercial scale-up of complex pharmaceutical intermediates far more economically viable. By eliminating the side reactions associated with aggressive organolithium reagents, this novel approach delivers a cleaner crude product, which subsequently simplifies the downstream purification workup and reduces solvent consumption.
Mechanistic Insights into LDA-Catalyzed Lithiation-Formylation
The core of this synthetic advancement lies in the precise mechanistic interaction between the bulky LDA base and the 2-chlorothiazole substrate, which facilitates a highly regioselective deprotonation at the C5 position of the thiazole ring. The steric bulk of the diisopropyl groups on the nitrogen atom of LDA prevents it from acting as a nucleophile towards the electrophilic centers, forcing it to act exclusively as a Brønsted base to abstract the acidic proton at the 5-position. This generates a stable 5-lithio-2-chlorothiazole intermediate which is resonance-stabilized by the adjacent nitrogen and sulfur atoms within the heterocyclic system, ensuring that the anion remains intact long enough to react with the formylating agent. Once the lithiated species is formed, the subsequent addition of N,N-dimethylformamide (DMF) or piperidine-1-carbaldehyde allows for a nucleophilic attack on the carbonyl carbon, effectively installing the formyl group onto the thiazole scaffold. The stability of the LDA-generated anion is crucial, as it prevents the decomposition or polymerization of the intermediate that often occurs with more reactive lithiating agents, thereby securing the high yields reported in the patent examples. This mechanistic clarity provides R&D teams with the confidence to optimize reaction parameters, knowing that the selectivity is inherent to the reagent choice rather than dependent on fragile kinetic control.
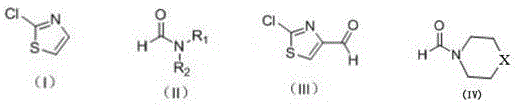
Controlling the impurity profile is another critical aspect of this mechanism, particularly regarding the avoidance of over-lithiation or attack on the chlorine substituent which could lead to dehalogenated byproducts. The use of LDA minimizes the risk of halogen-metal exchange, a common side reaction with aryl and heteroaryl halides when using smaller organolithium reagents like n-butyllithium. By preserving the chloro-substituent, the process ensures that the final product retains the necessary functionality for subsequent cross-coupling reactions, which is often the intended use of this intermediate in drug discovery pipelines. Furthermore, the quenching process described involves careful pH adjustment and extraction, which effectively removes the lithium salts and amine byproducts generated during the reaction, leading to a product with high chemical purity. The patent data indicates that this method consistently produces the target aldehyde with minimal impurities, reducing the need for extensive chromatographic purification and allowing for simpler crystallization or distillation techniques. For quality control laboratories, this means a more straightforward analytical profile and a reduced risk of failing specification limits due to unknown or difficult-to-quantify impurities, ensuring a reliable supply of high-purity thiazole derivatives for sensitive pharmaceutical applications.
How to Synthesize 2-Chloro-1,3-Thiazole-5-Formaldehyde Efficiently
The synthesis protocol outlined in the patent provides a clear and reproducible pathway for manufacturing this valuable intermediate, starting with the preparation of the reaction solutions under inert atmosphere conditions to prevent moisture interference. The process begins by dissolving 2-chlorothiazole in a suitable organic solvent such as tetrahydrofuran, followed by the controlled addition of the LDA solution at low temperatures to initiate the lithiation step. Detailed standardized synthesis steps see the guide below, which breaks down the precise molar ratios, temperature profiles, and workup procedures required to achieve the optimal yields demonstrated in the patent examples. Adhering to these parameters is essential for replicating the high efficiency of the process, particularly regarding the dropwise addition rates and the specific temperature maintenance periods that ensure complete conversion without thermal runaway. This structured approach allows manufacturing teams to translate the laboratory-scale success directly into pilot and commercial production environments with minimal deviation.
- Dissolve 2-chlorothiazole in THF and cool the solution to a temperature range between -78°C and -30°C to prepare for lithiation.
- Add lithium diisopropylamide (LDA) solution dropwise to the cooled mixture and maintain the temperature for 5 to 30 minutes to ensure complete deprotonation.
- Introduce the formylating agent (DMF or piperidine-1-carbaldehyde), warm to room temperature over 0.5 to 5 hours, and quench with acid to isolate the product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this LDA-mediated synthesis offers substantial benefits for procurement managers and supply chain heads who are tasked with optimizing costs and ensuring continuity of supply. The primary advantage lies in the drastic simplification of the reaction conditions, which eliminates the need for the extreme cryogenic infrastructure required by older methods, thereby reducing both capital expenditure on equipment and ongoing operational energy costs. By enabling the reaction to proceed at temperatures as high as -30°C, the process becomes more energy-efficient and easier to manage in standard industrial reactors, which directly contributes to cost reduction in pharmaceutical intermediates manufacturing without compromising on product quality. Additionally, the high selectivity of LDA means that raw material utilization is maximized, as less starting material is lost to side reactions, leading to a more sustainable and economically favorable material balance. This efficiency translates into a more competitive pricing structure for the final intermediate, allowing downstream partners to manage their own cost of goods sold more effectively while maintaining healthy margins. The robustness of the process also implies fewer batch failures and less waste generation, aligning with modern environmental compliance standards and reducing the burden on waste treatment facilities.
- Cost Reduction in Manufacturing: The elimination of aggressive reagents like n-butyllithium removes the necessity for complex safety measures and specialized handling equipment, which significantly lowers the operational overhead associated with the production line. Furthermore, the higher selectivity reduces the consumption of expensive starting materials and solvents that would otherwise be lost to byproduct formation, resulting in substantial cost savings on raw material procurement. The simplified workup procedure, which avoids extensive purification steps, further decreases the labor and utility costs per kilogram of product, making the overall manufacturing process leaner and more profitable. These cumulative efficiencies allow for a more aggressive pricing strategy in the market while maintaining high profitability, providing a distinct competitive advantage for suppliers who adopt this technology.
- Enhanced Supply Chain Reliability: The wider operating temperature range of this synthesis method makes the production process more resilient to minor fluctuations in cooling capacity or ambient conditions, reducing the risk of batch delays or cancellations due to equipment limitations. This robustness ensures a more consistent output rate, which is critical for maintaining reducing lead time for high-purity thiazole derivatives and meeting the just-in-time delivery expectations of global pharmaceutical clients. Moreover, the reagents used, such as LDA and DMF, are commercially available in bulk quantities from established chemical suppliers, minimizing the risk of raw material shortages that could disrupt the supply chain. This availability ensures that production schedules can be maintained reliably, providing procurement managers with the confidence to plan long-term contracts without fear of supply interruptions.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, as the reduced exothermicity and wider temperature window allow for safer operation in larger reactor volumes without the risk of thermal runaway. This ease of scale-up facilitates the rapid transition from pilot plant to full commercial production, enabling suppliers to quickly respond to increased market demand for this critical intermediate. Additionally, the reduction in side products and waste streams aligns with stricter environmental regulations, reducing the cost and complexity of waste disposal and treatment. This environmental compliance not only mitigates regulatory risk but also enhances the corporate sustainability profile of the manufacturing entity, which is increasingly important for multinational corporations seeking responsible partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 2-chloro-1,3-thiazole-5-formaldehyde, based on the specific data and advantages outlined in the patent literature. These insights are intended to clarify the operational benefits and technical feasibility for stakeholders evaluating this synthesis route for their supply chains. Understanding these details is crucial for making informed decisions about integrating this intermediate into broader drug development or manufacturing programs.
Q: Why is LDA preferred over n-butyllithium for this synthesis?
A: LDA offers superior dehydrogenation selectivity and does not react with the carbonyl group of the formylating agent, unlike n-butyllithium which causes significant side reactions and limits temperature flexibility.
Q: What is the expected yield range for this process?
A: According to patent data, the process achieves high yields ranging from 80% to 96% depending on the specific temperature conditions and scale, demonstrating robust efficiency.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the method operates effectively over a wider temperature range (-78°C to -30°C) compared to prior art, reducing cryogenic energy costs and facilitating easier commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Chloro-1,3-Thiazole-5-Formaldehyde Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of your pharmaceutical development pipelines, and we are committed to delivering this advanced synthesis technology to the global market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this LDA-mediated process are fully realized at an industrial level. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 2-chloro-1,3-thiazole-5-formaldehyde meets the highest standards required for drug substance manufacturing. Our commitment to technical excellence means that we do not just supply chemicals; we provide a partnership grounded in scientific expertise and reliable execution, giving you the confidence to focus on your core innovation activities while we manage the complexities of synthesis.
We invite you to engage with our technical procurement team to discuss how this optimized route can benefit your specific projects and to request a Customized Cost-Saving Analysis tailored to your volume requirements. By leveraging our capabilities, you can access specific COA data and route feasibility assessments that will help you validate the suitability of this intermediate for your applications. Let us help you streamline your supply chain and reduce your time-to-market with a partner who understands the nuances of fine chemical manufacturing and the demands of the global pharmaceutical industry.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
