Advanced Two-Step Synthesis of Metoprolol Intermediate for Scalable Pharma Production
Advanced Two-Step Synthesis of Metoprolol Intermediate for Scalable Pharma Production
The pharmaceutical industry continuously seeks robust synthetic pathways for cardiovascular therapeutics, particularly for beta-blockers like Metoprolol. A pivotal development in this domain is documented in patent CN109553513B, which discloses a highly efficient preparation method for the key intermediate, 4-(2-methoxyethyl)phenol. This compound serves as the foundational building block for Metoprolol, a widely prescribed medication for hypertension and angina. The structural complexity of the final active pharmaceutical ingredient (API) demands precise construction of the side chain, as illustrated in the molecular architecture below. By shifting away from traditional stoichiometric reagents toward catalytic processes, this technology addresses critical pain points in modern drug manufacturing, offering a streamlined approach that aligns with green chemistry principles while maintaining high purity standards essential for regulatory compliance.
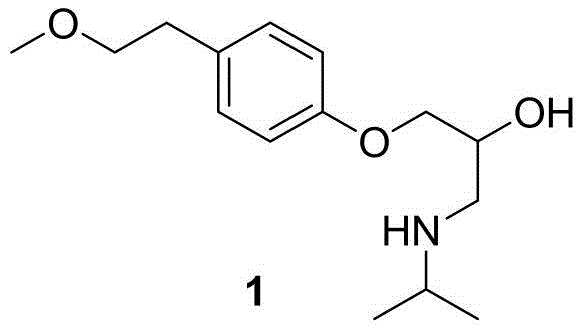
For procurement specialists and supply chain managers, the implications of this patent extend beyond mere chemical curiosity. The ability to source high-purity pharmaceutical intermediates through a shorter, safer synthetic route translates directly into supply chain resilience. Traditional methods often suffer from volatility in raw material costs due to the use of hazardous reagents like bromine or aluminum chloride, which require specialized handling and disposal infrastructure. In contrast, the methodology outlined in CN109553513B utilizes readily available starting materials such as p-bromophenol and methyl vinyl ether. This shift not only mitigates operational risks associated with toxic reagents but also stabilizes the cost structure of API manufacturing, making it a strategically superior choice for long-term production planning in the competitive cardiovascular drug market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 4-(2-methoxyethyl)phenol has been plagued by significant technical and environmental hurdles. One prevalent conventional route involves Friedel-Crafts acylation using chloroacetyl chloride and aluminum trichloride. While the raw materials are inexpensive, this process generates substantial amounts of corrosive waste and requires stoichiometric quantities of Lewis acids, complicating downstream purification and increasing the environmental footprint. Another common approach utilizes bromination of p-acetylphenol followed by reduction; however, this introduces elemental bromine, a highly toxic and volatile substance that poses severe safety risks during scale-up. Furthermore, nitration-based routes, though chemically feasible, involve dangerous diazotization steps and often yield unwanted ortho-substituted isomers, necessitating complex separation protocols that erode overall process efficiency and yield.
The Novel Approach
In stark contrast to these legacy methods, the innovation presented in patent CN109553513B offers a streamlined two-step sequence that bypasses these inherent dangers. The core of this novel strategy lies in a palladium-catalyzed coupling reaction between p-bromophenol and methyl vinyl ether, followed by a selective hydrogenation step. This approach effectively constructs the requisite carbon-oxygen and carbon-carbon bonds in a single catalytic cycle, eliminating the need for harsh acylation or nitration conditions. As depicted in the reaction scheme below, the process flows logically from simple precursors to the target intermediate with minimal byproduct formation. This elegance in design not only shortens the overall production timeline but also drastically simplifies the workup procedure, allowing for direct progression to the next step without rigorous intermediate purification, thereby enhancing throughput.
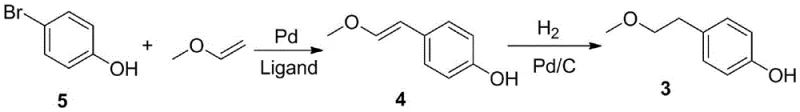
The transition to this catalytic methodology represents a paradigm shift in cost reduction in pharmaceutical intermediates manufacturing. By replacing stoichiometric metal reagents with catalytic systems, the process minimizes waste generation and reduces the burden on wastewater treatment facilities. Moreover, the mild reaction conditions—typically operating between 60°C and 100°C—lower energy consumption compared to high-temperature alternatives. For a reliable pharmaceutical intermediates supplier, adopting such a route ensures a more consistent supply of material with fewer batch-to-batch variations caused by difficult-to-control exothermic reactions. The use of methyl vinyl ether, a stable and commercially accessible reagent, further secures the supply chain against the fluctuations often seen with specialized acylating agents, making this route exceptionally robust for commercial scale-up.
Mechanistic Insights into Pd-Catalyzed Vinylation and Hydrogenation
The heart of this synthetic breakthrough is the palladium-catalyzed cross-coupling reaction, likely proceeding through a mechanism analogous to the Heck reaction or a specialized alkoxy-vinylation pathway. In the first step, the palladium catalyst, such as palladium acetate coordinated with a phosphine ligand like triphenylphosphine, activates the carbon-bromine bond of p-bromophenol. This oxidative addition forms a reactive organopalladium species that subsequently coordinates with methyl vinyl ether. The insertion of the alkene into the palladium-carbon bond, followed by beta-hydride elimination, yields the 4-(2-methoxyvinyl)phenol intermediate. The presence of a base, such as triethylamine, is crucial for neutralizing the hydrogen bromide byproduct and regenerating the active catalytic species. This mechanistic precision ensures high regioselectivity, preventing the formation of undesired isomers that commonly plague electrophilic aromatic substitution reactions.
Following the coupling, the second stage involves the catalytic hydrogenation of the vinyl double bond to form the ethyl side chain. Using a palladium-on-carbon (Pd/C) catalyst under moderate hydrogen pressure (30-50 atm), the olefinic bond is selectively reduced without affecting the aromatic ring or the ether linkage. This step is critical for impurity control, as over-reduction or hydrogenolysis of the ether bond must be avoided. The patent specifies that the crude product from the first step can be used directly, suggesting that the catalyst system is tolerant to minor impurities, which is a significant advantage for industrial processing. This tolerance reduces the need for intermediate isolation, saving both time and solvent. The result is a high-purity final product, often exceeding 97% purity after simple recrystallization, meeting the stringent specifications required for subsequent conversion into the final beta-blocker API.
How to Synthesize 4-(2-Methoxyethyl)phenol Efficiently
Implementing this synthesis requires careful attention to catalyst loading and reaction parameters to maximize yield and minimize residual metal content. The process begins with the dissolution of p-bromophenol in a suitable solvent like dioxane or DMF, followed by the addition of the palladium source and ligand. Maintaining an inert atmosphere is essential to prevent catalyst oxidation. Once the coupling is complete, the reaction mixture is concentrated, and the residue is subjected to hydrogenation in ethyl acetate. The simplicity of the workup—often involving just filtration and pH adjustment—makes this protocol highly attractive for pilot plant operations. For detailed operational parameters, including specific molar ratios and temperature profiles, please refer to the standardized synthesis guide provided below.
- React p-bromophenol with methyl vinyl ether using a palladium catalyst (e.g., palladium acetate) and phosphine ligand in a solvent like dioxane at 80-90°C.
- Hydrogenate the resulting 4-(2-methoxyvinyl)phenol intermediate using a palladium-carbon catalyst under 30-50 atm pressure at 50°C to yield the final product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented route offers profound benefits for procurement strategies and supply chain stability. The elimination of stoichiometric aluminum chloride and toxic bromine removes significant liability and disposal costs from the balance sheet. Traditional routes often incur hidden expenses related to the neutralization of acidic waste streams and the safe handling of hazardous volatiles. By transitioning to a catalytic system with recyclable palladium, manufacturers can achieve substantial cost savings over the lifecycle of the product. Furthermore, the use of commodity chemicals like p-bromophenol ensures that raw material sourcing remains stable, insulating the production schedule from the supply shocks that frequently affect specialty reagents. This reliability is paramount for maintaining continuous manufacturing lines and meeting delivery commitments to downstream API producers.
- Cost Reduction in Manufacturing: The shift from stoichiometric reagents to catalytic processes fundamentally alters the cost structure of production. By utilizing low loadings of palladium catalysts that can potentially be recovered, the direct material cost per kilogram of product is optimized. Additionally, the simplified purification steps reduce solvent consumption and labor hours associated with complex extractions and chromatography. The avoidance of expensive and hazardous reagents like bromine further lowers the operational expenditure, making the overall process economically superior to legacy methods that rely on heavy metal waste generation.
- Enhanced Supply Chain Reliability: Sourcing raw materials for this synthesis is straightforward, as p-bromophenol and methyl vinyl ether are widely produced industrial chemicals with established supply networks. This contrasts sharply with routes requiring niche acylating agents or unstable diazonium precursors. The robustness of the supply chain is further enhanced by the process tolerance; since the intermediate does not require high-purity isolation before the second step, variations in upstream quality have less impact on the final output. This resilience ensures that production timelines remain intact even when facing minor fluctuations in raw material specifications, guaranteeing on-time delivery for clients.
- Scalability and Environmental Compliance: The mild reaction conditions and absence of highly exothermic steps make this process inherently safer and easier to scale from kilogram to tonne levels. Regulatory bodies increasingly favor processes that minimize E-factor (mass of waste per mass of product), and this catalytic route excels in that metric by avoiding massive salt waste from Lewis acid complexes. The reduced environmental footprint simplifies the permitting process for new manufacturing facilities and aligns with corporate sustainability goals. Consequently, scaling up this technology presents fewer engineering challenges and regulatory hurdles compared to expanding capacity for traditional nitration or Friedel-Crafts processes.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is vital for stakeholders evaluating its implementation. The following questions address common concerns regarding catalyst performance, safety, and product quality. These insights are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for decision-making. Whether you are assessing the feasibility of technology transfer or evaluating vendor capabilities, these answers clarify the operational realities of this advanced manufacturing route.
Q: What are the primary advantages of this Pd-catalyzed route over traditional Friedel-Crafts acylation?
A: The novel route eliminates the need for stoichiometric amounts of aluminum trichloride, significantly reducing hazardous waste generation and simplifying post-treatment purification compared to traditional acylation methods.
Q: Is the palladium catalyst recoverable in this industrial process?
A: Yes, the patent indicates that while palladium catalysts have a higher initial cost, their usage is catalytic (low molar percentage) and they can be recycled in industrial production, leading to long-term cost optimization.
Q: How does this method improve impurity control compared to nitration routes?
A: By avoiding nitration and diazotization steps, this method prevents the formation of ortho-substituted byproducts and eliminates the safety risks associated with handling explosive diazonium salts, ensuring a cleaner impurity profile.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-(2-Methoxyethyl)phenol Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthetic routes requires a partner with deep technical expertise and proven execution capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patent CN109553513B are fully realized in practice. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 4-(2-methoxyethyl)phenol meets the exacting standards required for cardiovascular drug synthesis. Our commitment to quality assurance means that clients can rely on us for consistent supply without compromising on the chemical integrity of the intermediate.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can quantify the economic benefits of switching to this catalytic route for your specific volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project needs. Let us collaborate to optimize your production of Metoprolol intermediates, driving efficiency and value across your entire pharmaceutical manufacturing operation.
