Advanced Synthesis of 6-Methylene-17α-Hydroxyprogesterone Acetate for Commercial Scale-Up
Advanced Synthesis of 6-Methylene-17α-Hydroxyprogesterone Acetate for Commercial Scale-Up
The pharmaceutical industry continuously demands more efficient and cost-effective pathways for the production of critical steroid intermediates, particularly those serving as precursors for widely used hormonal therapies. Patent CN106977569B, published in June 2020, introduces a robust and innovative preparation method for 6-methylene-17α-hydroxyprogesterone acetate, a pivotal intermediate in the synthesis of Medroxyprogesterone acetate and Megestrol acetate. This novel methodology addresses long-standing challenges in steroid chemistry by streamlining the introduction of the 6-methylene group through a sophisticated sequence of acylation, Mannich reaction, and elimination. For R&D directors and procurement specialists, this technology represents a significant leap forward, offering a route that not only enhances product purity but also drastically simplifies the operational complexity associated with traditional synthesis protocols. By leveraging mild reaction conditions and readily available reagents, this process ensures a stable supply chain for high-value hormonal drugs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 6-methylene steroids has been plagued by inefficient multi-step sequences that rely on harsh conditions and expensive reagents. Prior art, such as the methods disclosed in US200912321 and US4322349, often necessitates the use of ethylene glycol dimethyl ether and triethyl orthoformate for etherification, followed by complex extraction and hydrolysis steps. These conventional routes frequently suffer from incomplete reactions, low yields ranging significantly below optimal levels, and the generation of difficult-to-remove impurities that compromise the final API quality. Furthermore, the reliance on specific pH adjustments and multiple solvent exchanges increases the environmental footprint and operational costs, creating bottlenecks for large-scale manufacturing. The need for rigorous purification techniques, such as activated carbon decolorization and repeated washing with specialized solvents like DMF, further exacerbates the production timeline and resource consumption.
The Novel Approach
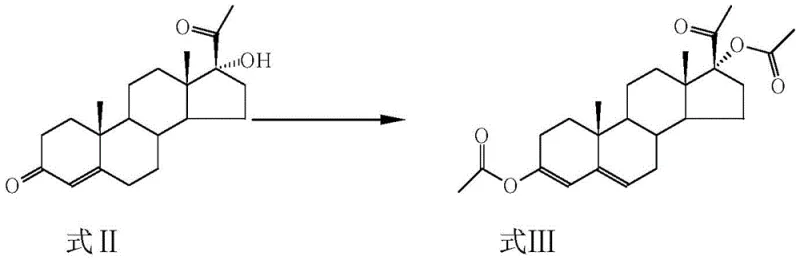
In stark contrast, the methodology outlined in CN106977569B employs a direct and elegant strategy that begins with the acylation of 17α-hydroxyprogesterone, effectively activating the molecule for subsequent functionalization. This initial step utilizes acetic anhydride not merely as a reactant but also as a solvent component, which simplifies the reaction matrix and enhances the efficiency of the 17-hydroxyl protection. The process seamlessly transitions into a Mannich reaction under weakly acidic conditions, allowing for the precise introduction of the aminomethyl group at the 6-position without the need for aggressive etherification agents. This streamlined approach minimizes the number of unit operations and eliminates the requirement for complex workup procedures associated with older patents. By integrating these steps into a cohesive workflow, the novel method achieves superior conversion rates and facilitates easier isolation of the intermediate, setting a new standard for steroid intermediate manufacturing.
Mechanistic Insights into the Acylation-Mannich-Elimination Sequence
The core innovation of this synthesis lies in the strategic activation of the steroid backbone to facilitate regioselective functionalization at the C6 position. The process initiates with the acylation of the 17α-hydroxyl group using acetic anhydride in the presence of catalytic amounts of strong acids such as sulfuric acid or p-toluenesulfonic acid. This transformation is critical as it stabilizes the D-ring and modifies the electronic environment of the molecule, rendering the C6 hydrogen atoms more susceptible to electrophilic attack. The reaction is typically conducted at temperatures ranging from -10°C to 60°C, allowing for precise control over the reaction kinetics and preventing unwanted side reactions at other sensitive sites on the steroid nucleus. The use of solvents like tetrahydrofuran or dichloromethane further aids in maintaining a homogeneous reaction mixture, ensuring consistent heat transfer and mass transport throughout the vessel.
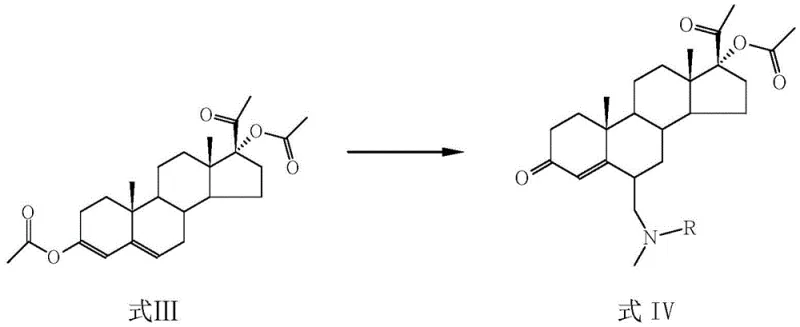
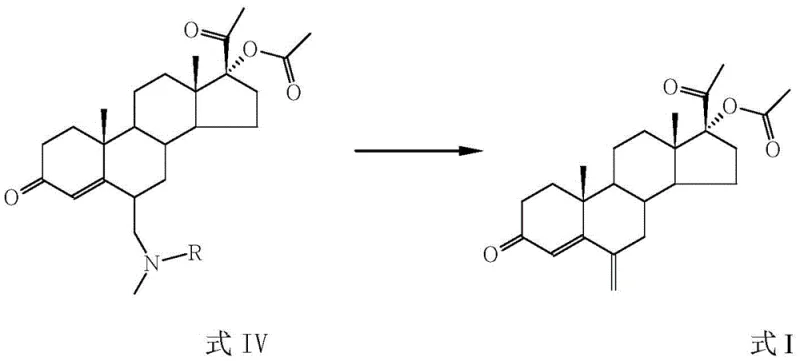
Following acylation, the intermediate undergoes a Mannich reaction with a reagent generated from dimethylamine or N-methylaniline and formaldehyde or trioxymethylene. This step introduces a tertiary amine moiety at the 6-position, forming a stable quaternary precursor that is essential for the final elimination step. The reaction proceeds efficiently within a temperature window of 30-60°C, where the weak acid atmosphere promotes the formation of the iminium ion intermediate necessary for the C-C bond formation. Subsequently, the tertiary amine intermediate is subjected to acid-mediated hydrolysis and elimination, often referred to as a Hofmann-type degradation, using mineral acids like hydrochloric or sulfuric acid. This final transformation cleaves the C-N bond and generates the exocyclic methylene double bond, yielding the target 6-methylene-17α-hydroxyprogesterone acetate with high stereochemical integrity and purity.
How to Synthesize 6-Methylene-17α-Hydroxyprogesterone Acetate Efficiently
Implementing this synthesis route requires careful attention to reagent stoichiometry and temperature control to maximize yield and minimize impurity profiles. The process is designed to be flexible, accommodating various acid catalysts and solvents while maintaining robust performance across different scales. Operators should ensure that the acylation step reaches completion before introducing the Mannich reagents to prevent competitive reactions. The final elimination step is particularly sensitive to acid concentration and temperature, requiring precise monitoring to avoid over-degradation of the steroid skeleton. For detailed operational parameters and safety guidelines, please refer to the standardized synthesis protocol provided below.
- Perform acylation of 17α-hydroxyprogesterone using acetic anhydride and an acid catalyst to form the 17-acetate intermediate.
- Conduct a Mannich reaction on the acetate intermediate using a formaldehyde-amine reagent to introduce the aminomethyl group at the 6-position.
- Execute an acid-mediated elimination (Hofmann degradation) to convert the tertiary amine into the final 6-methylene product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented process offers transformative benefits for procurement managers and supply chain leaders seeking to optimize their sourcing strategies for steroid intermediates. By eliminating the need for costly and specialized reagents like triethyl orthoformate, the method significantly reduces the raw material expenditure associated with each production batch. The simplification of the post-reaction workup, which avoids complex extraction and decolorization steps, translates directly into reduced labor costs and shorter cycle times, thereby enhancing the overall throughput of the manufacturing facility. Furthermore, the use of common industrial solvents and catalysts ensures that the supply chain remains resilient against fluctuations in the availability of niche chemicals, providing a stable foundation for long-term production planning.
- Cost Reduction in Manufacturing: The substitution of expensive etherifying agents with ubiquitous acetic anhydride and the reduction of solvent usage lead to a substantial decrease in the cost of goods sold. The streamlined process flow minimizes waste generation and energy consumption, contributing to a leaner and more economically viable production model that maximizes profit margins without compromising on quality standards.
- Enhanced Supply Chain Reliability: Utilizing widely available starting materials such as 17α-hydroxyprogesterone and common mineral acids mitigates the risk of supply disruptions caused by geopolitical or logistical issues. The robustness of the reaction conditions allows for consistent production schedules, ensuring that downstream API manufacturers receive their critical intermediates on time and in the required quantities to meet market demand.
- Scalability and Environmental Compliance: The mild reaction temperatures and the absence of heavy metal catalysts make this process inherently safer and easier to scale from pilot batches to multi-ton commercial production. The reduced generation of hazardous waste aligns with increasingly stringent environmental regulations, lowering the compliance burden and disposal costs for manufacturing sites while promoting sustainable chemical practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a clear understanding of the method's capabilities and limitations. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this route for their specific manufacturing needs.
Q: What are the key advantages of this synthesis route over traditional methods?
A: This method avoids the use of expensive etherifying agents like triethyl orthoformate and simplifies the purification process, leading to higher overall yields and reduced production costs compared to older patents like US4322349.
Q: What are the critical reaction conditions for the Mannich step?
A: The Mannich reaction is optimally conducted at temperatures between 30-60°C using solvents such as tetrahydrofuran or dichloromethane, ensuring complete conversion while minimizing byproduct formation.
Q: Is this process scalable for industrial manufacturing?
A: Yes, the process utilizes common industrial solvents and mild temperature ranges (-10 to 70°C), making it highly suitable for commercial scale-up from pilot plants to multi-ton production facilities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 6-Methylene-17α-Hydroxyprogesterone Acetate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving hormonal therapies. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous demands of global pharmaceutical clients. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that employ state-of-the-art analytical techniques to verify every batch. Our expertise in steroid chemistry allows us to navigate the complexities of this synthesis route, guaranteeing a consistent supply of 6-methylene-17α-hydroxyprogesterone acetate that meets the highest industry standards.
We invite you to collaborate with us to explore how this advanced synthesis method can optimize your supply chain and reduce your overall manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to support your next project, ensuring a seamless transition from development to commercial success.
