Revolutionizing Dicyanoethyl Cyclohexylamine Production with Recyclable Acidic Ionic Liquids
The chemical manufacturing landscape for specialized amine intermediates is undergoing a significant transformation driven by the need for higher purity and sustainable catalytic systems. Patent CN114890913B, published in May 2023, introduces a groundbreaking methodology for the preparation of dicyanoethyl cyclohexylamine, a critical intermediate widely utilized in the production of epoxy curing agents and isocyanates. This innovation addresses long-standing challenges in cyanoethylation reactions, specifically the formation of stubborn amide by-products and the difficulty of removing residual acids that poison downstream hydrogenation catalysts. By employing a novel acidic ionic liquid catalyst system, the patent outlines a route that achieves yields exceeding 98% while maintaining acid content below 50ppm. For R&D directors and procurement specialists seeking a reliable dicyanoethyl cyclohexylamine supplier, this technology represents a paradigm shift towards cleaner, more efficient, and economically viable manufacturing processes that eliminate the need for complex neutralization and purification steps traditionally associated with organic acid catalysts.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of dicyanoethyl tertiary amines from primary amines has been plagued by significant chemical inefficiencies when using traditional organic acid catalysts such as glycolic acid. As detailed in the background art of the patent, conventional one-step or glycolic acid-catalyzed processes suffer from a critical flaw: the carboxylic acid group of the catalyst readily reacts with the amine substrate to form stable amide by-products. These amide impurities are notoriously difficult to separate from the desired cyanoethylated product due to similar boiling points and polarity, leading to compromised product purity. Furthermore, the strong binding affinity between the acid catalyst and the amine product often results in residual acid remaining in the final material, even after high-temperature decomposition attempts. This residual acidity is catastrophic for downstream applications, particularly nitrile hydrogenation, where trace amounts of acid can irreversibly deactivate expensive noble metal catalysts, leading to reduced selectivity and increased operational costs for manufacturers.
The Novel Approach
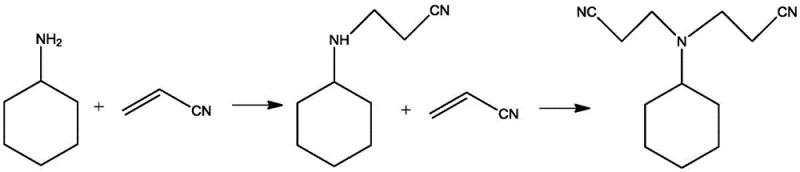
The methodology disclosed in patent CN114890913B overcomes these inherent limitations through a sophisticated two-step strategy utilizing a recyclable acidic ionic liquid catalyst. Unlike the chaotic one-pot reactions of the past, this novel approach first isolates the mono-cyanoethylation step using water as a mild catalyst, ensuring complete conversion of the primary amine before introducing the second equivalent of acrylonitrile. The introduction of the acidic ionic liquid in the second step provides a highly active yet selective catalytic environment that promotes the Michael addition without facilitating amide formation. This strategic separation of reaction stages, combined with the unique physicochemical properties of the ionic liquid, allows for the production of dicyanoethyl cyclohexylamine with exceptional purity. The process not only eliminates the generation of saline wastewater associated with neutralization steps but also enables the direct reuse of the catalyst, offering a compelling solution for cost reduction in pharmaceutical intermediates manufacturing and fine chemical production.
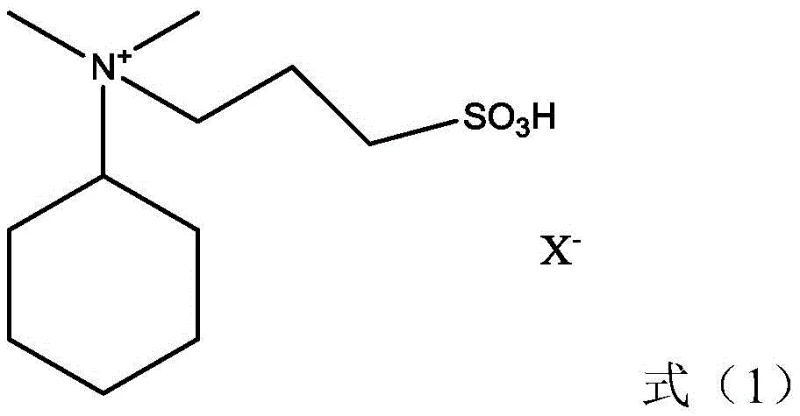
Mechanistic Insights into Acidic Ionic Liquid-Catalyzed Cyanoethylation
The core of this technological breakthrough lies in the specific design of the acidic ionic liquid catalyst, which typically features a [DMCHA-PS] cation structure paired with various anions such as chloride, bromide, or methanesulfonate. The cation, derived from N,N-dimethylcyclohexylamine and 1,3-propane sultone, possesses a structural similarity to the reaction intermediate, which facilitates the rapid activation of the hydrogen atom on the nitrogen of the monocyanoethylcyclohexylamine substrate. Simultaneously, the acidic anion creates a polarized environment that activates the carbon-carbon double bond of the acrylonitrile molecule. This dual-activation mechanism significantly lowers the energy barrier for the Michael addition reaction, allowing the process to proceed rapidly at moderate temperatures ranging from 60°C to 100°C. This is a marked improvement over prior art methods that often require harsher conditions or longer reaction times to achieve comparable conversion rates, thereby enhancing overall production efficiency and throughput.
Impurity control is meticulously managed through the physical properties of the catalyst system rather than just chemical selectivity. A defining feature of this ionic liquid is its ability to undergo phase separation from the organic product at low temperatures, typically between 10°C and 30°C. Upon cooling the reaction mixture, the system bifurcates into an aqueous phase containing the catalyst and an oil phase containing the crude dicyanoethyl cyclohexylamine. This physical separation ensures that the catalyst, along with any potential acidic residues, remains in the aqueous layer, preventing contamination of the organic product. Consequently, the resulting oil phase requires only simple vacuum distillation to remove excess acrylonitrile and water, yielding a product with acid content ≤50ppm and zero detectable amide by-products. This mechanism guarantees that the material is perfectly suited for sensitive downstream hydrogenation processes without risking catalyst poisoning.
How to Synthesize Dicyanoethyl Cyclohexylamine Efficiently
The synthesis protocol outlined in the patent provides a robust framework for scaling this reaction from laboratory benchtop to industrial production. The process begins with the controlled addition of acrylonitrile to cyclohexylamine in the presence of water to form the mono-substituted intermediate, followed by the introduction of the acidic ionic liquid to drive the second substitution. The precise control of molar ratios, particularly maintaining an excess of acrylonitrile in the first step, is critical to ensuring the complete consumption of the primary amine and preventing side reactions. Detailed standardized operating procedures regarding temperature ramps, addition rates, and phase separation techniques are essential for replicating the high yields and purity reported in the patent examples. For technical teams looking to implement this chemistry, the following guide summarizes the critical operational phases required to achieve optimal results.
- Perform mono-cyanoethylation of cyclohexylamine with acrylonitrile using water as a catalyst at 40-70°C to obtain monocyanoethylcyclohexylamine mother liquor.
- Add acidic ionic liquid catalyst (e.g., [DMCHA-PS]CH3SO3) to the mother liquor and react with additional acrylonitrile at 60-100°C to form dicyanoethyl cyclohexylamine.
- Cool the reaction mixture to 10-30°C for phase separation, recover the aqueous catalyst layer for reuse, and purify the organic layer via vacuum distillation.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this acidic ionic liquid catalytic system offers profound advantages for supply chain stability and cost management. Traditional methods relying on glycolic acid or other organic acids generate significant volumes of saline wastewater during the neutralization and workup phases, imposing heavy environmental compliance costs and waste disposal burdens on manufacturers. In contrast, the new process eliminates the need for alkali neutralization entirely, as the catalyst is recovered via phase separation. This drastic simplification of the post-treatment workflow translates directly into substantial cost savings by reducing utility consumption, waste treatment fees, and labor hours associated with complex purification protocols. Furthermore, the ability to recycle the catalyst multiple times without significant loss of activity reduces the raw material cost per kilogram of finished product, enhancing the overall economic competitiveness of the supply chain.
- Cost Reduction in Manufacturing: The elimination of expensive neutralization reagents and the reduction of wastewater treatment requirements significantly lower the variable costs associated with production. Additionally, the high catalytic activity allows for shorter reaction times and lower energy consumption compared to conventional thermal methods. The recyclability of the ionic liquid catalyst means that the effective cost of the catalyst per batch diminishes over time, providing a sustainable economic model that protects margins against fluctuating raw material prices.
- Enhanced Supply Chain Reliability: The robustness of this two-step process ensures consistent product quality, which is vital for maintaining uninterrupted supply to downstream customers in the epoxy and polyurethane industries. By avoiding the formation of hard-to-remove amide impurities, the risk of batch rejection due to specification failures is minimized. This reliability reduces the need for safety stock and allows for leaner inventory management, ensuring that high-purity dicyanoethyl cyclohexylamine is available exactly when needed for critical manufacturing schedules.
- Scalability and Environmental Compliance: The mild reaction conditions and the absence of hazardous by-products make this process highly scalable for commercial scale-up of complex amine intermediates. The technology aligns with green chemistry principles by minimizing waste generation and avoiding the use of volatile organic solvents for extraction, as the product separates naturally. This environmental compatibility simplifies regulatory permitting and reduces the risk of production shutdowns due to environmental non-compliance, securing long-term operational continuity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced cyanoethylation technology. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating this process into their existing manufacturing portfolios. The answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for decision-makers.
Q: Why is the acidic ionic liquid catalyst superior to glycolic acid for this synthesis?
A: Unlike glycolic acid, which reacts with amines to form difficult-to-remove amide by-products and leaves residual acid that poisons hydrogenation catalysts, the acidic ionic liquid catalyst in patent CN114890913B prevents amide formation entirely and achieves acid content levels below 50ppm through simple phase separation.
Q: How does the new process impact the quality of downstream hydrogenation products?
A: The process ensures the dicyanoethyl cyclohexylamine product contains no amide impurities and has extremely low acidity (≤50ppm). This prevents the deactivation of expensive hydrogenation catalysts in subsequent steps, significantly improving the selectivity and yield of the final amine products used in epoxy curing agents.
Q: Is the acidic ionic liquid catalyst reusable in this manufacturing process?
A: Yes, the catalyst features a unique phase-separation property at low temperatures (10-30°C). After the reaction, the catalyst remains in the aqueous phase while the product forms the oil phase, allowing the catalyst to be separated and directly recycled for the next batch without complex regeneration steps.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dicyanoethyl Cyclohexylamine Supplier
As the global demand for high-performance epoxy curing agents and specialty amines continues to grow, securing a supply of ultra-pure intermediates is more critical than ever. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging advanced catalytic technologies like the acidic ionic liquid system described in CN114890913B to deliver superior chemical solutions. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements without compromising on our stringent purity specifications. With rigorous QC labs dedicated to monitoring trace impurities such as amides and residual acids, we guarantee that every batch of dicyanoethyl cyclohexylamine meets the exacting standards required for sensitive downstream hydrogenation applications.
We invite procurement leaders and technical directors to collaborate with us to optimize their supply chains. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific production needs, demonstrating how our efficient manufacturing processes can reduce your total cost of ownership. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, and discover how our commitment to innovation can drive value and reliability in your operations.
