Advanced Dimerization Strategy for High-Purity Alpha-Hydroxy Acid Esters in Commercial Manufacturing
Introduction to Patent CN111269158A Technology
The chemical industry is constantly seeking more efficient pathways to synthesize complex intermediates, and patent CN111269158A presents a breakthrough methodology for the preparation of α-hydroxy acid ester compounds. This technology addresses long-standing challenges in organic synthesis by introducing a novel dimerization-esterification cascade that fundamentally alters the thermodynamic landscape of the reaction. Unlike traditional methods that struggle with equilibrium limitations and excessive waste generation, this approach utilizes a cyclic dimer intermediate to drive the reaction to completion with exceptional efficiency. For R&D directors and process chemists, this represents a significant opportunity to enhance the purity profiles of critical intermediates used in pharmaceutical and agrochemical applications. The patent details a robust protocol that not only improves yield but also aligns with modern green chemistry principles by facilitating solvent recovery.
From a commercial perspective, the implications of this technology are profound for supply chain stability and cost management. By enabling the rapid and high-efficiency recovery of alkyl alcohols, the process drastically reduces raw material consumption and waste liquid production. This is particularly relevant for the manufacturing of high-volume commodities like methionine hydroxyl analogues, where marginal improvements in efficiency translate to substantial economic advantages. As a reliable pharmaceutical intermediate supplier, understanding and adopting such innovative synthetic routes is essential for maintaining competitiveness in a global market that increasingly demands sustainable and cost-effective manufacturing solutions. The following analysis delves into the technical specifics and commercial viability of this dimerization strategy.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for α-hydroxy acid esters have historically been plagued by significant inefficiencies that hinder large-scale industrial adoption. The first category of conventional methods involves using α-hydroxy acid salts as raw materials, a process that is notoriously complicated, costly, and ill-suited for continuous large-scale production due to the extensive downstream processing required. The second category relies on α-hydroxy nitriles, which necessitate harsh hydrolysis conditions using concentrated sulfuric acid followed by esterification; this approach suffers from low yields, massive consumption of reaction media, and severe environmental burdens due to difficult-to-treat waste gas and liquid emissions. Furthermore, the third common method, direct esterification of α-hydroxy acids with alcohols under acid catalysis, while yielding better results, still requires a large excess of alcohol and generates significant amounts of wastewater, making it environmentally unfriendly and economically suboptimal for modern green manufacturing standards.
The Novel Approach
In stark contrast to these legacy methods, the novel approach described in the patent utilizes a strategic dimerization step that fundamentally simplifies the purification process and enhances overall efficiency. By first converting the α-hydroxy acid compound into a stable dimer intermediate, the reaction pathway bypasses the equilibrium constraints typical of direct esterification. This intermediate then undergoes depolymerization and esterification with the corresponding alkyl alcohol, a sequence that allows for the single-step formation of the target ester with minimal side reactions. The structural simplicity of the product stream means that purification is significantly easier, reducing the need for complex chromatographic separations or multiple recrystallizations. This streamlined workflow not only boosts throughput but also ensures a higher degree of consistency in product quality, which is critical for regulatory compliance in pharmaceutical supply chains.
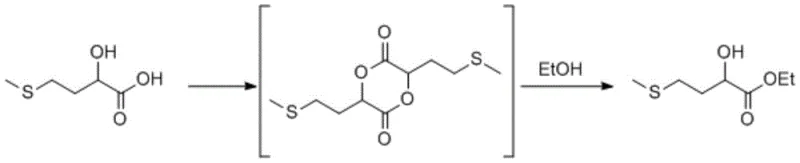
Mechanistic Insights into Acid-Catalyzed Dimerization and Alcoholysis
The core of this innovative synthesis lies in the formation and subsequent reactivity of the cyclic dimer intermediate, a structural motif that serves as a high-energy reservoir for the esterification process. Mechanistically, the initial step involves the acid-catalyzed condensation of two molecules of the α-hydroxy acid, accompanied by the removal of water, typically achieved through azeotropic distillation with solvents like toluene or n-heptane. This dehydration drives the equilibrium towards the formation of the 1,4-dioxane-2,5-dione derivative, effectively protecting the hydroxyl and carboxyl groups in a rigid cyclic structure. The stability of this dimer prevents premature polymerization or degradation, ensuring that the reactive functionality is preserved until the introduction of the alcohol nucleophile in the second stage.
Upon the addition of the alkyl alcohol and acid catalyst, the cyclic dimer undergoes ring-opening alcoholysis, a process that is thermodynamically favorable and kinetically rapid under reflux conditions. This depolymerization releases the monomeric ester units while regenerating the acid catalyst, creating a cycle that minimizes catalyst loading requirements. Crucially, because no water is generated in this second step, the reaction environment remains anhydrous, preventing the hydrolysis of the newly formed ester back to the acid. This mechanistic feature is key to achieving high conversions without needing a vast excess of alcohol, thereby simplifying the downstream recovery of unreacted alcohol via simple distillation. The result is a clean reaction profile with a narrow impurity spectrum, facilitating the production of high-purity intermediates suitable for sensitive biological applications.
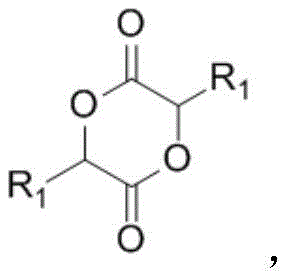
How to Synthesize Alpha-Hydroxy Acid Esters Efficiently
The practical implementation of this dimerization strategy offers a straightforward yet powerful protocol for synthesizing a wide range of α-hydroxy acid esters, from simple aliphatic derivatives to complex functionalized analogues. The process begins with the careful selection of an appropriate acid catalyst, such as phosphoric acid or p-toluenesulfonic acid, and an azeotropic solvent system to facilitate water removal during the dimer formation. Once the dimer intermediate is isolated or generated in situ, it is subjected to alcoholysis with the desired alcohol, where temperature control and catalyst concentration are optimized to ensure complete conversion without thermal degradation. The detailed standardized synthesis steps, including specific molar ratios and temperature profiles for various substrates, are outlined in the guide below to assist process engineers in replicating these high-yielding results.
- Perform dimerization reaction on alpha-hydroxy acid compounds using an acid catalyst and azeotropic water removal to form a cyclic dimer intermediate.
- Conduct depolymerization and esterification by reacting the dimer intermediate with the corresponding alkyl alcohol under acidic conditions.
- Recover excess alcohol via distillation, neutralize the mixture, and purify the final ester through vacuum distillation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this dimerization-based synthesis route offers compelling advantages that directly impact the bottom line and operational resilience. The primary benefit stems from the drastic reduction in raw material costs, particularly regarding the consumption of alkyl alcohols. In traditional direct esterification, a large molar excess of alcohol is required to push the equilibrium, much of which ends up in wastewater streams; however, this new method allows for the efficient recovery and recycling of the alcohol, significantly lowering the net consumption per kilogram of product. This efficiency translates into substantial cost savings over time, making the manufacturing process more economically viable even when raw material prices fluctuate in the global market.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the reduction in acid usage contribute to a leaner cost structure. By avoiding the need for complex neutralization and waste treatment steps associated with large volumes of acidic wastewater, the overall processing costs are significantly lowered. Furthermore, the high selectivity of the reaction minimizes the formation of by-products, which reduces the loss of valuable starting materials and lowers the cost of goods sold (COGS) through improved mass balance efficiency.
- Enhanced Supply Chain Reliability: The simplicity of the reaction conditions and the use of readily available commodity chemicals like toluene, ethanol, and sulfuric acid ensure that the supply chain remains robust against disruptions. The ability to recover solvents and alcohols on-site reduces dependency on external suppliers for fresh materials, thereby enhancing the continuity of production. This self-sufficiency is crucial for maintaining consistent delivery schedules to downstream customers, especially in the volatile landscape of pharmaceutical intermediate sourcing where reliability is paramount.
- Scalability and Environmental Compliance: From an environmental standpoint, the reduction in waste liquid and the absence of hazardous by-products make this process highly scalable without incurring prohibitive waste disposal costs. The green chemistry attributes of the method align with increasingly stringent global environmental regulations, reducing the risk of compliance-related shutdowns. This sustainability profile not only future-proofs the manufacturing asset but also appeals to end-users who are prioritizing eco-friendly supply chains in their vendor selection criteria.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this dimerization technology in industrial settings. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear picture of what potential partners can expect. Understanding these nuances is vital for making informed decisions about process adoption and integration into existing manufacturing lines.
Q: How does the dimerization method improve yield compared to direct esterification?
A: The dimerization route avoids the equilibrium limitations of direct Fischer esterification by forming a stable cyclic intermediate, which drives the reaction forward and minimizes side reactions, resulting in significantly higher isolated yields.
Q: What are the environmental benefits of this synthesis route?
A: This method allows for the efficient recovery and recycling of alkyl alcohol solvents and eliminates the generation of large volumes of acidic wastewater typically associated with traditional sulfuric acid-catalyzed direct esterification processes.
Q: Is this process suitable for scaling up methionine hydroxyl analogues?
A: Yes, the patent specifically highlights the synthesis of 2-hydroxy-4-methylthiobutyric acid esters, demonstrating that the mild conditions and simple workup make it highly scalable for producing methionine precursors industrially.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Hydroxy Acid Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced synthetic methodologies like the dimerization route described in CN111269158A for producing high-value intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory successes are seamlessly translated into industrial reality. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that employ state-of-the-art analytical techniques to verify every batch. Whether you require methionine hydroxyl analogues for animal nutrition or specialized esters for pharmaceutical synthesis, our infrastructure is designed to meet the highest standards of quality and consistency.
We invite you to collaborate with us to leverage these technological advancements for your specific application needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how this efficient synthesis route can optimize your budget. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate why we are the preferred partner for sustainable and high-performance chemical solutions.
