Scalable Production of N-Methyl Glutamine Derivatives for Advanced Peptide Therapeutics
Scalable Production of N-Methyl Glutamine Derivatives for Advanced Peptide Therapeutics
The pharmaceutical industry's relentless pursuit of potent antiviral and cytotoxic agents has placed significant focus on the Callipeltin family of marine natural products. Specifically, the synthesis of complex depsipeptides like Callipeltin E requires highly specialized, non-proteinogenic amino acid building blocks that are often difficult to source commercially. Patent CN112174854A, published in early 2021, addresses this critical bottleneck by disclosing a robust, solution-phase preparation method for (S)-2-((9H-fluorene-9-methoxycarbonyl)methylamino)-5-amino-5-oxo pentanoic acid. This compound serves as a pivotal intermediate for constructing the unique peptide backbone found in Callipeltins, which have demonstrated promising anti-HIV and anti-fungal activities. By shifting away from traditional, resource-intensive methodologies, this innovation offers a streamlined pathway that aligns perfectly with modern green chemistry principles and cost-efficiency mandates.
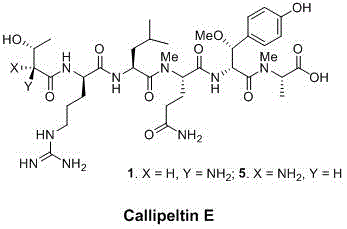
For R&D directors evaluating synthetic routes, understanding the structural complexity of the target molecule is paramount. As illustrated in the structure of Callipeltin E, the presence of N-methylated residues and specific stereochemistry demands precise control during intermediate synthesis. The disclosed process ensures the retention of chirality while introducing the necessary N-methyl group through a clever cascade reaction, thereby providing a reliable supply of high-purity pharmaceutical intermediates essential for downstream drug discovery programs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of modified amino acids for peptides like Callipeltins has relied heavily on solid-phase peptide synthesis (SPPS) or multi-step solution routes involving expensive chiral pool starting materials. As depicted in prior art references, conventional approaches often utilize protected amino acids attached to insoluble polymer resins. While effective for small-scale library generation, these methods suffer from severe limitations when scaled for commercial supply. The requirement for vast excesses of coupling reagents, the high cost of functionalized resins, and the substantial solvent consumption create an economic burden that is unsustainable for large-scale manufacturing. Furthermore, the cleavage steps often generate complex mixtures requiring extensive purification, leading to lower overall yields and increased environmental waste.
The Novel Approach
In stark contrast, the methodology presented in CN112174854A leverages a solution-phase strategy that begins with L-glutamic acid, one of the most abundant and cost-effective chiral building blocks available in the fine chemical market. This novel approach circumvents the need for solid supports entirely, utilizing a sequence of protection, cyclization, and reductive functionalization that can be performed in standard glass-lined reactors. By employing paraformaldehyde as both a protecting group precursor and a methyl source, the process achieves atom economy that is rarely seen in traditional peptide chemistry. This shift not only drastically simplifies the operational workflow but also transforms the cost structure of producing these high-value intermediates, making them accessible for broader therapeutic applications.
Mechanistic Insights into Oxazolidine-Mediated Reductive Methylation
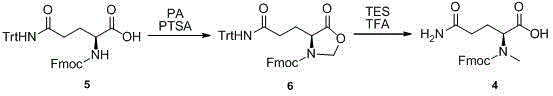
The core chemical innovation of this process lies in the formation and subsequent transformation of a cyclic oxazolidine intermediate. In the second step, the Fmoc-protected glutamic acid reacts with paraformaldehyde under acidic catalysis (PTSA) in refluxing toluene. This dehydration condensation results in the formation of a five-membered oxazolidine ring (Compound 2), which effectively masks both the alpha-amino group and the gamma-carboxylic acid functionality. This dual protection strategy is mechanistically elegant because it prevents unwanted side reactions during the subsequent amidation step while activating the system for the final transformation. The rigidity of the cyclic structure also helps in maintaining the stereochemical integrity of the alpha-carbon, ensuring the final product meets the stringent enantiomeric purity requirements of the pharmaceutical industry.
The final step involves a sophisticated reductive cleavage using triethylsilane (TES) and trifluoroacetic acid (TFA). Mechanistically, the acidic conditions open the oxazolidine ring, generating an iminium ion intermediate in situ. The hydride source (TES) then selectively reduces this iminium species, resulting in the introduction of the methyl group onto the nitrogen atom while simultaneously liberating the free carboxylic acid and amide functionalities. This one-pot reductive methylation/deprotection sequence is highly efficient, bypassing the need for isolated alkylation steps which often suffer from over-alkylation or racemization. The result is a clean conversion to the target N-methyl glutamine derivative with reported HPLC purity exceeding 98%, demonstrating the robustness of this mechanistic design.
How to Synthesize (S)-2-((9H-fluorene-9-methoxycarbonyl)methylamino)-5-amino-5-oxopentanoic acid Efficiently
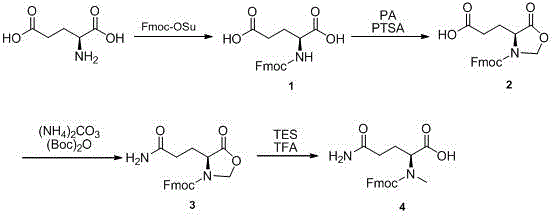
The synthesis protocol outlined in the patent provides a clear, four-step roadmap that is amenable to scale-up without requiring specialized equipment beyond standard chemical processing infrastructure. The process begins with the aqueous protection of L-glutamic acid, followed by an azeotropic dehydration to drive the cyclization. The subsequent amidation is performed under mild conditions using ammonium carbonate, avoiding the use of hazardous ammonia gas. Finally, the reductive workup yields the crude product which can be purified via simple crystallization or minimal chromatography. For detailed operational parameters, stoichiometry, and safety considerations, please refer to the standardized synthesis guide below.
- Protect L-glutamic acid with Fmoc-OSu in sodium bicarbonate solution to form Compound 1.
- React Compound 1 with paraformaldehyde and PTSA in refluxing toluene to generate the cyclic oxazolidine Compound 2.
- Perform acylation on Compound 2 using ammonium carbonate and di-tert-butyl dicarbonate to yield Compound 3.
- Treat Compound 3 with triethylsilane and trifluoroacetic acid in dichloromethane to afford the final target Compound 4.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition from solid-phase to this optimized solution-phase chemistry represents a significant opportunity for cost reduction in pharmaceutical intermediate manufacturing. The elimination of expensive polymeric resins and the use of commodity chemicals like L-glutamic acid and paraformaldehyde fundamentally lowers the bill of materials. Moreover, the high yields reported in each step—ranging from 92% to 98%—accumulate to provide a superior overall process mass intensity compared to legacy methods. This efficiency translates directly into a more stable pricing model for long-term supply agreements, shielding downstream manufacturers from the volatility often associated with specialty peptide building blocks.
- Cost Reduction in Manufacturing: The process eliminates the need for costly chromatographic purification of intermediates, a step that typically consumes significant resources and time in peptide synthesis. By relying on crystallization and extraction for isolation, the method reduces solvent usage and labor costs substantially. Additionally, the avoidance of transition metal catalysts means there is no need for expensive metal scavenging steps or rigorous testing for heavy metal residues, further streamlining the quality control budget and reducing the total cost of ownership for the intermediate.
- Enhanced Supply Chain Reliability: Sourcing L-glutamic acid is far more secure than sourcing specialized protected amino acids or custom resins, as it is a bulk fermentation product with a global supply base. This raw material availability ensures that production schedules are not disrupted by vendor shortages. The robustness of the reaction conditions, which tolerate room temperature operations for several steps, also reduces energy consumption and equipment wear, contributing to a more reliable and continuous manufacturing output that can meet the demanding lead times of clinical and commercial projects.
- Scalability and Environmental Compliance: The synthesis avoids the generation of hazardous waste streams associated with resin cleavage cocktails. The solvents used, such as ethyl acetate and toluene, are well-understood and easily recycled in modern facilities. The high atom economy of the reductive methylation step minimizes waste generation per kilogram of product. This alignment with green chemistry principles facilitates easier regulatory approval and environmental permitting, allowing for seamless commercial scale-up of complex pharmaceutical intermediates from pilot plants to multi-ton production facilities without significant process re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, aiming to clarify the feasibility and advantages for potential partners and licensees.
Q: What are the primary advantages of this synthesis route over solid-phase methods?
A: This solution-phase method utilizes inexpensive L-glutamic acid as a starting material and eliminates the need for expensive resin supports and large volumes of solvents typically associated with solid-phase synthesis, significantly reducing raw material costs and waste generation.
Q: Does the process require column chromatography for purification?
A: No, a key benefit highlighted in the patent is that the intermediate products do not require purification by chromatographic columns, which simplifies the workflow and enhances scalability for industrial production.
Q: What is the chemical role of triethylsilane in the final step?
A: Triethylsilane acts as a reducing agent in the presence of trifluoroacetic acid to facilitate the reductive methylation and deprotection sequence, converting the cyclic intermediate directly into the N-methylated target amino acid.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (S)-2-((9H-fluorene-9-methoxycarbonyl)methylamino)-5-amino-5-oxopentanoic acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of next-generation antiviral therapeutics. Our technical team has thoroughly analyzed the route disclosed in CN112174854A and validated its potential for industrial application. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and consistency. Our facility is equipped with stringent purity specifications and rigorous QC labs capable of verifying the structural integrity and enantiomeric excess of every batch, guaranteeing that the material you receive is ready for immediate use in sensitive peptide coupling reactions.
We invite you to collaborate with us to leverage this advanced technology for your pipeline. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can accelerate your timeline and optimize your budget for Callipeltin-based drug development programs.
