Advanced One-Pot Synthesis of R-Propylene Carbonate for Scalable Pharmaceutical Manufacturing
Advanced One-Pot Synthesis of R-Propylene Carbonate for Scalable Pharmaceutical Manufacturing
The pharmaceutical industry continuously seeks robust, scalable, and environmentally sustainable pathways for producing critical chiral intermediates, particularly those serving as the backbone for antiviral therapies. A pivotal advancement in this domain is detailed in Chinese Patent CN111875577B, which discloses a highly efficient preparation method for R-propylene carbonate, a key precursor in the synthesis of Tenofovir Disoproxil Fumarate, a widely used medication for treating HIV and Hepatitis B. This patent introduces a novel catalytic system utilizing an S-salen Co(II) complex that enables a one-pot kinetic resolution and carbonation process. Unlike traditional multi-step syntheses that suffer from yield losses and excessive waste generation, this innovative approach achieves exceptional stereochemical control, delivering products with content and chiral E.E. values exceeding 99.5%. For global procurement leaders and R&D directors, this technology represents a significant leap forward in process intensification, offering a streamlined route that minimizes operational complexity while maximizing the recovery of valuable chiral materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of R-propylene carbonate has been plagued by inefficiencies inherent in multi-step synthetic routes. Conventional methodologies typically rely on a two-step process where propylene oxide first undergoes kinetic resolution to yield R-propylene glycol, followed by a transesterification reaction with dimethyl or ethyl carbonate. This traditional pathway is not only operationally cumbersome but also economically burdensome due to the generation of stoichiometric amounts of byproducts such as methanol or ethanol and waste alkali. Furthermore, the ester exchange step often induces partial racemization, limiting the final chiral purity of the R-propylene carbonate to approximately 97-98% e.e., which is insufficient for high-grade pharmaceutical applications without further costly purification. Alternative enzymatic or high-pressure methods have also been explored, yet they frequently require extended reaction times exceeding 30 hours or necessitate harsh conditions that compromise catalyst stability and increase energy consumption, thereby inflating the overall cost of goods sold for API manufacturers.
The Novel Approach
In stark contrast to these legacy processes, the method disclosed in patent CN111875577B revolutionizes the synthesis through a sophisticated one-pot strategy that integrates kinetic resolution and cycloaddition seamlessly. By employing a tailored S-salen Co(II) catalyst in conjunction with specific catalytic assistants C1 (inorganic iodides) and C2 (organic ammoniums), the process achieves rapid conversion rates under mild conditions. The innovation lies in the synergistic effect of the co-catalysts, which activate the cobalt center and facilitate the nucleophilic attack of carbon dioxide on the epoxide ring with high stereoselectivity. This approach eliminates the need for intermediate isolation of propylene glycol, thereby preventing the racemization issues associated with high-temperature esterification. Moreover, the reaction system is designed for circularity; the catalyst and co-catalysts can be recovered via simple filtration and distillation after the reaction, allowing them to be directly reused in subsequent batches. This closed-loop capability drastically reduces raw material consumption and aligns perfectly with modern green chemistry principles, offering a compelling value proposition for cost reduction in API manufacturing.
Mechanistic Insights into S-Salen Co(II) Catalyzed Kinetic Resolution
The core of this technological breakthrough resides in the precise orchestration of the S-salen Co(II) catalyst, which acts as a chiral Lewis acid to differentiate between the enantiomers of propylene oxide. The mechanism initiates with the activation of the cobalt center by air and the iodide co-catalyst (C1), forming a highly reactive species capable of selectively coordinating with one enantiomer of the epoxide. This selective coordination lowers the activation energy for the ring-opening of the specific enantiomer, leading to the formation of the corresponding carbonate while leaving the other enantiomer largely unreacted as S-propylene oxide. The presence of the organic ammonium co-catalyst (C2) is critical, as it serves as a nucleophilic promoter that assists in the insertion of carbon dioxide into the metal-alkoxide bond, accelerating the cyclization step. This dual-catalyst system ensures that the reaction proceeds with high turnover frequency, minimizing the residence time required to reach completion.
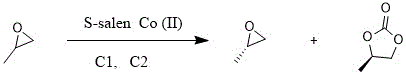
Furthermore, the structural integrity of the catalyst plays a pivotal role in maintaining high enantioselectivity throughout the reaction lifecycle. The S-salen ligand framework provides a rigid chiral environment around the cobalt ion, effectively shielding one face of the substrate and directing the incoming carbon dioxide to attack from a specific trajectory. This steric control is essential for achieving the reported >99.5% e.e. values, as even minor deviations in the catalyst geometry could lead to the formation of the undesired S-enantiomer of the carbonate. The robustness of this ligand system also contributes to the catalyst's longevity, allowing it to withstand the reaction conditions without significant degradation or leaching of the metal center.
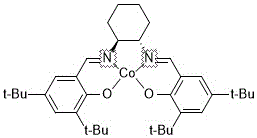
From an impurity control perspective, the mechanism inherently suppresses the formation of polymeric byproducts and oligomers that are common in uncatalyzed epoxide-CO2 copolymerization. The specific ratio of catalyst to co-catalysts, optimized at a mass ratio of roughly 500:6-8:2-4:2-4 (epoxide:catalyst:C1:C2), ensures that the active species concentration remains within an ideal window. If the catalyst loading is too low, the reaction rate diminishes, prolonging the process and increasing the risk of side reactions; conversely, excessive loading offers diminishing returns on rate acceleration while unnecessarily driving up material costs. This balanced mechanistic approach ensures that the final product profile is clean, simplifying downstream purification and ensuring that the R-propylene carbonate meets the stringent quality specifications required for antiviral drug synthesis.
How to Synthesize R-Propylene Carbonate Efficiently
Implementing this advanced synthesis route requires careful attention to the sequential addition of reagents and the control of reaction parameters to maximize yield and optical purity. The process begins with the activation of the catalyst system, followed by the controlled introduction of carbon dioxide under pressure to drive the equilibrium towards the carbonate product. The operational simplicity of this method makes it highly attractive for technology transfer teams looking to scale up production without requiring exotic equipment or hazardous reagents. The following section outlines the generalized procedural framework derived from the patent examples, providing a foundational understanding of the unit operations involved in this transformative process.
- Mix propylene oxide with S-salen Co(II) catalyst and catalytic assistant C1 (inorganic iodide), then introduce air for activation.
- Cool the mixture, add catalytic assistant C2 (organic ammonium), and introduce carbon dioxide at 0.4-0.6 MPa pressure for reaction.
- Distill the reaction mixture to recover S-propylene oxide, filter to recycle catalysts, and vacuum distill the mother liquor to obtain R-propylene carbonate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis method translates into tangible strategic advantages that extend beyond mere technical performance. The shift from a linear, waste-generating process to a circular, one-pot operation fundamentally alters the cost structure of producing this critical intermediate. By eliminating the need for separate hydrolysis and esterification reactors, manufacturers can significantly reduce capital expenditure on equipment and lower the utility costs associated with heating, cooling, and solvent recovery across multiple stages. The ability to recycle the expensive chiral cobalt catalyst and the organic co-catalysts directly back into the process creates a substantial buffer against raw material price volatility, ensuring more predictable long-term pricing for downstream API producers.
- Cost Reduction in Manufacturing: The economic impact of this process is driven primarily by the elimination of stoichiometric waste and the recovery of high-value catalysts. In traditional methods, the consumption of bases and the disposal of alcohol byproducts represent a continuous drain on operating margins. By contrast, this catalytic system operates with near-atomic efficiency regarding the carbon dioxide fixation, and the recovery of the S-salen Co(II) complex means that the cost of the chiral inducer is amortized over many batches rather than being a single-use expense. This structural change in the bill of materials allows for a significant reduction in the variable cost per kilogram of R-propylene carbonate, enhancing the competitiveness of the final antiviral medication in the global market.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the simplification of the manufacturing workflow. A one-pot process inherently reduces the number of potential failure points, such as transfer losses between reactors or delays in intermediate quality testing. The robustness of the catalyst system, which tolerates standard industrial grades of propylene oxide and carbon dioxide, reduces dependency on ultra-high-purity feedstocks that might be subject to supply constraints. Furthermore, the shortened overall cycle time, achieved by avoiding the lengthy isolation and purification steps of intermediate glycols, enables manufacturers to respond more agilely to fluctuations in market demand, ensuring a steady flow of material to API synthesis lines.
- Scalability and Environmental Compliance: As regulatory pressures regarding industrial emissions intensify globally, the "zero three-waste" claim of this process offers a distinct compliance advantage. The absence of waste alkali and organic solvent effluents simplifies the environmental permitting process and reduces the liability associated with hazardous waste disposal. From a scalability standpoint, the reaction conditions (moderate pressure and temperature) are well within the capabilities of standard stainless steel pressure vessels found in most fine chemical plants, facilitating a smooth transition from pilot scale to commercial tonnage production without the need for specialized high-pressure autoclaves or cryogenic setups.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this R-propylene carbonate synthesis technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, aiming to clarify the operational benefits and feasibility for potential licensees or manufacturing partners. Understanding these nuances is crucial for making informed decisions about process adoption and supply chain integration.
Q: What is the primary advantage of the S-salen Co(II) catalyst system in this patent?
A: The primary advantage is the ability to achieve high chiral purity (>99.5% e.e.) in a one-pot process while allowing for the complete recycling and reuse of the expensive cobalt catalyst and co-catalysts, significantly lowering production costs.
Q: How does this method compare to traditional two-step synthesis routes?
A: Traditional methods often involve a separate hydrolysis step followed by esterification, generating waste alkali and alcohols. This patented method combines kinetic resolution and carbonation in a single vessel, eliminating intermediate isolation and reducing three-waste discharge.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly states the method is suitable for industrial production due to its controllable reaction process, reasonable catalyst loading ratios, and the elimination of complex purification steps associated with byproduct removal.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable R-Propylene Carbonate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced catalytic processes requires a partner with deep technical expertise and a proven track record in process development. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN111875577B are fully realized in practical manufacturing settings. We maintain stringent purity specifications and operate rigorous QC labs equipped to verify chiral purity and trace impurities, guaranteeing that every batch of R-propylene carbonate we supply meets the exacting standards required for GMP pharmaceutical synthesis. Our commitment to quality assurance ensures that your downstream API production remains uninterrupted and compliant with global regulatory bodies.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the potential economic benefits specific to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to evaluate the superior quality and reliability of our R-propylene carbonate offerings compared to conventional market alternatives.
