Advanced Manufacturing of Ethyl 5-n-propylpyrazole-3-carboxylate via Novel Metal Sodium Catalysis
Advanced Manufacturing of Ethyl 5-n-propylpyrazole-3-carboxylate via Novel Metal Sodium Catalysis
The pharmaceutical and agrochemical industries constantly seek robust, scalable pathways for critical heterocyclic intermediates. A pivotal advancement in this domain is documented in patent CN1264703A, which outlines a highly efficient process for synthesizing ethyl 5-n-propylpyrazole-3-carboxylate. This compound serves as a fundamental building block for pyrazolopyrimidinone drugs and exhibits potential biological activities including anti-arteriosclerosis effects. The disclosed methodology represents a significant departure from historical precedents by replacing conventional basic catalysts with metallic sodium in the initial Claisen condensation step. This strategic modification not only drives the chemical equilibrium favorably towards product formation but also simplifies downstream purification, addressing long-standing inefficiencies in the production of this valuable pharmaceutical intermediate.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical literature reveals a trajectory of suboptimal processes for generating the requisite enol sodium salt precursor. Early reports, such as those by Earl in 1945, described dropping ketone-oxalate mixtures into sodium methoxide solutions, requiring stirring for 4 to 6 hours followed by an overnight standing period. Subsequent attempts by Gruber in 1950 utilized sodium in ethanol but necessitated a staggering 24-hour reaction time to achieve a meager yield of merely 35%. Even later optimizations by Seki in 1984, which employed reflux conditions for 8 hours, only managed to elevate yields to approximately 68%. Beyond the temporal inefficiency, the subsequent cyclization step in these legacy methods often relied on benzene as an extraction solvent. Benzene is a known carcinogen with severe toxicity profiles, posing significant occupational health risks and environmental compliance burdens. Furthermore, the high boiling point of benzene often led to product discoloration during solvent removal, compromising the quality and purity of the final active pharmaceutical ingredient (API) intermediate.
The Novel Approach
The innovative protocol detailed in the patent data fundamentally re-engineers the catalytic landscape of the Claisen condensation. By introducing metallic sodium directly into the reaction matrix containing diethyl oxalate and 2-pentanone, the process leverages the trace ethanol present in the ester to generate sodium ethoxide in situ. This generated alkoxide initiates the condensation, and crucially, the continuous consumption of the byproduct ethanol by the excess metallic sodium prevents the reverse reaction, effectively locking the equilibrium in the forward direction. This mechanistic shift allows the first step to complete in just 0.5 to 1.5 hours with excellent crystallization properties. In the second stage, the cyclization with hydrazine hydrate is accelerated to completion within 1 hour. The substitution of benzene with dichloromethane (DCM) for extraction further refines the process; DCM's lower boiling point facilitates gentle solvent recovery, preserving the pristine white needle-like morphology of the final crystals and ensuring a total process time of merely 3 to 4 hours.
Mechanistic Insights into Metal Sodium Catalyzed Claisen Condensation
The core innovation lies in the dynamic interplay between the metallic sodium and the reaction byproducts. In a standard base-catalyzed Claisen condensation, the accumulation of alcohol can drive the equilibrium backward, limiting conversion. However, in this system, the metallic sodium acts as a scavenger for the ethanol produced during the ester-enolate transformation. As the reaction proceeds, the metal reacts with the generated ethanol to form additional sodium ethoxide and hydrogen gas. This dual function—consuming the inhibitory byproduct while replenishing the catalytic species—creates a self-reinforcing cycle that drives the reaction to near-completion rapidly. The resulting 1-ethoxycarbonyl-3-oxohexenol sodium salt precipitates readily from the solution, a physical property that greatly aids in isolation and purity enhancement without the need for complex workup procedures typically associated with liquid-liquid extractions at this stage.
Following the isolation of the sodium salt, the cyclization mechanism proceeds through a nucleophilic attack by hydrazine on the electrophilic carbonyl centers. The presence of the electron-withdrawing ester group on the sodium salt significantly increases the positive charge density on the alpha and gamma carbons, rendering them highly susceptible to nucleophilic substitution and addition-elimination sequences. The formation of the stable five-membered pyrazole ring is thermodynamically favorable and kinetically rapid, confirmed by tracking studies showing full conversion within 60 minutes. The use of glacial acetic acid serves to neutralize the sodium salt gently, releasing the free beta-keto ester which immediately undergoes condensation with hydrazine. This seamless transition minimizes the exposure of reactive intermediates to harsh conditions, thereby suppressing the formation of polymeric byproducts or degradation species that often plague heterocyclic synthesis.
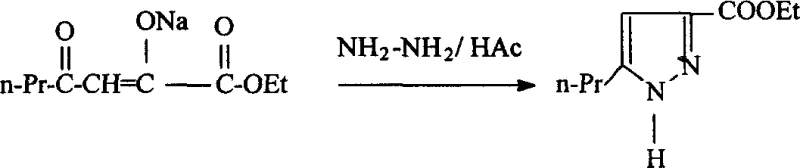
How to Synthesize Ethyl 5-n-propylpyrazole-3-carboxylate Efficiently
Implementing this synthesis requires precise control over stoichiometry and addition rates to manage the exothermic nature of the sodium reaction. The molar ratio of metallic sodium to 2-pentanone to diethyl oxalate is optimized at 1:(1~1.2):(1~1.5). The process begins with the addition of metallic sodium to the ester, followed by the slow滴加 (dropwise addition) of the ketone to maintain a gentle boil, ensuring safe hydrogen evolution. Once the sodium salt is isolated and purified via ethanol recrystallization, it is subjected to cyclization using an 85% hydrazine hydrate solution in the presence of acetic acid. The detailed operational parameters, including temperature ramps and specific workup protocols necessary for GMP compliance, are outlined in the technical guide below.
- Perform Claisen condensation of 2-pentanone and diethyl oxalate using metallic sodium as a catalyst substitute to generate the enol sodium salt intermediate.
- React the isolated sodium salt with hydrazine hydrate and glacial acetic acid under reflux conditions to effect ring closure.
- Purify the crude product via dichloromethane extraction and recrystallization from ethanol to obtain high-purity white needle-like crystals.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this novel synthetic route offers profound strategic benefits beyond mere chemical elegance. The drastic reduction in cycle time from multiple days to under 4 hours translates directly into increased asset turnover and throughput capacity. Facilities can produce significantly more batches per month using the same reactor volume, effectively expanding capacity without capital expenditure on new equipment. Furthermore, the elimination of benzene removes a major regulatory hurdle and reduces the costs associated with hazardous waste disposal and worker safety monitoring. The improved crystallization behavior of the intermediate ensures consistent particle size distribution and purity, reducing the risk of batch failures and reprocessing costs that often erode profit margins in fine chemical manufacturing.
- Cost Reduction in Manufacturing: The replacement of expensive or inefficient catalyst systems with commodity-grade metallic sodium significantly lowers raw material costs. Additionally, the high yield exceeding 71% in the final step and over 60% overall means less starting material is wasted per kilogram of product. The energy consumption is also drastically reduced due to the shorter reaction times and lower boiling point of the extraction solvent, leading to substantial utility savings. By eliminating the need for prolonged reflux and overnight standing, the process optimizes labor utilization and reactor occupancy, driving down the fully loaded cost of goods sold (COGS) for this critical intermediate.
- Enhanced Supply Chain Reliability: Shorter manufacturing cycles inherently reduce lead times, allowing suppliers to respond more agilely to fluctuating market demands. The robustness of the reaction, evidenced by its tolerance and high conversion rates, minimizes the risk of supply disruptions caused by failed batches. The use of common, readily available solvents like dichloromethane and ethanol ensures that the supply chain is not vulnerable to shortages of exotic or highly regulated reagents. This reliability is crucial for downstream pharmaceutical manufacturers who require just-in-time delivery to maintain their own production schedules for finished dosage forms.
- Scalability and Environmental Compliance: The process is explicitly designed for industrialization, with features such as easy filtration of the solid sodium salt intermediate facilitating scale-up from pilot to commercial plants. The avoidance of benzene aligns with increasingly stringent global environmental regulations regarding volatile organic compounds (VOCs) and carcinogen usage. The cleaner reaction profile results in a simpler impurity spectrum, easing the burden on quality control laboratories and accelerating the release of materials for clinical or commercial use. This environmental and operational efficiency positions the manufacturer as a sustainable partner in the global pharmaceutical supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthesis pathway. Understanding these nuances is essential for technology transfer teams evaluating the feasibility of adopting this process for large-scale production. The answers are derived directly from the experimental data and mechanistic insights provided in the patent documentation, ensuring accuracy and relevance for process chemists.
Q: How does the metal sodium method improve yield compared to traditional alkoxide catalysts?
A: By utilizing metallic sodium directly, the reaction consumes generated ethanol in situ to form sodium ethoxide, shifting the equilibrium to the right and preventing reverse reactions, thereby increasing yield from historical lows of 35% to over 60%.
Q: What are the safety advantages regarding solvent selection in this process?
A: The process replaces toxic benzene, traditionally used for extraction, with dichloromethane (DCM). DCM has a lower boiling point, reducing thermal stress during solvent removal and minimizing product discoloration.
Q: What is the total processing time for this synthesis route?
A: The entire synthetic sequence, including both the condensation and cyclization steps, can be completed within 3 to 4 hours, significantly faster than the 24+ hour cycles of prior art methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ethyl 5-n-propylpyrazole-3-carboxylate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory patent to commercial reality requires more than just a recipe; it demands deep process engineering expertise. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the efficiencies demonstrated in patent CN1264703A are fully realized at an industrial level. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of ethyl 5-n-propylpyrazole-3-carboxylate meets the exacting standards required for API synthesis. Our commitment to quality ensures that the impurity profiles remain tightly controlled, safeguarding the integrity of your downstream drug products.
We invite pharmaceutical and agrochemical companies to leverage our technical capabilities for their supply chain needs. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical constraints. We encourage potential partners to contact our technical procurement team to request specific COA data and route feasibility assessments. Let us demonstrate how our optimized manufacturing processes can enhance your supply chain resilience and drive value for your organization through superior chemical solutions.
