Advanced Synthesis of Bis-Aminopropyl Alicyclic Tertiary Amines for High-Performance Epoxy Curing Applications
The chemical industry is currently witnessing a paradigm shift in the synthesis of high-performance epoxy curing agents, driven by the urgent need for materials that offer superior thermal stability and mechanical strength. Patent CN114853633B introduces a groundbreaking preparation method for bis-aminopropyl alicyclic tertiary amines, a critical class of compounds that serve as essential intermediates in the formulation of advanced polymer systems. This technology addresses the longstanding limitations of traditional alicyclic amine modification, which often suffer from incomplete reactions and significant environmental burdens due to excessive wastewater generation. By implementing a sophisticated two-step nitrile ethylation strategy coupled with an innovative mother liquor recycling and washing protocol, this method achieves a target product yield of ≥93% while drastically simplifying the downstream purification process. For R&D Directors and Procurement Managers seeking a reliable epoxy curing agent intermediate supplier, understanding the mechanistic advantages of this patent is crucial for optimizing supply chain resilience and reducing overall manufacturing costs in the competitive fine chemical sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the modification of alicyclic amines to enhance their performance in epoxy resin applications has been plagued by significant technical and environmental inefficiencies. Conventional methods, such as those disclosed in US 4321354, typically rely on the use of acetic acid as a catalyst for the addition of acrylonitrile to diamines. While this approach can improve reaction rates, it necessitates a subsequent neutralization step using large quantities of alkali solution to remove unreacted acid, resulting in the production of substantial volumes of saline wastewater that pose severe environmental disposal challenges. Furthermore, traditional one-step synthesis routes often struggle with the steric hindrance effects inherent in secondary amines, leading to low conversion rates for the desired tertiary amine products and necessitating complex, energy-intensive purification steps to remove unreacted starting materials and by-products. These inefficiencies not only inflate the cost reduction in fine chemical manufacturing but also create bottlenecks in the commercial scale-up of complex amine intermediates, making it difficult for producers to meet the stringent quality and sustainability standards demanded by high-end applications such as wind turbine blades and marine protective coatings.
The Novel Approach
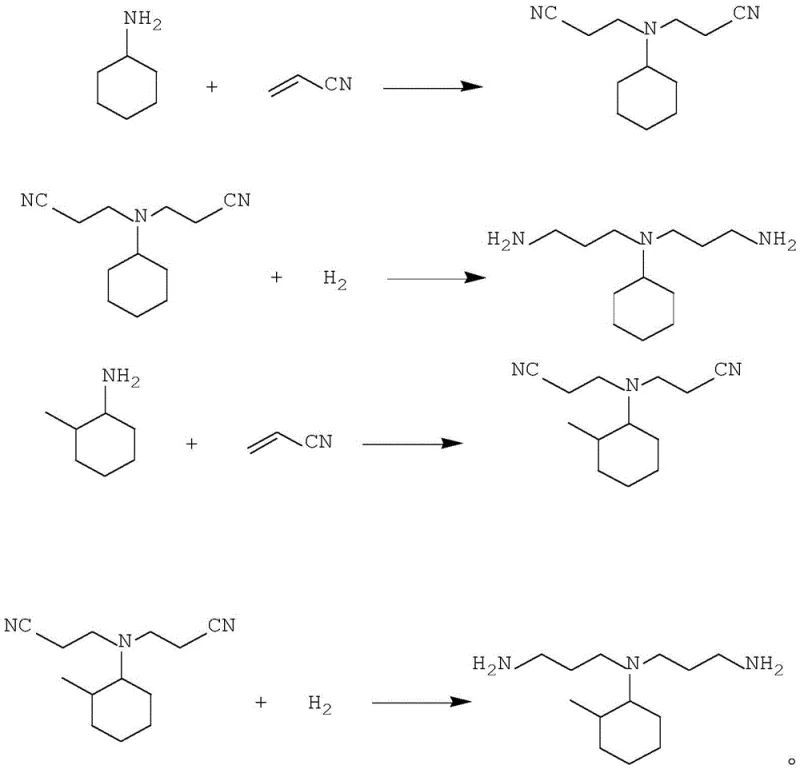
In stark contrast to these legacy processes, the novel approach detailed in CN114853633B employs a strategic two-step nitrile ethylation sequence that fundamentally alters the reaction kinetics and separation efficiency. By first converting the alicyclic primary amine into a mono-nitrile ethyl secondary amine and then subsequently reacting this intermediate with additional acrylonitrile, the process effectively bypasses the steric barriers that limit direct tertiary amine formation. The most transformative aspect of this methodology is the utilization of the mono-nitrile ethyl secondary amine mother liquor, specifically fraction B, to wash the bis-nitrile ethyl tertiary amine mother liquor. This ingenious phase separation technique allows for the thorough removal of residual acid catalysts and water-soluble impurities without the need for external alkali neutralization, thereby eliminating the generation of saline wastewater entirely. This streamlined workflow not only enhances the purity of the intermediate bis-nitrile ethyl product but also facilitates the recycling of acid-carrying mother liquors back into the reaction system, significantly improving atom economy and reducing the consumption of raw materials and auxiliaries in large-scale production environments.
Mechanistic Insights into Nitrile Ethylation and Hydrogenation
The core chemical innovation lies in the precise control of the nitrile ethylation reaction conditions and the subsequent phase behavior of the reaction mixture. In the first step, the alicyclic primary amine reacts with acrylonitrile in the presence of a minimal amount of water catalyst at temperatures ranging from 20°C to 80°C, favoring the formation of the mono-adduct. The reaction mixture is then strategically divided, with a portion reserved for the washing step and the remainder proceeding to the second ethylation stage where an acidic catalyst such as glycolic acid or hydrochloric acid is introduced to drive the formation of the bis-nitrile ethyl tertiary amine. The critical mechanistic advantage is observed during the washing phase, where the reserved mother liquor acts as a selective extraction solvent, pulling residual acid and polar impurities into the lower aqueous phase while leaving the organic tertiary amine product in the upper layer. This separation is so effective that the catalyst content in the organic phase can be reduced to below 1000 ppm, and preferably below 100 ppm, prior to the final hydrogenation step, which is vital for protecting the activity of the sensitive hydrogenation catalysts used in the subsequent stage.
Following the purification of the bis-nitrile ethyl intermediate via distillation to remove light components, the final transformation involves catalytic hydrogenation to convert the nitrile groups into primary amine functionalities. This step typically employs Raney series catalysts, such as Raney nickel or Raney cobalt, under hydrogen pressures of 1 MPa to 10 MPa and temperatures between 50°C and 150°C. The high purity of the feedstock entering this stage, ensured by the preceding washing and distillation steps, is paramount for maintaining catalyst longevity and achieving high conversion rates. The resulting bis-aminopropyl alicyclic tertiary amine exhibits a purity profile where the target product content can exceed 94%, with minimal residual primary or secondary amine impurities. This level of control over the impurity spectrum is essential for R&D teams focused on developing high-purity epoxy curing agents that require consistent reactivity and curing profiles. The integration of these reaction steps into a cohesive, continuous-friendly process demonstrates a sophisticated understanding of process chemistry that translates directly into operational reliability and product consistency.
How to Synthesize Bis-Aminopropyl Alicyclic Tertiary Amine Efficiently
The synthesis of this high-value intermediate requires precise adherence to the patented two-step protocol to ensure optimal yield and purity. The process begins with the controlled addition of acrylonitrile to the alicyclic primary amine, followed by the critical separation and recycling of mother liquors to maximize resource efficiency. Detailed standard operating procedures regarding specific molar ratios, temperature ramps, and pressure controls are essential for replicating the high yields reported in the patent data. For technical teams looking to implement this route, it is crucial to understand that the efficiency of the washing step directly correlates with the performance of the final hydrogenation catalyst. This structured approach ensures that the transition from laboratory scale to industrial production is managed with the highest regard for safety and quality control standards.
- Perform stepwise nitrile ethylation of alicyclic primary amine with acrylonitrile to form mono- and bis-nitrile ethyl intermediates.
- Utilize mono-nitrile ethyl secondary amine mother liquor to wash the bis-nitrile ethyl tertiary amine mother liquor for acid removal and phase separation.
- Conduct catalytic hydrogenation of the purified bis-nitrile ethyl intermediate using Raney nickel or cobalt catalysts to yield the final tertiary amine.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented synthesis route offers profound advantages for procurement managers and supply chain heads focused on cost optimization and sustainability. The elimination of the alkali neutralization step represents a significant reduction in utility consumption and waste treatment costs, as there is no longer a need to purchase, store, and handle large quantities of caustic soda or other neutralizing agents. Furthermore, the ability to recycle the acid-carrying mother liquor back into the reaction system drastically reduces the consumption of fresh catalyst and raw materials, leading to substantial cost savings in the overall manufacturing budget. For supply chain professionals, this process enhances reliability by simplifying the production workflow and reducing the dependency on complex waste disposal infrastructure, which can often be a bottleneck in chemical manufacturing regions with strict environmental regulations. The robustness of the process also implies a more stable supply of high-purity intermediates, reducing the risk of production delays caused by purification failures or catalyst deactivation issues commonly associated with dirtier synthesis routes.
- Cost Reduction in Manufacturing: The process architecture inherently lowers production costs by removing the need for expensive alkali neutralization and the associated wastewater treatment facilities. By recycling the mother liquor containing residual acid catalyst, the consumption of fresh acidic catalysts is significantly minimized, which directly impacts the variable cost of goods sold. Additionally, the high yield of the target product means that less raw material is wasted on by-products, improving the overall atom economy of the synthesis. This efficiency translates into a more competitive pricing structure for the final epoxy curing agent, allowing manufacturers to maintain healthy margins even in volatile raw material markets. The reduction in processing steps also lowers energy consumption related to heating, cooling, and agitation, further contributing to the overall economic viability of the production line.
- Enhanced Supply Chain Reliability: The simplified process flow reduces the number of potential failure points in the manufacturing chain, ensuring a more consistent and reliable supply of the critical amine intermediate. By avoiding the generation of hazardous saline wastewater, the facility mitigates the risk of regulatory shutdowns or fines related to environmental compliance, which can severely disrupt supply continuity. The use of readily available raw materials such as cyclohexylamine and acrylonitrile, combined with standard hydrogenation catalysts like Raney nickel, ensures that the supply chain is not dependent on exotic or scarce reagents. This accessibility allows for flexible sourcing strategies and reduces the lead time for high-purity amine intermediates, enabling downstream customers to plan their production schedules with greater confidence and accuracy.
- Scalability and Environmental Compliance: The technology is designed with industrial scale-up in mind, utilizing standard unit operations such as distillation and phase separation that are easily adaptable to large-scale reactors. The absence of saline wastewater generation aligns perfectly with increasingly stringent global environmental regulations, making the process future-proof against tightening discharge limits. This green chemistry approach not only reduces the environmental footprint of the manufacturing site but also enhances the brand value of the final product in markets that prioritize sustainability. The ability to scale from pilot plants to multi-ton production without significant process re-engineering ensures that the technology can meet growing market demand for high-performance epoxy curing agents without compromising on quality or compliance standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the patent specifications and are intended to provide clarity on the process capabilities and limitations. Understanding these details is vital for stakeholders evaluating the feasibility of integrating this intermediate into their existing supply chains. This transparency ensures that all parties have a clear understanding of the technical parameters and commercial benefits associated with this advanced manufacturing method.
Q: Why is a two-step nitrile ethylation process preferred over a one-step method for alicyclic amines?
A: A two-step process overcomes the steric hindrance associated with secondary amines, significantly improving the yield of the tertiary amine product compared to direct one-step addition which often stalls at the secondary amine stage.
Q: How does this patent address environmental concerns regarding wastewater in amine synthesis?
A: The process eliminates the need for alkali neutralization of acid catalysts by using a mother liquor washing strategy, thereby preventing the generation of saline wastewater and reducing environmental compliance costs.
Q: What catalysts are suitable for the hydrogenation step in this synthesis route?
A: Raney series catalysts, specifically Raney nickel or Raney cobalt, are preferred for the hydrogenation step due to their high activity in converting nitrile groups to primary amines under moderate pressure and temperature conditions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bis-Aminopropyl Alicyclic Tertiary Amine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of next-generation epoxy curing agents. Our team of CDMO experts possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated two-step nitrile ethylation process described in CN114853633B can be successfully translated into a robust industrial operation. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which utilize advanced analytical techniques to verify the absence of critical impurities and ensure batch-to-batch consistency. Our infrastructure is designed to handle complex amine chemistries safely and efficiently, providing our partners with a secure and reliable source of supply that supports their long-term R&D and commercialization goals.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs through the adoption of this superior synthesis route. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production volumes and quality requirements. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing you to make informed decisions about integrating this high-performance intermediate into your product portfolio. By partnering with NINGBO INNO PHARMCHEM, you gain access to not just a chemical supplier, but a strategic ally dedicated to driving innovation and efficiency in the fine chemical industry.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
